Abstract
Objective
This study aimed to investigate the expression of O-linked glycoprotein glycans in tissue of patients with cholangiocarcinoma compared with adjacent normal tissue.
Methods
Sixty patients with cholangiocarcinoma were included in the study. Permethylated O-linked glycans from intrahepatic cholangiocarcinoma tissue and adjacent normal tissue were analyzed using nano-spray ionization-linear ion trap mass spectrometry. Histochemistry of peanut agglutinin lectin was used for detection and localization of galactose (Gal) 1, N-acetyl-galactosamine (GalNAc) 1.
Results
O-linked glycans from patients with cholangiocarcinoma were composed of di- to hexa-saccharides with a terminal galactose and sialic acids (N-acetylneuraminic acid [NeuAc]). A total of eight O-linked glycan structures were detected. Gal1GalNAc1 and Gal2 N-acetyl-glucosamine 1 GalNAc1 expression was significantly higher in tissue from patients with cholangiocarcinoma compared with adjacent normal tissue, while NeuAc1Gal1GalNAc1 expression was significantly lower. High Gal1GalNAc1 expression was significantly associated with the late stage of cholangiocarcinoma (stages II–IV), lymphatic invasion, and vascular invasion.
Conclusion
Our study shows expression of O-linked glycans in progression of cholangiocarcinoma and highlights the association of Gal1GalNAc1 with lymphatic and vascular invasion of cholangiocarcinoma.
Keywords
Introduction
Cholangiocarcinoma (CCA) is an aggressive malignancy of the bile duct epithelium. CCA persists as a major health problem in northeastern Thailand, particularly in areas where the liver fluke Opisthorchis viverrini infection is a major risk factor. 1 The incidence of CCA is rare worldwide, but it is relatively high in areas where liver flukes are endemic as found in East and Southeast Asia. However, there has been an increasing incidence of CCA reported in England, the USA, and Australia.2,3 Typically, diagnosis of CCA occurs when this disease is at advanced stages, leading to limited treatment options and poor prognosis. Novel target biomarkers for CCA are required for early detection of this disease when resection can be curative, and for therapeutic efficacy when chemotherapy is appropriate with or without resection.
More than 50% of human proteins are glycosylated and this structure plays an important role in cell–cell interactions, immune recognition, tissue development, and malignancy.4,5 Altered protein glycosylation may result in abnormal changes in protein structure, biological function, and molecular recognition. 6 Investigation of tumor-related glycoprotein may lead to discovery of potential biomarkers, as well as discovery of therapeutic novel targets.
Study in the field of protein glycosylation in CCA in vitro and in vivo is growing. Sialyl Lewis a expression in patients’ tissues is related to a poor prognosis in CCA. 7 A study that used a monoclonal antibody of the glycans for mucin 5ACS from serum of patients with CCA showed that the level of a glycan epitope (S121) was related to prognosis and was specific to CCA. 8 In an animal model, S121 was found to be expressed in the cytoplasm and apical surface of biliary epithelium at the early stage of the tumor and increased with progression of the tumor. 9 Further immunohistochemical studies showed overexpression of N-acetyl-glucosamine (GlcNAc) 10 and O-GlcNAc transferase in tissue of patients with CCA. 11 Another study in serum of patients with CCA by sandwich ELISA showed that the glycan epitope CA-S27 may have immunodiagnostic value for the prognosis or diagnosis of CCA. 12 A lectin microarray-based sero-biomarker for detection of O-linked glycosylation of CCA was found. 13 In CCA cell lines, a change in O-linked glycan expression in different histological types of CCA was observed. 14 Our recent study of patients with CCA showed increased N-linked glycoprotein glycan expression in the serum of patients with CCA compared with healthy controls. 15
To better understand the association of glycoprotein glycans with clinicopathological features, glycans that are expressed in patients with CCA need to be investigated. Therefore, the present study aimed to determine the structural details and quantity of O-linked glycans from the tissue of patients with CCA compared with normal tissue. Knowledge of specific O-linked glycans may help to understand the mechanisms of carcinogenesis, development, and metastasis of CCA, and may also have application for clinical diagnosis and/or effective therapeutic treatment.
