Abstract
Objective
Pneumothorax after neurosurgical procedures is very rare and incompletely understood. This study was performed to explore the clinical characteristics and pathogenesis of pneumothorax after neurosurgery.
Methods
We retrospectively evaluated patients admitted from December 2016 to April 2019 for treatment of spontaneous intracranial hemorrhage. The inclusion criteria were neurosurgical procedures (open surgeries or endovascular intervention) performed under general anesthesia, no performance of central venous puncture during surgery, and occurrence of pneumothorax immediately after the neurosurgical procedure.
Results
Eight patients developed pneumothorax after neurosurgical procedures for spontaneous intracranial hemorrhage under general anesthesia. Of the eight patients, seven had aneurysmal subarachnoid hemorrhage and one had left temporal–parietal hemorrhage. The lung injury prediction score (LIPS) was 3, 4, 5, 6, and 9.5 in three, one, two, one, and one patient, respectively. During the operation, volume-controlled ventilation (tidal volume, 8–10 mL/kg) was selected for all patients.
Conclusions
Neurogenic pulmonary edema, inappropriate mechanical ventilation, and stimulation by endotracheal intubation might conjointly contribute to postoperative pneumothorax. To avoid this rare entity, mechanical ventilation with a low tidal volume or low pressure during general anesthesia should be adopted for patients with hemorrhagic cerebrovascular diseases involving the temporal lobe and a LIPS of >3.
Keywords
Introduction
In general, pulmonary surfactant stabilizes the size of the alveoli and prevents the alveoli from overexpanding and rupturing. The visceral pleura covers the surface of the lung. When lung disease or an external force induces rupture of the lung tissue and visceral pleura, or when a subtle emphysema bubble near the surface of the lung ruptures, escape of the air from the lung and bronchi into the pleural cavity can cause pneumothorax. 1 This gas compresses the lung tissue, and severely affected patients can develop breathing difficulties, low blood oxygen saturation, and carbon dioxide retention. This may progress to unconsciousness and coma; if not addressed in time, pneumothorax often causes death. 2
Lung disease is a common cause of pneumothorax. 3 However, pneumothorax caused by surgeries that are remote from the lung, such as shoulder arthroscopy, gastrostomy, and nephrectomy, is relatively rare.4–6 Pneumothorax is also rarely reported after craniotomy or craniectomy, and the mechanism is not fully understood. 7 In the present study, we evaluated the data of eight patients who developed pneumothorax immediately after undergoing neurosurgical interventions at our institution. To our knowledge, this is the largest case series ever reported in neurosurgical patients.
Materials and methods
We retrospectively evaluated the data of patients admitted to our institution from December 2016 to April 2019 for treatment of spontaneous intracranial hemorrhage. The inclusion criteria were the performance of a neurosurgical procedure (open surgery or endovascular intervention) under general anesthesia, no central venous puncture during surgery, and occurrence of pneumothorax immediately after the neurosurgical procedure.
Imaging examination
After hospitalization, computed tomography (CT), CT angiography, or digital subtraction angiography was performed as indicated to determine the cause of hemorrhage. The Hunt–Hess grade was determined in patients with subarachnoid hemorrhage (SAH). 8 Pulmonary CT was performed preoperatively for all patients. Acute lung injury or acute respiratory distress syndrome (ARDS) was predicted according to the lung injury prediction score (LIPS). 9 If pneumothorax was suspected postoperatively, a bedside X-ray or pulmonary CT scan was performed.
Intervention
Closed thoracic drainage was performed immediately upon confirmation of pneumothorax. Ventilator-assisted breathing, oxygen inhalation, and blood gas monitoring were then initiated. Before removal of the thoracic drainage tube, a chest CT scan was performed. The indications for thoracic drainage withdrawal were good pulmonary re-expansion, no air exiting the thoracic tube, no or only a small amount of pleural effusion, and well-controlled pneumonia.
Ethics
This study was approved by the Ethics Committee at The First Hospital of Jilin University (approval number: 2019-319). Informed consent for participation in the study or use of the medical data was obtained from each patient. Written consent was obtained from the patients for publication of this manuscript and any accompanying images.
