Abstract
Objective
We investigated whether the recipient’s complement system function, kidney graft endothelial ultrastructural injury, and microRNA (miRNA) expression before transplantation may be associated with the risk of posttransplant de novo thrombotic microangiopathy (TMA).
Methods
Complement system function assessment, histological and ultrastructural examination of preimplantation and kidney graft biopsies, and microRNA assessment were performed on kidney transplant recipients (KTRs) with de novo TMA.
Results
On the basis of the clinical course, histological findings, and miRNA patterns, the following two de novo TMA phenotypes were observed: a self-limiting disease that was localized to the kidney graft and a systemic disease that progressed to graft failure without timely treatment. Decreased alternative complement pathway activity and ultrastructural endothelial injury before transplantation were confirmed in all five KTRs and four of five KTRs, respectively, but they did not correlate with de novo TMA severity.
Conclusions
Alternative complement pathway abnormalities in KTRs and endothelial ultrastructural injury on preimplantation biopsy might be associated with de novo posttransplant TMA, although they did not predict posttransplant TMA severity (localized vs. systemic). The specific miRNA expression patterns in preimplantation kidney graft biopsies demonstrated a borderline statistically significant difference and might provide more accurate information on posttransplant TMA severity.
Keywords
Introduction
Posttransplant thrombotic microangiopathy (TMA) is a recognized serious and debilitating complication of kidney transplantation that results in graft dysfunction and inferior long-term graft outcomes. It affects 6.4% of kidney graft biopsies, among which the etiology remains unclear in 33%. 1 Posttransplant TMA can occur as a recurrent disease in a recipient with a history of atypical hemolytic uremic syndrome (aHUS) or as a de novo disease. De novo TMA is more common than recurrent TMA, and it is associated with a worse prognosis. 2 Posttransplant de novo TMA is most frequently diagnosed in the initial 3 to 6 months after transplantation, but it can occur at any time after kidney transplantation. It can manifest in various phenotypes, from localized (renal-limited) TMA to systemic TMA with its classic triad of thrombocytopenia, microangiopathic hemolytic anemia, and decrease in kidney function. In the acute stage, evidence of endothelial injury with platelet aggregation (thrombosis), fibrinoid necrosis, and glomerular ischemia can be seen.2–4
The etiopathogenesis of de novo TMA is poorly understood. De novo TMA has been associated with risk factors before and after transplantation, such as ischemia–reperfusion injury, the use of certain drugs (i.e., calcineurin inhibitors, mammalian target of rapamycin [mTOR] inhibitors, infection, and antibody-mediated rejection [AMR]). However, because transplant recipients are usually exposed to the same risk factors, it remains unknown why only a small number of recipients develop TMA.2,3
Genetic abnormalities in complement regulatory genes have recently been reported in one-third of patients with de novo TMA, 5 which suggests that complement abnormalities may play an important role in the deleterious inflammatory reactions that can contribute to endothelial dysfunction, TMA, and graft loss.4,6 In our previous study, we showed that ultrastructural (by electon microscopy (EM)) assessment of endothelial injury severity in the kidney graft, which presented as unremarkable on light microscopy, was associated with a poor graft outcome. 7 In addition, microRNAs (miRNAs), which are small non-coding RNAs, have emerged as an important regulator in the development of immune and inflammatory responses; some miRNAs also have a role in vascular/endothelial (miR-126) and glomerular (miR-150 and miR-223) function.8,9
Because the endothelium is the main target for complement, we investigated the hypothesis that a complement abnormality in the recipient and endothelial injury in the kidney graft might be important risk factors for posttransplant de novo TMA and a poor kidney graft outcome. In addition, a previously undiagnosed complement abnormality in combination with the transplantation procedure might trigger TMA in some patients. Finally, we selected five miRNAs that are involved in immune cell development and activity at different stages (miR-126, miR-146a, miR-150, miR-155, and miR-223), and investigated whether any of these miRNAs may be associated with posttransplant de novo TMA and kidney graft outcome.
Patients and methods
Patients
We retrospectively examined a complete national cohort of kidney transplant recipients (KTRs) in whom we performed an indication graft biopsy between January 2018 and December 2018 (n = 54). Kidney graft biopsies were indicated in the case of delayed graft function (DGF, which is defined as at least one dialysis session during the first week after transplant), persistent posttransplant graft dysfunction or an increase in serum creatinine that is greater than 20% from the baseline without other evident causes, or unexplained persisting or newly diagnosed proteinuria. Five kidney transplant recipients with histologic evidence of posttransplant de novo TMA were identified and further assessed.
