Abstract
Granular cell tumors (GCTs) are rare submucosal neoplasms, with tumors in the oral mucosa accounting for about a third of all cases. In contrast, GCT is a rare anal neoplasm, with fewer than 30 cases of perianal GCT reported in the literature. We report the case of a 36-year-old woman with a perianal lump with no obvious local lesion as the main clinical complaint. The tumor was completely resected and histologically confirmed as a GCT. The patient remained under continuous follow-up. GCT is difficult for surgeons and pathologists to diagnose, and biopsy and immunohistochemical analysis are prerequisites for an accurate diagnosis. An integrated understanding of GCT in terms of its differential diagnosis will contribute to better identification and more appropriate treatment of this disease.
Keywords
Introduction
Granular cell tumors (GCTs) are rare neoplasms originating from the pituitary stalk or posterior pituitary lobe. GCTs were originally thought to originate from ectopic embryonic tissues, and were referred to as labyrinthine tumors. However, GCTs originating from embryonic myocytes were subsequently found to have a similar morphology to histiocytes, and the neoplasm was considered to be a benign myoblastoma. GCT is therefore also referred to as granulosa cell myoblastoma. 1
The relative incidence of GCT in men and women is 1:3, with a predominance in the 40- to 50-year age group. 2 It is a submucosal neoplasm that can occur in any part of the body. The most common regions are the head and neck (70%), of which 30% occur in the tongue, with other common sites being the breast (5%–15%) and the proximal limbs. 3 The slow growth of the tumor results in few clinical symptoms. Its clinical manifestations include firm-textured nodules 0.5 to 3 cm in diameter, high or not above the skin, occasionally verrucous, with unclear boundaries, and some patients may have tenderness or itching. The pathogenesis of this disease has been controversial for many years, though recent immunohistochemical studies have shown that this disease originates from the neuroectoderm, and some scholars believe that the disease originates from Schwann cells.4,5
GCT is diagnosed by pathology tests. Tumors are located in the dermis or subcutaneous tissue, and have unclear boundaries with no envelope. Tumor cells are mostly flaky and distributed between collagen bundles, and may also be streaked between collagen bundles. The tumor cells are also large and irregular, round, or oval in shape, often with multiple nuclei, and can fuse with each other, leading to unclear boundaries between cells. The cytoplasm is eosinophilic with many fine granules, and nuclear mitosis can be seen. Tumor cells stain positive for periodic acid-Schiff (PAS), S-100 protein, neuron-specific enolase (NSE), and CD68. 3 In addition, the epidermis usually shows pseudoepithelioma-like hyperplasia, which can be easily misdiagnosed as squamous cell carcinoma (SCC), and infiltration of tumor cells around nerves can sometimes be seen.
Benign GCT has a good prognosis. GCT in soft tissue can be treated with extensive local excision. However, malignant GCT often metastasizes to the regional lymph nodes and to the lung, liver, and bone, resulting in a poor prognosis, with no standardized treatment. Malignant GCT displays specific morphological and biological behaviors. Although a few early cases can be treated by simple surgical excision, a comprehensive treatment plan should generally be based on the specific anatomical location of the tumor. In this way, maximal physiological function of the affected region can be preserved, while the risks of local recurrence and distant metastasis can be reduced, with the ultimate goal of improving the cure rate.
As of 2020, fewer than 30 cases of perianal GCT had been reported in the literature. In this study, we report a rare case of perianal GCT, and consider the need to make an accurate and clear differential diagnosis in light of the rarity of benign neurogenic anal tumors. We describe the challenges for surgeons and pathologists.