Results
Preoperative condition
Eight patients (six women, two men) aged 41 to 64 years (mean, 50.1 years) were identified. Seven patients presented with SAH that was found to be secondary to a ruptured intracranial aneurysm. One patient presented with left temporal–parietal hemorrhage but had no other intracranial lesions that might lead to bleeding. The Hunt–Hess grade in the patients with aneurysmal SAH was II, III, and IV in three, three, and one patient, respectively. The LIPS was 3, 4, 5, 6, and 9.5 in three, one, two, one, and one patient, respectively. Of the seven patients with intracranial aneurysms, four had a middle cerebral artery aneurysm, one had an anterior communicating artery aneurysm, one had a posterior communicating artery aneurysm, and one had a carotid–ophthalmic artery aneurysm.
Intraoperative management
Endotracheal intubation and general anesthesia were performed for all patients. All patients underwent volume-controlled ventilation during the operation, and the tidal volume was 8 to 10 mL/kg. Of the seven patients with intracranial aneurysms, five underwent microsurgical clipping via the lateral supraorbital approach and two underwent endovascular coiling. The patient with left temporal–parietal hemorrhage underwent hematoma evacuation and craniectomy.
Pneumothorax confirmation and treatment
The endotracheal tube was retained before detection of pneumothorax for all patients. Four patients developed sudden dyspnea and were subsequently found to have pneumothorax during transfer from the operating bed to the transfer bed. The other four patients developed dyspnea and pneumothorax during transfer from the operation room to the ward.
Upon the occurrence of dyspnea and hypoxemia, simple respirator-assisted breathing was initiated, followed by a pulmonary CT scan (five patients) or bedside X-ray examination (three patients). Thoracic closed drainage was performed after confirmation of pneumothorax, and the time interval from diagnosis of pneumothorax to closed thoracic drainage was 12 to 50 minutes (mean, 28.8 minutes).
Prognosis
One patient died of cerebral infarction 1 day postoperatively. Among the remaining seven patients, the duration of thoracic drainage ranged from 3 to 25 days (mean, 11.4 days). Six patients underwent ventilator-assisted breathing for aggravation of lung injury. At the 3-month follow-up, the modified Rankin scale score was 0, 1, and 5 in five, one, and one patient, respectively. The patients’ detailed clinical information is shown in Table 1. Typical cases are presented in Figures 1 to 3.
Patients’ clinical data.
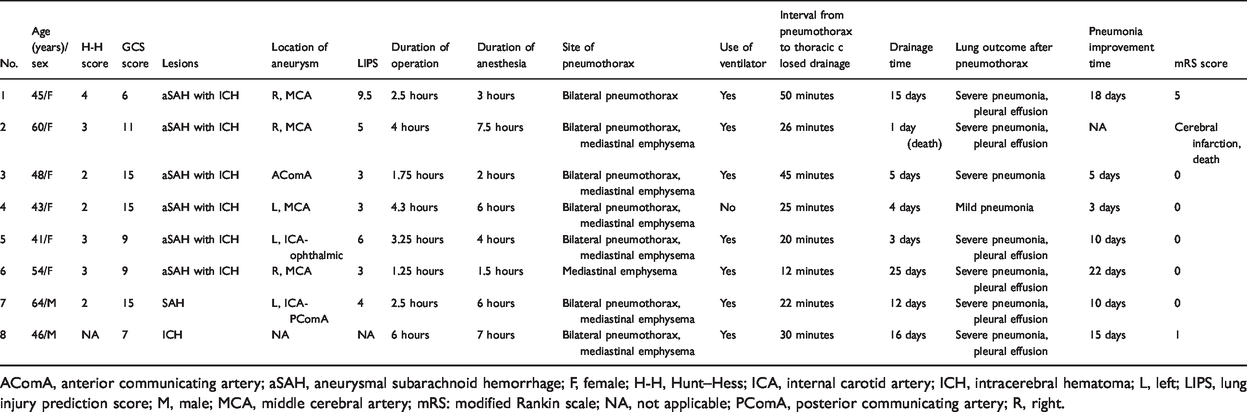
AComA, anterior communicating artery; aSAH, aneurysmal subarachnoid hemorrhage; F, female; H-H, Hunt–Hess; ICA, internal carotid artery; ICH, intracerebral hematoma; L, left; LIPS, lung injury prediction score; M, male; MCA, middle cerebral artery; mRS: modified Rankin scale; NA, not applicable; PComA, posterior communicating artery; R, right.