Materials and methods
Reagents
Sodium borohydride, sodium hydroxide, and mucin glycoprotein standard were obtained from Sigma-Aldrich; Merck KGaA (Darnstadt, Germany). D-galactose (Gal), N-acetyl-D-glucosamine (GlcNAc) was purchased from Millipore (Billerica, MA, USA).
Patients and samples
Intrahepatic CCA tissue and adjacent normal tissue were obtained from the Liver Fluke and Cholangiocarcinoma Research Institute, Khon Kaen University, Thailand. Written informed consent was obtained from each subject. The Ethics Committee of Khon Kaen University approved the study protocol (registration number: HE521209).
Protein powder preparation
Protein powder from CCA tissue and adjacent normal tissue was obtained as previously described. 16 Briefly, 100 mg of each tissue was homogenized separately on ice in cold 50% methanol. The homogenate was then extracted in a solvent (chloroform: methanol: water, 4:8:3) for 2 hours at room temperature. The extracts were centrifuged at 2500 × g for 15 minutes at room temperature. The resulting pellets were then dried under nitrogen and stored at –20°C until further use.
Release of O-linked glycans by reductive β-elimination
Preparation of O-linked glycans by reductive β-elimination was performed as previously described. 16 Briefly, 1 mg of protein powder of each of the tissues was subjected to reductive β-elimination in 100 mM NaOH, which contained 1.0 M sodium borohydride at 45°C for 18 hours. The reaction mixture was neutralized with 10% acetic acid and desalted in a column of Dowex®AG50W-X8 (H+) (Sigma-Aldrich; Merck KGaA). 17 The mixture was eluted with 5% acetic acid, lyophilized, and then boric acid was removed by evaporation with methanol. The released O-linked glycans were purified by a Sep-Pak C18 cartridge column (Waters Corporation, Milford, MA, USA). 18
Permethylation of glycans
Permethylation of released O-linked glycans was performed under water-free conditions in 500 µL of dimethyl sulfoxide (DMSO), 10 µg of NaOH, and 150 µL of methyliodide as previously described. 19 Briefly, 200 µL of DMSO was added to the O-linked glycan sample in a screw-cap tube. After 1 minute of sonication, 300 µL of prepared base (10 µg of NaOH in DMSO) and 150 µL of methyliodide were added to the sample mixture. The sample mixture was vortexed vigorously at maximum speed for 5 minutes and then sonicated for 10 minutes. The permethylated glycans were then extracted using 2 mL of nanopure water and 2 mL of dichloromethane for five times. The permethylated glycans in dichloromethane were then dried under nitrogen.
Mass spectrometry
Permethylated O-linked glycans were analyzed using nano-spray ionization-linear ion trap mass spectrometry (MS) as previously described. 18 Briefly, permethylated glycans were dissolved in 1 mM NaOH in 50% methanol and infused directly into a linear ion trap mass spectrometer (LTQ Orbitrap Discovery; Thermo Fisher Scientific, Inc., Waltham, MA, USA). MS and MS/MS (at 28% collision energy) analysis was performed in a positive ion mode and identified using the nomenclature described by Domon and Costello. 20 The prevalence of each glycan in each profile was quantified by comparing its signal intensity to the sum of signal intensities for all identified glycans in the profile, thereby yielding the % total profile.