Case presentation
A 36-year-old Chinese woman was hospitalized on 28 December 2018 with a complaint of anal pressing pain and an anal mass. Anorectal physical examination showed no redness or ulcers in the perianal skin. Preoperative digital anal examination showed moderate anal tightness, a smooth rectum wall, and no swelling or mass. There were also no significant changes in the rectal wall when the mass was pulled. The perianal skin and lower rectal mucosa were smooth, and no ulcers, fistulas, or purulent secretion were found under anoscopy. The tumor was palpable 6 cm from the anal margin at a 7 o'clock direction, with a range of about 5 cm × 5 cm (Figure 1). Perianal ultrasound suggested an inflammatory mass, with no significant relationship between the mass and the rectum (Figure 2). The clinical diagnosis was a perianal mass for further examination. The decision was made to perform excision and biopsy. Under general anesthesia, the perianal area was prepped and draped in a sterile manner and the anal mass was identified. Local anesthesia with 25% macaine was injected into the skin around the mass, and an elliptical incision was made around the mass, about 0.5 cm from the edge of the mass. The subcutaneous tissue was taken down by electrocautery. During surgery, the tumor was found to be spreading subcutaneously, with unclear boundaries. It had a hard texture, no capsule, and no fluctuation (indicating a solid mass) and was sent for pathology. The skin was closed using interrupted subcuticular stitches and Monocryl.

Perianal mass with pressing pain at 7 o’clock direction.

Results of perianal ultrasound showing a hypoechoic mass with clear edges (red arrow) approximately 3.07 cm × 2.24 cm in size.