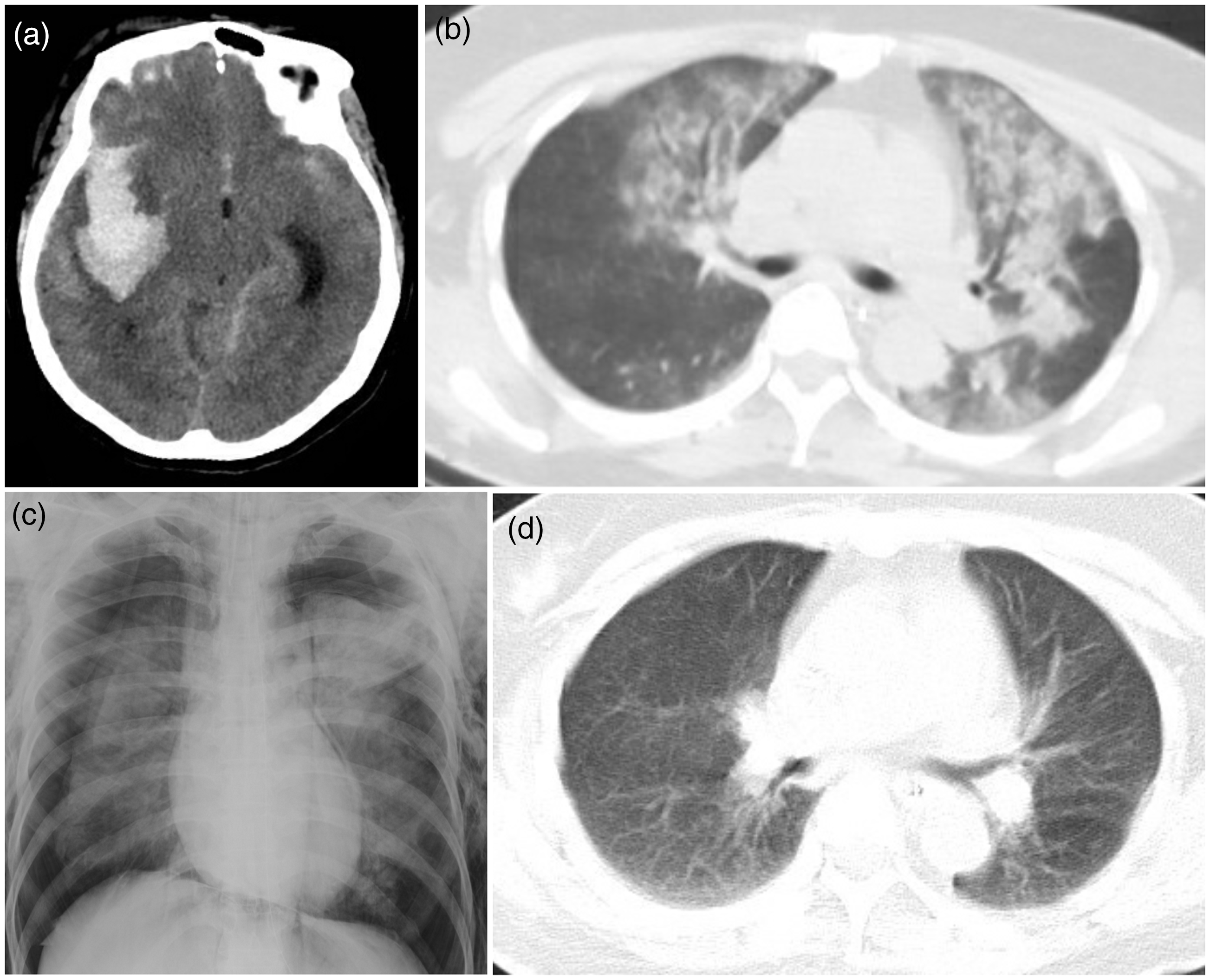
Typical case (Patient 1). (a) Head CT shows SAH and a hematoma in the right sylvian fissure. (b) Preoperative pulmonary CT shows patchy inflammation and exudation in the bilateral lungs. (c) Bedside X-ray of the lung after surgery shows that the bilateral lungs are squeezed by air. (d) Pulmonary CT shows resolution of pneumothorax and inflammation 1 month after thoracic closed drainage.