KTRs with posttransplant TMA (TMA group) included patients with the following: (i) an indication kidney biopsy that was performed within 3 months after transplantation; (ii) histological diagnosis of TMA without criteria of AMR (no anti-HLA donor-specific antibodies [DSA] and C4d negative) in accordance with the Banff classification; 10 (iii) stored serum samples taken before transplantation; (iv) unremarkable histological results in the pretransplant kidney graft biopsy; and (v) no history of aHUS.
The control group included five recipients who were as follows: (i) matched on the basis of age and time after transplant; (ii) had an uneventful posttransplant clinical course with stable kidney function and no need for an indication biopsy within the first year after transplantation; (iii) had stored plasma samples that were taken before transplantation; (iv) unremarkable histology of pretransplant kidney graft biopsy; and (v) no history of aHUS.
The following data were recorded for each patient: recipient’s age and gender, original kidney disease, percentage of panel reactive antibodies (PRA), cold ischemia time and anastomosis time, type of immunosuppressive therapy, occurrence of and time period between transplantation and histological diagnosis of de novo TMA, and laboratory findings during the posttransplant period, including serum creatinine, estimated glomerular filtration rate (eGFR), lactate dehydrogenase (LDH), haptoglobin, signs of microangiopathic hemolytic anemia, and thrombocyte count. KTRs with posttransplant TMA and a functioning graft and control group patients also underwent a surveillance graft biopsy 12 months after transplantation.
The study was approved by the Republic of Slovenia National Medical Ethics Committee on 23 January 2016 (approval number 0120-715/2015-2). All patients provided written informed consent.
Histological assessment
Kidney graft biopsy samples (pretransplant-, indication- and surveillance biopsies) were formalin-fixed paraffin-embedded, cut, and stained using several levels in accordance with the standard histological protocol, including hematoxylin/eosin (HE)-, Periodic Acid Schiff (PAS)-, trichrome-, vanGieson/Weigert, and silver stain. C4d (QUIDEL, San Diego, CA, USA) staining was performed on frozen sections in accordance with the manufacturer’s instructions.
Lesions were evaluated using light microscopy and immunofluorescence and were scored/graded in accordance with the Banff classification. 10 None of the biopsies met the criteria for T-cell-mediated rejection or AMR. There was no significant endarteritis. In vessels with signs of TMA, there were few inflammatory cells.
Histological criteria for glomerular and vascular TMA included at least two glomerular signs of TMA (i.e., endothelial swelling in uncollapsed glomeruli, bloodless glomeruli, fragmentation of erythrocytes in the subendothelium, mesangiolysis, or glomerular capillary thrombosis) and at least one of the vascular signs of TMA in at least three arteries/arterioles (i.e., obliteration of the arterial/arteriolar lumen due to endothelial swelling or arterial/arteriolar fibrinoid necrosis). Immunofluorescence microscopy was used to demonstrate fibrin/fibrinogen deposition in the glomeruli and within the vessel walls. Glomerular TMA was confirmed by ultrastructural examination.
Ultrastructural assessment
Kidney graft biopsy samples were fixed in 10% buffered formalin, postfixed in 1% osmium tetroxide, dehydrated in ethanol and propylene oxide, and embedded in Epon (Serva Electrophoresis, Heidelberg, Germany). Ultrathin slices were contrasted with uranyl acetate and lead citrate and examined with a JEM-1200 EX II transmission electron microscope (Tokyo, Japan). Ultrastructural criteria for glomerular or vascular thrombotic microangiopathy were subendothelial widening, endothelial cell swelling, and the presence of intracellular debris in the capillary lumen. The changes were scored semi-quantitatively from (0) to (3), as follows: (0) no change; (1) mild; (2) moderate; and (3) severe changes.
Complement system assessment
Analyses of complement involvement were performed retrospectively on frozen serum samples, i.e., serum samples taken before transplantation from all kidney recipients in both the TMA and control groups (stored at −80°C for less than 1 year).