Gross examination showed a gray-yellow mass measuring 4 cm × 3 cm × 3 cm (Figure 3). Hematoxylin and eosin staining revealed nests of mononuclear cells with abundant eosinophilic granular cytoplasm, small central nucleoli, and a small number of vascular structures in the tumor tissues, with no hemorrhage or necrosis. Immunohistochemical examination showed notable and strong positivity for NSE, vimentin, and S-100. The immunostaining pattern also showed slight positive expression of Ki-67. However, cytokeratin AE1/AE3, epithelial membrane antigen, synuclein, chromogranin A (CgA), CD34, smooth muscle actin, desmin, and neurofilament proteins were all negative (Figure 4). These findings were compatible with a GCT. The patient was followed-up for 19 months after the operation, with no recurrence to date.
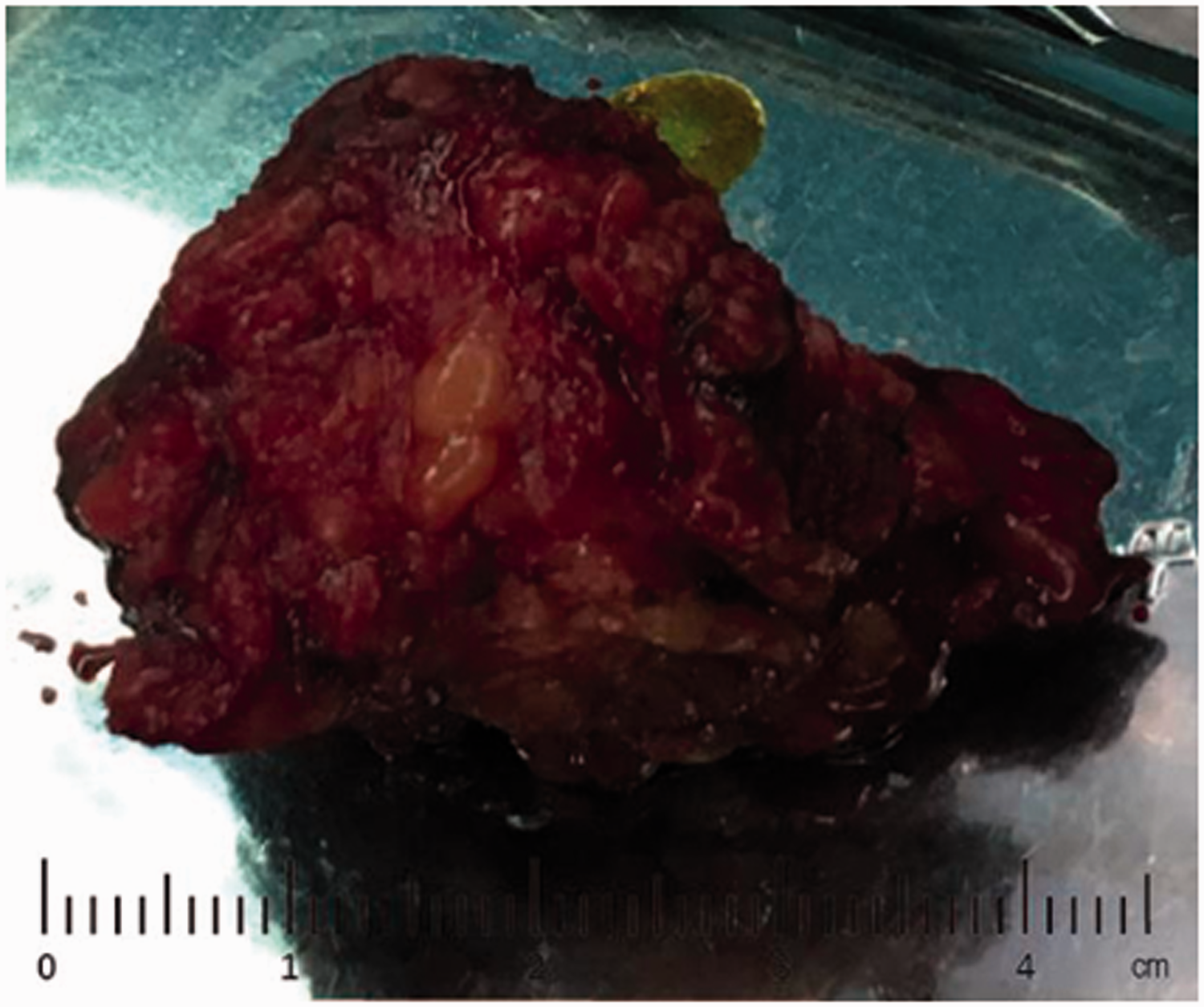
Gross appearance of tumor after complete resection.

Histological staining and immunostaining of the tumor after complete resection. (a) Hematoxylin and eosin-staining. Immunostaining for (b) S-100, (c) neuron-specific enolase, (d) vimentin, (e) Ki-67 (darker portion indicates positive expression), and (f) AE1/AE3. (All × 2). Scale bars = 20 μm.
The study was approved by the ethics committee of Beijing Dongfang Hospital and conducted in accordance with the provisions of the Declaration of Helsinki. All patient details have been de-identified, and the patient provided written informed consent for treatment and publication of this report.
Discussion
GCT is a rare tumor that usually occurs singly, but can occur in different areas at the same time or successively. 6 Since its first report in 1926, 7 GCT has been reported in various parts of the body, with the most common sites being the oral cavity and tongue followed by the skin and subcutaneous tissue, while GCTs in the lower digestive tract, especially in the perianal areas, are relatively rare. Perianal GCT is a neurogenic tumor originating from the submucosa and characterized by Schwann cell differentiation. In our case, the tumor, located around the anus and away from the skin, was considered to be in a rare location for GCT.