Typical case (Patient 6). (a) Head CT shows SAH and a hematoma in the right sylvian fissure. (b) Preoperative pulmonary CT shows scattered strip-like inflammation in the lower lobes of the bilateral lungs. (c) Pulmonary CT shows mediastinal emphysema and subcutaneous emphysema in the anterior chest wall. (d) Pulmonary CT 1 month after thoracic closed drainage shows complete resolution of the pneumothorax.
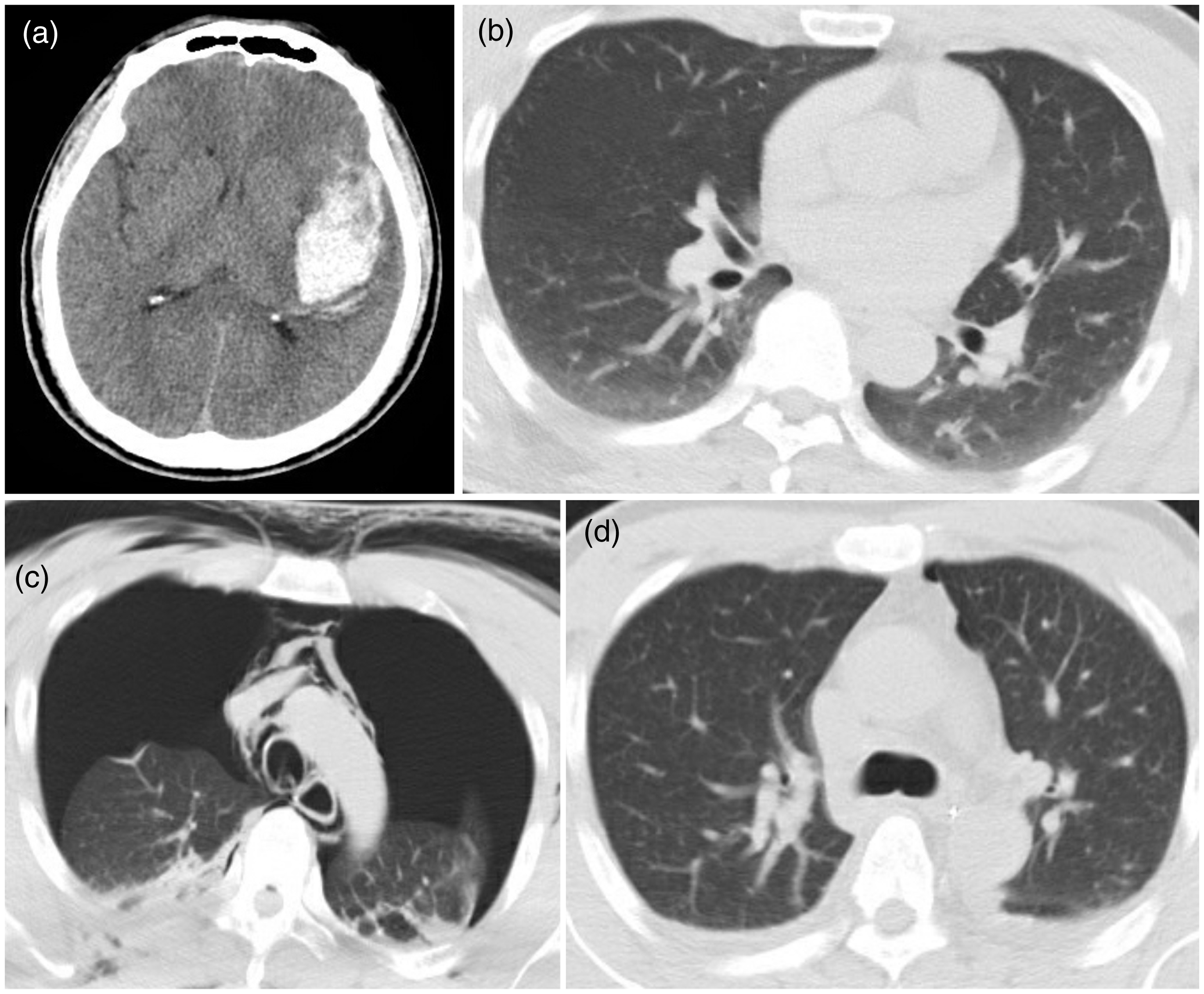
Typical case (Patient 8). (a) Head CT shows SAH and a hematoma in the left sylvian fissure. (b) Preoperative pulmonary CT shows patchy high-density shadows in the bilateral lungs. (c) Postoperative pulmonary CT reveals gas-induced collapse of lung tissue. Subcutaneous and mediastinal emphysema are also present. (d) Pulmonary CT 1 month after closed drainage shows complete resolution of pneumothorax.