The functional activity of the classical complement pathway and the alternative pathway was determined using validated enzyme immunoassay protocols, in accordance with the manufacturer’s instructions (Complement System Screen Wieslab®, Euro Diagnostica AB, Malmoe, Sweden) and the results were expressed as a percentage of the positive standard serum, using the following formula: activity = 100% × (mean A450 (sample) − mean A450 (negative control)/(mean A450 (standard serum) − mean A450 (negative control)). The normal range (mean ± two standard deviations [SDs], n = 40) for classical pathway activity was found to be 66% to 113%; however, the normal range for alternative pathway activity was established by the manufacturer (30% to 113%). Serum C3 and C4 protein concentrations were assessed using nephelometry on the Atellica NEPH 630 System (Siemens Healthineers, Marburg, Germany), and the validated diagnostic protocols were provided by the manufacturer. Factor B, factor H, and factor I concentrations were quantified using radial immunodiffusion in accordance with the described procedures that were obtained from the manufacturer (The Binding Site, Birmingham, UK). Normal ranges established by the manufacturer for factors B (205–400 mg/L) and H (345–590 mg/L) were used. Applying this technique, the normal range (mean ± 2 SD, n = 30) for factor I was found to be 28 to 48 mg/L. Anti-factor H IgG antibody concentration was measured using a validated enzyme immunoassay (GA Generic Assays GmbH, Dahlewitz, Germany), in accordance with the manufacturer’s instructions. Results were expressed in arbitrary units per mL and the normal range for the anti-factor H IgG levels (<10 U/mL) that were established by the manufacturer was used. The hemolytic assay for C3 nephritic factor (C3NeF) was performed following the procedure described by Rother. 11 Values were given as the percent hemolysis of the cells, with 10% hemolysis as the normal limit.
RNA isolation and quantitative real-time PCR
miRNAs were analyzed in pretransplant and posttransplant indication kidney biopsies from all five recipients in the TMA group. Paired tissue samples were cut into 10-µm-thick samples from formalin-fixed paraffin-embedded tissue blocks. For the isolation procedure, three to four 10-µm-thick sections were used. Total RNA isolation was performed using an AllPrep DNA/RNA FFPE Kit (Qiagen, Hilden, Germany) in accordance with the manufacturer’s protocol. Briefly, 1 mL of Xylene (Merck, Kenloworth, NJ, USA) was added for deparaffinization and RNA was eluted in 30 µL of nuclease-free water. The concentration was measured using NanoDrop-1000 and Qubit by High-sensitivity RNA assay (Thermo Fisher Scientific, Waltham, MA, USA).
miRNAs (miR-126, miR-146a, miR-150, miR-155, and miR-223) were analyzed using the miRCURY LNA miRNA PCR system (Qiagen). As the reference gene, miR-103a-3p was used in accordance with the manufacturer’s instructions and a previous publication. 12 All the reagents were from Qiagen, except where otherwise indicated. Quantitative real-time polymerase chain reaction (qPCR) was performed using Rotor Gene Q. For reverse transcription, a miRCURY LNA RT kit was used in 10 µL of reaction master mix containing 10 ng of total RNA, according to the manufacturer’s instructions. The resulting RT was diluted ten-fold and 3 µL was used in 10 µL of reaction master mix, according to the manufacturer’s instructions. All the qPCR reactions were performed in duplicate. Before qPCR, RNA samples were pooled and RT and qPCR were then performed, as described above, except using 100 ng of total RNA. Efficiency was tested for each analyzed miRNA using ten-fold dilutions, and qPCR was performed in triplicate. The signal was collected at the endpoint of each cycle. Following amplification, melting curve analysis of the PCR products was performed to verify the specificity and identity. Melting curves were acquired on the SYBR channel using a ramping rate of 0.7°C/60 s for 60 to 95°C.
Statistical analysis
Average values of functional tests (CPA [classic pathway activity] and APA [alternative pathway activity]) of complement activation were expressed as the median with the interquartile range, and differences between groups were compared using the non-parametric Mann–Whitney U-test.
To present relative gene expression, the calculated ΔCq in patients with TMA was used and tested for statistical significance using either the Mann–Whitney U-test (independent samples) or Wilcoxon Signed-Rank test (paired tissue samples, pre- and post-transplant biopsy). Correlations and associations were analyzed using Spearman’s rank correlation coefficient (Spearman’s rho). For all statistical analyses, SPSS version 24.0 (IBM, Armonk, NY, USA) was used with a cut-off point at p<0.05, which was considered to be significant.