Lectin histochemistry
Biotinylated peanut agglutinin (PNA) (Vector Laboratories, Burlingame, CA, USA) was used for detection of Gal1GalNAc1. Paraffin sections of CCA tissues were deparaffinized in xylene, and then hydrated in downgraded ethanol and distilled water. The antigens were unmasked by heating each section (in 0.1 mol/L citrate buffer, pH of 6.0) in a pressure cooker. The sections were then treated with absolute methanol containing 5% hydrogen peroxide for 30 minutes at room temperature. After washing with phosphate-buffered saline and blocking nonspecific binding (20% normal horse serum for 30 minutes at room temperature), sections were (a) incubated with biotinylated lectin PNA (dilution of 1:20) for 2 hours at room temperature, and then (b) incubated with the VECTASTAIN® ABC HRP Kit (Vector Laboratories) according to the manufacturer’s instructions. The sections were visualized with 3,3ʹ-diaminobezidine-tetrahydrochloride, liquid DAB+ (Dako, Carpinteria, CA, USA), and counterstained with hematoxylin and eosin. The staining results were evaluated as the frequency of PNA-positive cells at the tumor area. Staining was classified into four scoring categories as follows: 0, negative; 1+, 1% to 10%; 2+, 11% to 50%; and 3+, >50%. Scores of 0 and 1+ were categorized as low expression and scores of 2+ and 3+ as high expression for statistical analysis.
Statistical analysis
The prevalence of each glycan in tissue of patients with CCA versus adjacent normal tissues was reported as the mean rank (% total profile). The Mann–Whitney U test was used for determining differential expression of the O-linked glycans between the groups. Fisher’s exact test was used for determining the association between O-linked glycan expression and the clinicopathological features of CCA. IBM SPSS version 22.0 (IBM Corp., Armonk, NY, USA) was used for analysis. P<0.05 indicated a statistically significant difference.
Results
Patients
Sixty patients with intrahepatic CCA tissue and their adjacent normal tissue were included in the study (mean age, 54.78 ± 9.00 years; 17 women and 43 men).
Altered expression of O-linked glycans in tissue of patients with CCA
The mass profiles for the O-linked glycans of tissue from patients with CCA and adjacent normal tissue are shown in Figure 1. Seven monosaccharide compositions yielding eight glycan structures were identified from each tissue. These structures were Gal1, N-acetyl-galactosamine (GalNAc) 1 (structure 1), N-acetylneuraminic acid (NeuAc) 1Gal1GalNAc1 (structure 2a and 2b), Gal2GlcNAc1GalNAc1 (structure 3), NeuAc1Gal1GlcNAc1GalNAc1 (structure 4), NeuAc2Gal1GalNAc1 (structure 5), NeuAc1Gal2GlcNAc1GalNAc1 (structure 6), and NeuAc2Gal2GlcNAc1GalNAc1 (structure 7).

Mass spectrometry spectra show differential expression of O-linked glycans in tissue of patients with cholangiocarcinoma compared with adjacent normal tissue. The glycan profiles (normal versus tumor) show predominance of Gal1GalNAc1 (structure #1) and Gal2GlcNAc1GalNAc1 (structure #3) in tumor tissue versus normal tissue. m/z, mass/charge ratio; Gal, galactose; GalNAc, N-acetyl-galactosamine; GlcNAc, N-acetyl-glucosamine; NeuAc, N-acetylneuraminic acid.
Gal1GalNAc1 and Gal2GlcNAc1GalNAc1 were significantly higher in tissue of patients with CCA compared with adjacent normal tissue (P = 0.003 and P = 0.002, respectively). By contrast, the isobaric structure of NeuAc1Gal1GalNAc1 was significantly lower in tissue of patients with CCA compared with adjacent normal tissue (P = 0.002) (Table 1). The association between O-linked glycan expression and clinicopathological features of CCA was further analyzed. High Gal1GalNAc1 expression in tissue of patients with CCA was associated with vascular invasion (P = 0.018). Low NeuAc1Gal1GalNAc1 expression was also associated with an advanced stage of CCA (P = 0.018) (Table 2).
O-linked glycan expression in tissue of patients with cholangiocarcinoma compared with normal tissue.