Discussion
Pneumothorax is defined as gas accumulation in the pleural cavity. Spontaneous pneumothorax is caused by spontaneous rupture of lung tissue. Traumatic pneumothorax is caused by chest wall or lung trauma. 10 Iatrogenic pneumothorax is defined as traumatic pneumothorax due to medical interventions, such as thoracic puncture, bronchoscopic biopsy, central venous puncture, or mechanical positive-pressure ventilation. 5 The spontaneous occurrence of pneumothorax after neurosurgical procedures is extremely rare; only two cases have been reported to date.7,11 One patient had sustained penetration of the diaphragm during navigation of a ventriculoperitoneal shunt, and the other patient showed no direct pulmonary injury.
In the present study, we examined the data of eight patients who developed spontaneous pneumothorax after neurosurgical procedures under endotracheal intubation and general anesthesia. These patients developed pneumothorax during the postoperative process of waking up and being transferred. The cause of pneumothorax was unclear in these patients but is presumably associated with pneumonia, neurogenic pulmonary edema, or pulmonary embolism.12,13 Severe pneumonia and pulmonary edema can progress to ARDS. Pneumothorax can occur in patients with ARDS undergoing mechanical positive-pressure ventilation. 14 In this study, all patients had varying degrees of neurogenic pulmonary edema. Although no aspiration pneumonia was noted, the LIPS showed that the lung injury was severe and that the possibility of ARDS was very high. 9
Patients with intracranial hemorrhage might develop a sharp increase in the intracranial pressure and damage to the insula or thalamus. The locus coeruleus–sympathetic–adrenal medullary system is stimulated in such patients and produces a substantial amount of catecholamine, which is the neuroendocrine reaction during severe stress. This severe stress reaction can result in neurogenic pulmonary edema.15,16 In addition, the release of inflammatory mediators during systemic stress can cause damage to the alveoli. 17 Seven of the eight patients in this series had a hematoma in the temporal lobe, which might have compressed the insula and thalamus.
In patients with neurogenic pulmonary edema, volume-controlled ventilation (tidal volume of 8–10 mL/kg) during anesthesia can induce pneumothorax. 18 Hence, for patients with intracranial hemorrhage, the preoperative lung CT scan should be carefully evaluated. For those with neurogenic pulmonary edema, regardless of the degree of severity, protective ventilation with a lower tidal volume and appropriate positive end-expiratory pressure should be administered. For patients with an ideal body weight, the tidal volume should be set at 6 to 8 mL/kg, and the platform pressure should be kept at a low level. 19 If the platform pressure is sustained at a persistently high level, pressure-controlled ventilation should be selected. 20
All eight patients in this study received volume-controlled ventilation and had a high tidal volume, increasing the risk of lung injury. Although the patients did not exhibit symptoms of pneumothorax during general anesthesia, the use of an anesthesia apparatus might have caused further injury to the lung. During transfer of the patients, the endotracheal tube could have induced a bucking response and increased the pulmonary pressure. A substantial amount of gas would have entered the pleural cavity through the damaged lung tissue in such a case, causing pneumothorax.
Limitations
This was a retrospective study with a limited number of patients, and a statistical analysis could not be performed. Additionally, only patients with intracranial hemorrhage were included in the study; we excluded those admitted for treatment of other cerebral diseases, which might have compromised the conclusion. Finally, none of the patients in this series underwent bronchoscopy, which might have been helpful to explore the cause of pneumothorax.