Results
Clinical characteristics of kidney transplant recipients
Five patients with posttransplant de novo TMA and five control patients without posttransplant kidney complications were retrospectively assessed. All recipients in both groups received a first kidney transplant from a deceased donor. Maintenance immunosuppression consisted of mycophenolate mofetil (MMF), steroids (withdrawn 5 days after transplantation in two patients in the control and in one patient in the TMA group), and tacrolimus (TAC) with target trough levels 8 to 12 ng/mL during the first 3 months posttransplant and 5 to 8 ng/mL thereafter. The average age at transplantation was 54 ± 5 years in the TMA group and 51 ± 19 years in the control group. The cold ischemia time was 18.4 ± 6.7 hours in the TMA group and 15.7 ± 8.8 hours in the control group. The anastomosis time was 56 ± 23 minutes in the TMA group and 44 ± 19 minutes in the control group. Two recipients in the TMA group and one recipient in the control group had pretransplant PRA detected. Anti-HLA DSA were absent in all recipients. Three recipients in the TMA group and no recipient in the control group experienced DGF. The baseline demographic and transplant-related characteristics of the TMA and control patient groups are presented in Table 1.
Baseline patient demographic and transplantation-related characteristics.

*No biopsy of the native kidney.
**Rapid steroid withdrawal on day 5 posttransplant.
M, male; F, female; c, control; p, patient; FSGS, focal segmental glomerulosclerosis; ADPKD, autosomal dominant polycystic kidney disease; VUR, vesicouretheral reflux; TIN, tubulointerstitial nephritis; DM, diabetes mellitus; DGF, delayed graft function; PRA, panel reactive antibodies; TAC, tacrolimus; MMF, mycophenolate mofetil; SW, steroid withdrawal; Ig, immunoglobulin; A, major histocompatibility complex, class I A; B, major histocompatibility complex, class I B; DR, major histocompatibility complex class II DR.
Clinical characteristics, additional treatments, and graft outcome in patients with TMA are presented in Table 2 and Figure 1. The time between transplantation and onset of TMA was less than 1 month in all five recipients. In three recipients with DGF, systemic signs of hemolysis (systemic TMA), microangiopathic hemolytic anemia, increased LDH, decreased haptoglobin, thrombocytopenia, and a rapid decrease in eGFR appeared within the first week after transplantation. In two recipients, graft dysfunction without microangiopathic hemolytic anemia and no systemic signs of hemolysis persisted without the need for hemodialysis, necessitating a kidney biopsy within 2 weeks after transplantation, which revealed signs of TMA (localized TMA) (Table 2).
Clinical characteristics, additional treatments, and graft outcome in patients with TMA.

TMA, thrombotic microangiopathy; HLA, human leukocyte antigen; DSA, donor-specific antibodies; PF, plasmapheresis; TAC, tacrolimus; EVR, everolimus; IVIG, intravenous immunoglobulin; DGF, delayed graft function.

Time to TMA diagnosis (kidney graft biopsy) and kidney graft outcome in TMA patients. Three patients were treated with eculizumab (arrows indicate first introduction of eculizumab).
Two patients with histologically proven posttransplant localized TMA recovered spontaneously with no specific additional treatment. These two recipients had no apparent clinical reasons for the persisting graft dysfunction. The other three patients with systemic TMA received additional treatment; two of them, who were treated with eculizumab immediately after diagnosis, experienced improved kidney graft function, while the patient with delayed eculizumab treatment (1 week after histologic diagnosis) lost the graft (Table 2, Figure 1).
Additional inquiry into the paired kidneys that were transplanted elsewhere revealed a favorable posttransplant clinical course (data not shown).
Histological features of kidney transplant biopsies
Pretransplant kidney biopsies (TMA and control group)
Using light microscopy, glomeruli in all pretransplant biopsies from TMA and control patients showed normal kidney parenchyma. None of the pretransplant biopsies showed histologic signs of glomerular or vascular TMA. However, light microscopy revealed mild to moderate acute tubular injury in all pretransplant biopsies in the TMA and control patient groups. There were no significant deviations among biopsies, and one pretransplant biopsy showed marked arteriolar hyalinosis.
Posttransplant indication kidney biopsies (TMA group)
Diffuse glomerular and vascular TMA was found in three of five recipients (systemic TMA). Light microscopy revealed glomerular TMA (at least two of the following signs: endothelial swelling, bloodless glomeruli, fragmentation of erythrocytes in the subendothelium, mesangiolysis, or glomerular capillary thrombosis) and vascular TMA (obliteration of the arterial/arteriolar lumen due to endothelial swelling or arterial/arteriolar fibrinoid necrosis in at least three arteries/arterioles) (Figure 2). It also revealed the following signs of microvascular injury in accordance with the Banff criteria: glomerulitis and peritubular capillaritis; and mild endarteritis of the interlobular sized arteries. All patients with systemic TMA presented with severe endothelial changes, including glomerular capillary thrombosis and/or vascular fibrinoid necrosis. There were also signs of acute tubular injury, which was accompanied by diffuse interstitial edema without significant interstitial infiltrate.