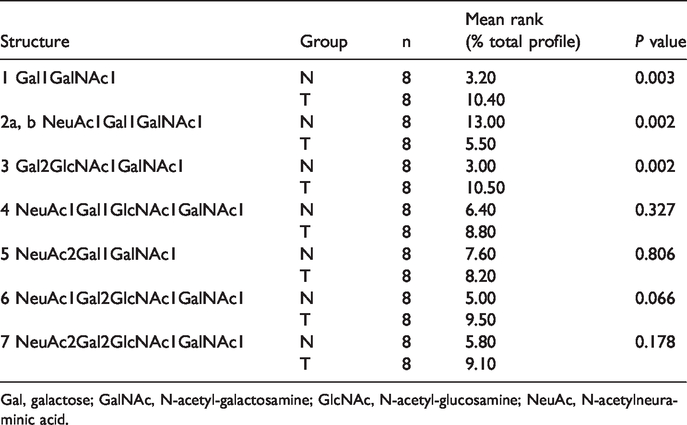
Gal, galactose; GalNAc, N-acetyl-galactosamine; GlcNAc, N-acetyl-glucosamine; NeuAc, N-acetylneuraminic acid.
O-linked glycan expression and clinicopathological features of patients with cholangiocarcinoma.
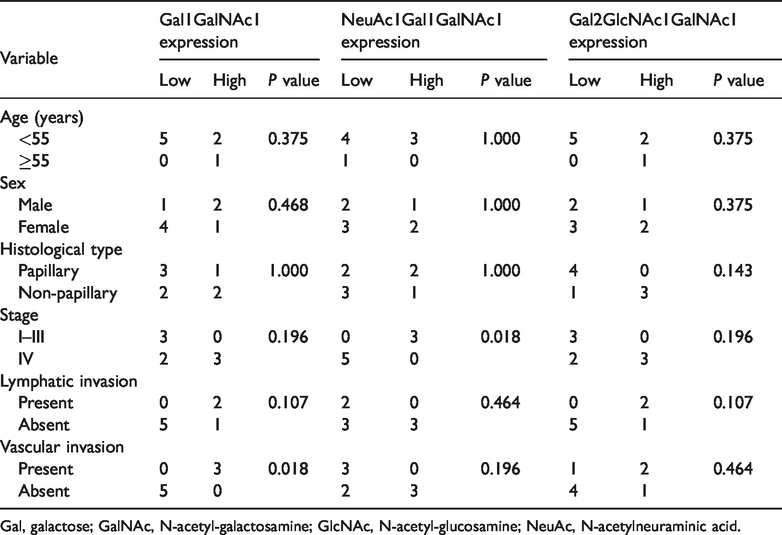
Gal, galactose; GalNAc, N-acetyl-galactosamine; GlcNAc, N-acetyl-glucosamine; NeuAc, N-acetylneuraminic acid.
High Gal1GalNAc1 expression is associated with advanced tumor stage, and lymphatic and vascular invasion of CCA
We further investigated Gal1GalNAc1 expression in 60 intrahepatic CCA tissues by histochemistry of PNA lectin. Gal1GalNAc1 was expressed in the cell membrane and cytoplasm of tumor cells, especially in the epithelium lining of the bile ducts (Figure 2.). Thirty-four CCA tissues were classified as high expression of Gal1GalNAc1 (56.67%), with specific membrane and cytoplasmic staining (scores of 2+ and 3+). The remaining 26 CCA tissues (43.33%) showed negative or partial staining and were classified as low expression (scores of 0 and 1+). The associations between Gal1GalNAc1 expression and clinicopathological features were further analyzed using univariate analysis. High Gal1GalNAc1 expression was significantly associated with tumor staging (P =0.002), lymphatic invasion (P = 0.033), and vascular invasion (P = 0.043) (Table 3).

Histochemistry of lectin shows high Gal1GalNAc1 expression in cholangiocarcinoma (CCA) tissue. (a) Hematoxylin and eosin (H&E) staining of CCA tissue, (b) peanut agglutinin (PNA)-positive staining of CCA tissue, (c) H&E staining of bile duct epithelium in CCA tissue, and (d) PNA-positive staining of bile duct epithelium in CCA tissue (arrows). Original magnification, ×400.
Association between Gal1GalNAc1 expression and clinicopathological features of patients with cholangiocarcinoma (n = 60).