Although the etiology and histogenesis of GCT are not completely clear, they can still be characteristic. Perianal GCT may manifest as perianal discomfort or hemorrhage, but most cases involve asymptomatic masses. Its preoperative diagnosis is difficult because of its unclear symptoms, and it may be misdiagnosed as other diseases, such as abscess or teratoma. GCT needs to be differentiated from lipoma, hemangioma, fibroma, leiomyoma, sebaceous cyst, and superficial myxoma. The current gold standard for the diagnosis of GCT is histopathological and immunohistochemical examination. 8 The histopathological manifestations of GCT are characterized by a neoplastic mass with unclear boundaries in the dermis and subcutaneous tissues, including large neoplastic cells with nests or cords, and eosinophilic granules. Immunohistochemical tests often show positivity for NSE, S-100, and CD57, but some GCTs have been S-100-negative. 9 The expression of S-100 indicates that GCTs originated from Schwann cells in the neuroectoderm. Unlike in adults, patients with congenital GCT were negative for S-100, and there was no pseudoepithelial hyperplasia on the surface of the tumors. 10 GCT tissues also express vimentin and CD68. Expression of the macrophage marker CD68 contributes to the aggregation of lysosomes in the cytoplasm but does not reflect the origin of the tumor cells. This supports the hypothesis that GCT represents a non-specific degeneration process of mesenchymal cells via self-phagocytosis. In the current case, the pathological results showed positive immunostaining for S-100, NSE, and vimentin, supporting the diagnosis of a neurogenic tumor. Meanwhile, negativity for CgA could exclude an endocrine origin, while negativity for AE1/AE3 excluded an epidermal origin. The cytoplasm showed granular changes, consistent with the appearance of GCT. Because the epidermis of GCT often shows pseudoepithelioma-like proliferation, it is easily misdiagnosed as SCC, and a pathological diagnosis is required to differentiate between them. SCC is a malignant tumor of the epidermis and its appendages that shows granules and desmosomes in cells under electron microscopy. However, intracellular granular PAS staining is negative in SCC, in contrast to GCT.
Most GCTs present with benign biological behaviors, 11 but less than 2% of GCTs are diagnosed as malignant GCTs (MGCT). 12 However, benign and malignant tumors have similar histological changes, making it difficult to differentiate between them. Malignant cases may still present with benign clinical manifestations and histomorphology. Gambou divided MGCTs into two groups: a clinically malignant but histologically benign type (type I) and a clinically malignant and histologically malignant type (type II). 13 Type II is easier to diagnose than type I. However, opinions on MGCT typing differ and there is a lack of uniform histological criteria. MGCT is usually associated with rapid growth, diameter greater than 4 cm, cell necrosis, spindle-shaped tumor cells, marked cell atypia, active mitosis (more than 2/10 high-power fields (HPF), ×20), high nuclear-plasma ratio, local recurrence, and high p53 and Ki-67 expression. 14 In the current case, Ki-67 (positive in 2%) indicated a higher risk of MGCT; however the inactive mitotic image (less than 2/HPF) and the non-invasive cell growth temporarily excluded the possibility of MGCT.
Some studies suggest that asymptomatic GCT needs no special treatment and requires only regular follow-up15,16; however, most scholars believe that such tumors should be treated with complete surgical excision, without excessive loss of normal tissue.17–19 Local excision of the tumor and surrounding normal tissues of at least 1 cm is the main treatment option, while selective endoscopic resection can be used to target small early tumors. Although most tumors with safe margins rarely recur, the metastasis rate of malignant lesions was over 50%, and the 3-year mortality rate was about 30% to 50%.10,11 Extensive local resection and local lymph node dissection are the main treatment methods for MGCT. GCT is insensitive to radiation, which may cause malignant changes, and radiotherapy is therefore not a treatment option. In addition, chemotherapy has been shown to have no significant beneficial effect on the clinical course of GCT. Some apparently recurrent cases may be due to multiple nodules and/or incomplete local resection, while benign GCT seldom recurs, and a cure can be achieved if the correct surgical treatment is chosen. The most common sites of metastasis of MGCT are the local lymph nodes and lungs, with an average survival time of 2.5 years. 10 Patients should be re-examined regularly after tumor resection to exclude recurrence. Endoscopy can help to detect recurrence in the submucosa of the lumen, while computed tomography and magnetic resonance imaging are helpful for identifying small or concealed recurrences. The current patient remained under follow-up.
Conclusion
Perianal GCT is a rare disease that is difficult to diagnose clinically, but which has a low malignant-transformation rate. However, even though lesions in the perianal region might have a clinically benign appearance, a thorough preoperative examination, including histopathology, should be performed. GCT should also be kept in mind during the diagnosis. Epithelial cells show similar pseudoepithelioma-like proliferation in GCT and SCC, and biopsy and immunohistochemical analysis are thus important for making an accurate diagnosis and applying the appropriate treatment. In light of the high recurrence rate, patients must be closely monitored after treatment.
Footnotes
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