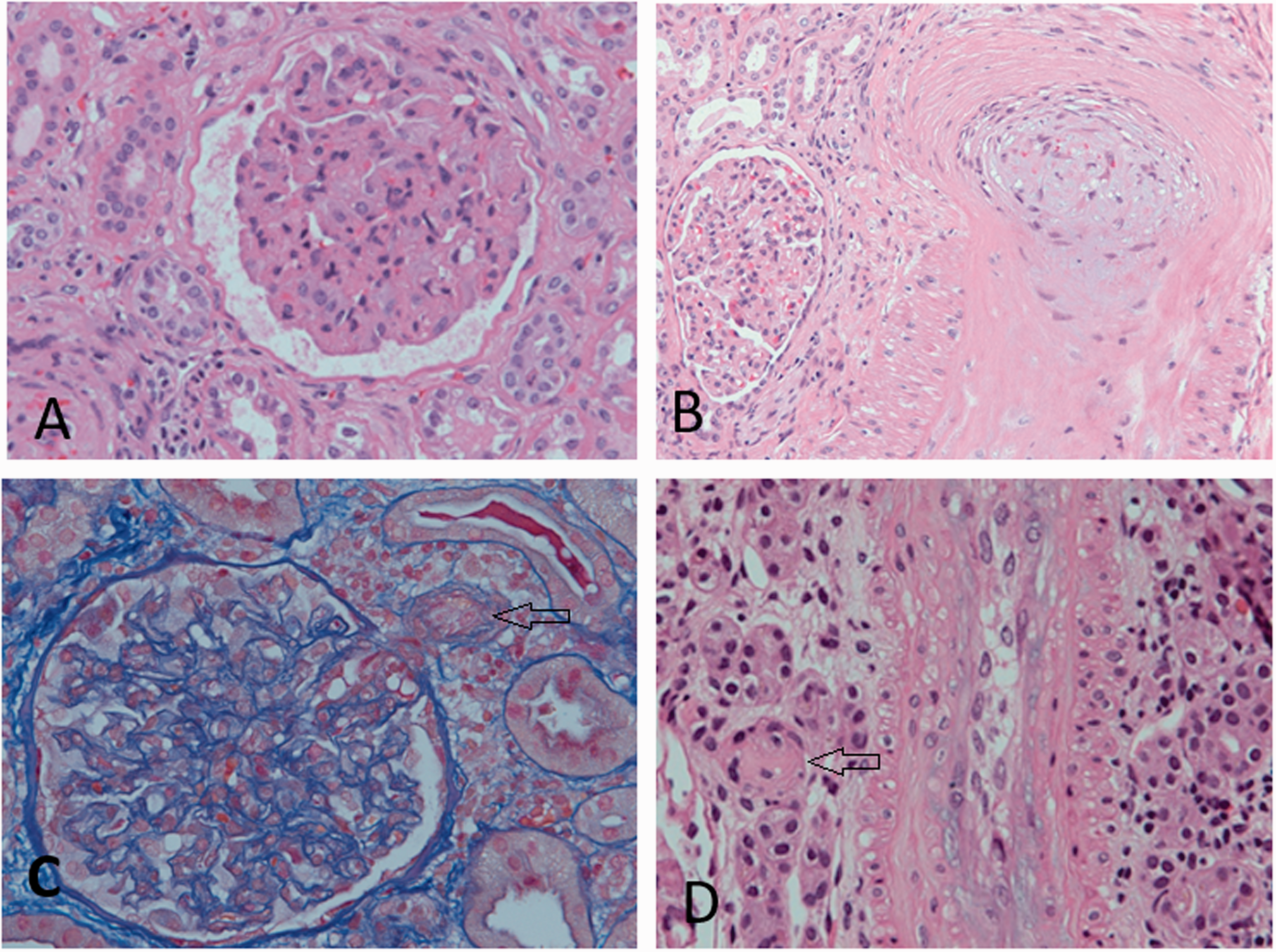
Glomerular and vascular TMA in three patients with systemic disease on posttransplant indication kidney biopsy (a–c). a. Glomerular endothelial swelling and mesangiolysis with fragmentation of erythrocytes in slightly collapsed glomerulus. b. Mucoid intimal hyperplasia with obliteration of the lumen and fragmentation of erythrocytes in small arteries. c. TMA in the hilar arteriole with obliteration of the lumen (arrow). d. Vascular TMA with arteriole obliteration (arrow) in a patient with self-limiting disease without clinical signs of TMA.