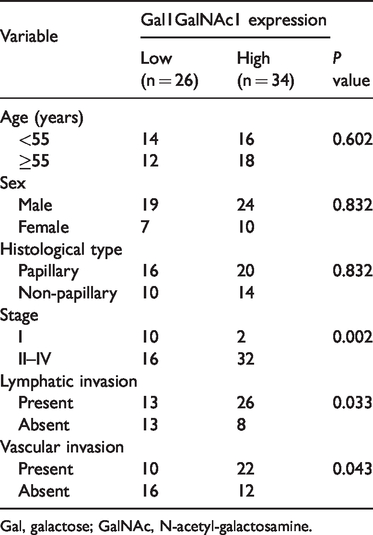
Gal, galactose; GalNAc, N-acetyl-galactosamine.
Discussion
Changes in glycan expression have been observed in various chronic diseases and in malignancy. In this study, we showed comparative glycan expression from tissue of patients with CCA using MS.
Our study showed that Gal1GalNAc1 (structure 1) was significantly elevated in tissue of patients with CCA compared with adjacent normal tissue. Gal1GalNAc1, core 1, or T antigen—known as a pan-carcinoma antigen—has been reported across many types of cancers (e.g., breast cancer, colon cancer, gastric cancer, pancreatic cancer, respiratory cancer, and melanoma). 21 However, truncated O-linked glycans (e.g., Tn, sialyl-Tn) were not detected in this study because of a limitation of the technique used. T antigen interacts with galectin-3, which facilitates tumor cell interaction with endothelial cells and platelets during the early stages of cancer metastasis. 22 Therefore, high Gal1GalNAc1 expression in CCA may be associated with progression and metastasis of tumors.
The present study showed a significant increase in the extended core 2 (Gal2GlcNAc1GalNAc1, structure 3) in tissue of patients with CCA compared with normal tissue. Core 1 is converted to the core 2 structure by the core 2 β6-GlcNAc-transferases C2GnT1 and C2GnT2. C2GnT1 expression is correlated with vascular invasion and lymphatic metastasis. 23 Core 2 O-linked glycans are part of the tumor defense systems against natural killer cell immunity. 24 Therefore, a significant increase in the core 2 family in CCA tissue suggests that they have an important role in progression of tumors in CCA.
We observed the isobaric structure NeuAc1Gal1GalNAc1 (structure 2a and 2b) in tissue of patients with CCA. This aberrant expression is consistent with that of CCA cell lines. 14 However, NeuAc1Gal1GalNAc1 was significantly lower in the tissue of patients with CCA compared with adjacent normal tissue. Competition of NeuAc1Gal1GalNAc1 (sialylated core 1) and the core 2 structure, which compete for the core 1 substrate, has been reported in breast cancer. 25 Therefore, decreased NeuAc1Gal1GalNAc1 expression in CCA might be related to increased expression of the extended core 2 family in CCA cells.
The present study also showed an association between O-linked glycan expression and staging, and lymphatic and vascular invasion of patients with CCA. High Gal1GalNAc1 expression was associated with an advanced tumor stage, and lymphatic and vascular invasion in tissues of patients with CCA. Glycans can regulate proliferation, invasion, metastasis, and angiogenesis of tumors. 26 These findings suggest that O-linked glycan expression, especially that of Gal1GalNAc1 and Gal2GlcNAc1GalNAc1, plays an important role in progression of CCA.
In conclusion, the current study shows important information on O-linked glycans in the tissue of patients with CCA. Increased expression of Gal1GalNAc1 (core 1) and extended core 2 (Gal2GlcNAc1GalNAc1) in patients with CCA suggests that glycans have a major role in growth and progression of tumors. Further investigation into the specific glycoproteins or specific glycosyltranferases in tissue and serum of patients with CCA is required for understanding the mechanism of this cancer and may lead to the discovery of novel target biomarkers or new target for treatment.
Footnotes
Acknowledgements
The authors thank the patients and their families for their participation. The authors also thank the Liver Fluke and Cholangiocarcinoma Research Institute, Khon Kaen University for providing samples from the patients. The authors appreciate assistance by Mr. Bryan Roderick Hamman for English language presentation of the present study.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Thailand Research Fund under Grant no. RSA6180017.