Two of the five patients had no clinical manifestation of hemolysis, and they showed localized TMA lesions in the kidney biopsy. One of them presented with isolated glomerular TMA (endothelial swelling and focal fragmentation of the erythrocytes in the mesangium), while the other showed obliteration of the arterial/arteriolar lumen due to endothelial swelling in some arteries/arterioles and focal arteriolar fibrinoid necrosis that was consistent with vascular TMA (Figure 2).
Surveillance kidney biopsies (TMA and control group)
All 12-month surveillance kidney biopsies of TMA and control patients showed unremarkable changes without any signs of rejection or TMA. A patient with isolated vascular TMA displayed the following mild chronic vascular changes: vascular narrowing of up to 30% of the luminal area by fibrointimal thickening of some small arteries without breach of internal elastic lamina. There was no arterial inflammation.
Ultrastructural features
Ultrastructural examination revealed no signs of endothelial injury in pretransplant kidney biopsies in the control group. There were only mild alterations of endothelial cells in the glomeruli, peritubular capillaries, and arterioles, which are usually seen on pretransplant biopsies (such as microvillus transformation of endothelial cell membranes (arcade structures)). In the TMA group, all but one pretransplant biopsy showed the following scarce to moderate endothelial changes: swelling of the glomerular and peritubular capillary and arteriolar endothelium (Figure 3).

Ultrastructural microscopy of kidney graft biopsy. a. TMA group (four of the five patients) showed mild to moderate swelling of the glomerular and b. peritubular capillary endothelial cells in pretransplant biopsies. c. Patient with no endothelial changes in the glomerular and d. peritubular capillaries in the pretransplant biopsy showed e. severe endothelial swelling in glomeruli and f. in peritubular capillaries in the posttransplant biopsy. g. A patient with localized vascular TMA showed milder glomerular endothelial swelling in posttransplant biopsy compared with h. pretransplant biopsy.
On posttransplant indication biopsies, four of the five patients showed more severe endothelial injury of all compartments compared with the pretransplant biopsy. A patient with localized vascular TMA showed milder glomerular endothelial swelling and increased swelling of the capillary/arteriolar endothelium that was consistent with vascular TMA (Figure 3).
The results of light and electron microscopy of the kidney biopsies are shown in Table 3 (posttransplant indication kidney biopsies) and Table 4 (pretransplant kidney biopsies).
Histological and ultrastructural features of posttransplant indication kidney biopsies in patients with TMA.

TMA, thrombotic microangiopathy; t, tubulitis; i, interstitial infiltrate; ti, total inflammation; ptc, peritubular capillaritis; v, endarteritis; vascular necrosis, fibrinoid necrosis of arteries/arterioles; g, glomerulitis; EM, electron microscopy.
Pretransplant serum complement levels and pretransplant graft histological and ultrastructural features in the TMA and control patient groups.
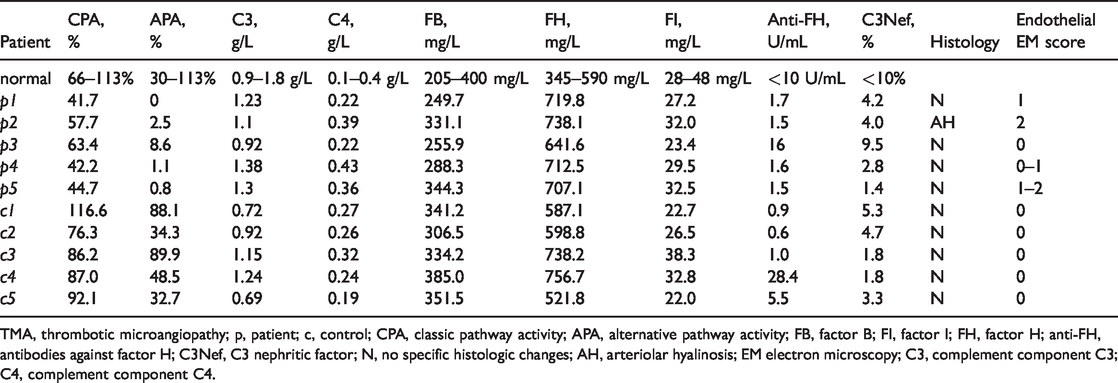
TMA, thrombotic microangiopathy; p, patient; c, control; CPA, classic pathway activity; APA, alternative pathway activity; FB, factor B; FI, factor I; FH, factor H; anti-FH, antibodies against factor H; C3Nef, C3 nephritic factor; N, no specific histologic changes; AH, arteriolar hyalinosis; EM electron microscopy; C3, complement component C3; C4, complement component C4.
Pretransplant complement abnormalities
All recipients in the TMA group consistently had CPA and APA that were below the lower normal limit and a significantly lower CPA (p<0.001) and APA (p = 0.002) compared with recipients in the control group. In the control group, all CPA and APA values were within the normal range (only C1 had a slightly increased CPA [116.2%; range, 66% to 113%]).
In the TMA group, the median CPA was 44.7% (the 25th and 75th percentiles were 42.2% and 57.1%, respectively) compared with 87% (the 25th and 75th percentiles were 86.2% and 92.1%, respectively) in the control group (p<0.001).
In the TMA group, the median APA was 1.1% (the 25th and 75th percentiles were 0.8% and 2.5%, respectively) compared with the median APA of 48.5% (the 25th and 75th percentiles were 34.3% and 88.1%, respectively) in the control group (p = 0.002).
Two recipients in the TMA group and three recipients in the control group had slightly decreased serum factor I levels before transplantation. In each group, antibodies against factor H were detected in one recipient. Additionally, there were no obvious differences in the measured complement components between recipients with localized and systemic TMA (Table 4).
MicroRNA expression in pre- and post-transplant kidney biopsies (TMA group)
Two miRNA expression patterns were observed within the TMA group. Posttransplant kidney biopsy samples with systemic TMA (three of the five patients) showed significantly higher expression in four of the five miRNAs (miR-146a, miR-150, miR-155, and miR-223) than in the pretransplant biopsy samples (p<0.05). No significant difference in miRNA expression was observed between pre- and post-transplant biopsy samples in patients with localized TMA (Figure 4). In pretransplant biopsies, miR-126, miR-146a, and miR-223 showed a similar expression pattern in both recipients with systemic and recipients with localized TMA. miR-150 and miR-155 expression in pretransplant biopsies differed among patients with localized vs. systemic TMA, but this difference was not significant. This observation was additionally confirmed using the Spearman correlation coefficient, which showed an association between miR-150 and miR-155 expression and sample groups (Rho = 0.828, p = 0.042).

miRNA expression in pre- and post-transplant kidney biopsy samples from recipients with posttransplant de novo TMA. a. miRNA expression in post-transplant kidney biopsy samples of recipients with localized and systemic TMA compared with pretransplant biopsy samples. b. miR-150 and miR-155 expression in pre-transplant kidney biopsy samples from recipients with localized and systemic TMA.
Discussion
In the present study, we showed that recipients with posttransplant de novo TMA, which was unrelated to AMR and with no previous known history of aHUS, had dysregulated classical and particularly alternative complement pathway in their serum before transplantation. However, control recipients with an unremarkable clinical course after transplantation had normal pretransplant activity of both complement pathways. A favorable posttransplant clinical course for paired kidneys that were transplanted elsewhere supported the important role of recipient’s complement system in kidney graft function. Our study corroborates the findings of other studies that a dysregulated complement system in the transplant candidate may be associated with a greater risk of posttransplant complications.3–6
One possible explanation for the posttransplant de novo TMA in the five recipients is that they may have had undiagnosed, clinically overt abnormalities of the complement pathway before transplantation. Because complement alternative pathway abnormalities have been described in membranous nephropathy, IgA nephropathy, and polycystic kidney disease, and they were also diagnosed in three patients with posttransplant TMA, the TMA after transplantation in these patients may be associated with previously undiagnosed alternative pathway abnormalities that are related to their original kidney disease.13–15
In our patients, a dysregulated complement system in transplant candidates was not associated with the severity of histological features in the transplanted kidney or in the clinical course of TMA after transplantation. Three recipients experienced systemic TMA with significant graft dysfunction that required dialysis treatment, while two of the five recipients experienced slowly improving kidney graft function without systemic TMA manifestations. Although a kidney biopsy revealed localized TMA in these two recipients, the graft function was restored spontaneously without any additional treatment. However, kidney graft function in recipients with systemic TMA improved only after introduction of eculizumab, a complement inhibitor that was previously shown to be successful in various etiologies of posttransplant TMA, including AMR and medication associated TMA.16–21
We previously reported that ultrastructurally assessed severe endothelial injury, even in unremarkable biopsy reports on light microscopy in the preimplantation kidney biopsy, was associated with posttransplant kidney graft dysfunction. 7 Consistent with this analysis, we tested whether the severity of ultrastructurally assessed endothelial cell injury before transplantation could be related to a different histological and clinical picture of TMA. Most patients (four of the five) with de novo TMA received kidney grafts with ultrastructural signs of widespread endothelial injury, which is in contrast to none of the patients in the control group who all had unremarkable endothelial ultrastructure, but the severity of pretransplant endothelial injury did not correlate with the severity of posttransplant complications. Electron microscopy revealed that recipients who received a kidney with moderate endothelial injury developed a milder form of posttransplant TMA, and recipients who received a kidney with mild or no endothelial injury developed a severe form of systemic posttransplant TMA.
Because morphological and ultrastructural assessments are based on structural alterations of tissue and cells, which are a chronological consequence of the previous molecular events, morphological and ultrastructural assessments offer an insight into kidney disease and injury that originates in the donor. However, injury that is triggered during the organ procurement process may not be seen at the structural level in a such short time. Ischemia and reperfusion are critical factors for graft injury. 22 Because kidneys are procured within a relatively short time, during which mostly molecular alterations take place, we speculated that a molecular-level biomarker that is involved in regulating the immune and inflammatory responses could be a prognostic factor for early kidney graft injury, and one such prognostic factor may be miRNAs.
As expected, analysis of the five selected miRNAs that are involved in immune cell development and activity at different stages as well as in vascular/endothelial and glomerular function (miR-126, miR-150, miR-146a, miR-155, and miR-223)8,9 showed increased expression in all posttransplant biopsies with systemic TMA compared with expression in pretransplant biopsies. The results, thus, confirmed that miR146a, miR-150, miR-155, and miR-223 are involved in underlying detrimental posttransplant processes. Additionally, there was no significant difference in miRNA expression between pre- and post-transplant biopsy samples in patients with localized TMA, indicating that there are two different miRNA expression patterns in systemic compared with localized phenotypes of de novo TMA.
To further evaluate which miRNAs might be useful as an early warning signal of underlying detrimental mechanisms within the kidney graft, miRNA expression in pretransplant biopsy samples was correlated with that of posttransplant kidney complications. The results demonstrated that upregulated miR-155 and miR-150 expression in pretransplant biopsies might be associated with a greater risk of posttransplant systemic TMA. miRNA expression in both pretransplant kidney biopsies might, therefore, provide more accurate information regarding the severity of posttransplant TMA and kidney transplant outcomes than the ultrastructurally assessed degree of endothelial injury before transplantation. Although both miR-150 and miR-155 are involved in various processes,23–25 it is interesting to speculate that miR-155 upregulation might be associated with down-regulation of complement factor H, 26 while miR-150 may be associated with hypoxia-induced processes. 27 The possible association might be through direct or indirect regulation of factors that are involved in regulating the complement system.
The present study has several limitations, including a retrospective analysis of the transplanted patient cohort, the small number of patients, and lack of genetic analysis of genes that are involved in complement regulation. Our results, therefore, need to be explored further and evaluated in a larger prospective study cohort.
To conclude, our study indicated that dysregulation of complement system activity in KTRs and ultra-structurally observed endothelial injury in the kidney graft before transplantation may play an important role in triggering de novo TMA after transplantation, although neither could differentiate between localized vs. systemic TMA. A specific miRNA expression pattern in the kidney graft before transplantation, which was upregulated miR-150 and miR-155, was associated with an increased risk of posttransplant systemic TMA. However, this observation should be verified in a larger independent cohort of patients with posttransplant TMA.
Footnotes
Author contributions
All authors participated in research design, performance of the research, and writing of the manuscript. MA, ŽVH, and NK collected data. EB and SS participated in data analysis. MP, MA, AAR, and NK performed critical review.
Data availability
The data used to support the findings of this study are included within the article.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
