Abstract
Objective
There is growing evidence that ultraviolet B (UVB) irradiation can change the expression profile of microRNAs (miRNAs) in immortalized human epidermal melanocytes (Pig-1). We aimed to investigate the effect of miR-340 on regulating UVB-induced pigmentation.
Methods
Real-time quantitative PCR (qRT-PCR) was used to evaluate the expression of miR-340 in Pig-1 cells. Immunoblotting analysis, qRT-PCR, and luciferase reporter assays were used to detect the potential target of miR-340. The sodium hydroxide dissolution assay was used to assess the effect of miR-340 on changes in melanin content.
Results
Expression of miR-340 was reduced in human Pig-1 cells after UVB irradiation. We found a negative correlation between miR-340 and melanocyte inducing transcription factor (MITF) in Pig-1 cells after UVB irradiation. Knockdown and overexpression of MITF in Pig-1 cells down- and upregulated melanogenesis, respectively. Overexpression of miR-340 inhibited MITF expression, reduced the amount of melanin, and suppressed expression of multiple key molecules involved in the pigment synthesis pathway, whereas knockdown of miR-340 showed the opposite results.
Conclusions
Our results showed that miR-340 inhibited melanogenesis by regulating the downstream molecules of MITF and its signaling pathways, suggested that miRNA-340 may be a new target for the clinical treatment of UVB-induced pigmentation.
Keywords
Introduction
A variety of endogenous and exogenous factors can affect skin color, such as the proportion of various pigments. 1 For most pigment in the skin, 2 the synthesis and distribution of melanin are the determining factors. 3 Melanin is derived from tyrosine catalyzed by tyrosinase in melanocytes (pigment-producing cells), 4 and the activity of tyrosinase is affected by many factors, the most important of which is exposure to ultraviolet B (UVB) radiation.5,6 UVB radiation on the skin promotes the transportation of melanosomes (melanin-containing granules) from melanocytes to the surrounding keratinocytes, where most pigment is found. 7 UVB can also activate tyrosinase in melanocytes, leading to an increase in the number of melanocytes, which is one of the most important factors in pigmentation. 8 Hyperpigmentation skin diseases associated with UVB are very common in clinical dermatology, and include chloasma, freckles, and sunspots. 9 These diseases often have a hidden onset, are difficult to treat, and seriously affect the normal social and quality of life of patients. To improve the prognosis of UVB-induced pigmentation skin diseases, the first steps are to study their pathogenesis and identify effective therapeutic targets for treatment.
MicroRNAs (miRNAs) are a class of single-stranded, small, noncoding RNAs, 21 to 23 nucleotides long, that induce mRNA degradation of target genes, inhibiting protein translation by binding to the 3′-untranslated region (UTR) of mRNAs. 10 Studies have shown that the expression profiles of a large number of miRNAs are changed after UV irradiation, which plays roles in the regulation of DNA damage, photoaging, cell survival, carcinogenesis, and pigmentation, among others.11,12 By means of miRNA microarrays, differential miRNA expression profiles have been investigated, 13 and pronounced expression changes of miRNAs are known to occur within the first hours following UVB irradiation, which indicates an earlier regulation of miRNAs than most transcriptional responses. 14 A number of studies have shown a relationship between miRNAs and skin.15–17 Although early studies demonstrated a functional role of miRNA-340 (miR-340) in regulating brain morphogenesis 18 and modulating reorganization of the actin cytoskeleton, 19 key roles for miR-340 in regulating UVB-induced melanosome transport 20 and UVB-mediated retinal pigment epithelium cell damage 21 have been reported recently. However, the functional roles and potential molecular mechanisms of miR-340 in UVB-induced pigmentation skin disease are underinvestigated.
Melanocyte inducing transcription factor (MITF) is a transcription factor involved in regulation of many types of cells, including melanocytes, osteoclasts, and mast cells. 22 Various studies have demonstrated that the functional roles of MITF in targeting genes involved in melanin formation and normal melanocyte development depend on three major pigmentation enzymes: tyrosinase (TYR), tyrosine related protein 1 (TYRP1), and dopachrome tautomerase (DCT).23–25 A report showed that in melanoma cells, miR-340 can bind specifically with the 3′-UTR of MITF and mediate the degradation of its mRNA, reducing the expression and activity of MITF protein. 26 However, the mechanism of MITF in UVB-induced pigmentation has not been fully elucidated.
In the present study, we investigated the effect of miR-340 on UVB-induced pigmentation, identified the downstream regulator MITF, investigated the roles of miR-340 and MITF in the regulation of pigmentation, and explored the potential underlying mechanisms. The aim of this study was to provide a basis and new target for individualized treatment of UVB-induced pigmentation skin disease by exploring the regulatory mechanisms of miR-340 and MITF in the occurrence and development of melanocytes.
Materials and methods
Ethical statement
This study did not involve animal or human subjects; thus, ethical permission from the relevant institutional review boards was not applicable.
Cell culture
The immortalized human epidermal melanocyte cell line Pig-1 was used in this study. The cell line was cultured in HEMA-LP PROLIF (M254/HMGS-2, Invitrogen, Carlsbad, CA, USA) with 5% (vol/vol) fetal bovine serum (HyClone, Logan, UT, USA). Cell cultures were incubated in a constant-temperature incubator at 37°C under 5% CO2.
UVB irradiation
For UVB irradiation of cells, the culture medium was replaced with PBS. Cells were then exposed to 100 mJ/cm2 irradiation from a germicidal 15-W UV lamp (305 nm) at a distance of 7 cm for different times up to 24 hours. After UVB irradiation, cells were cultured in fresh medium for another 24 hours.
RNA preparation and real-time quantitative PCR
The miRNAs were extracted in accordance with the manufacturer’s instructions (Qiagen, Hilden, Germany). The concentration and quality of total RNA were measured by UV absorbance at 260 and 280 nm and confirmed by gel electrophoresis. The primer sequences 5′-TGAGTCTTCAAGAGGGAGCC-3′ (forward) and 5′-GTTGTTCAACGGGCCATGAT-3′ (reverse) were used to detect the content of miR-340 with the Reverse Transcription System Bestar qPCR RT kit (#2220, DBI Bioscience, Shanghai, China) and ABI 7500 Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). Additionally, sample RNAs were extracted by using Trizol reagent (Invitrogen). The cDNAs were synthesized using the Reverse Transcription System Bestar qPCR RT kit in accordance with the manufacturer’s instruction and using the ABI 7500 Real-Time PCR System (Applied Biosystems). GAPDH and U6 were used as internal reference genes. The primer sequences were as follows: MITF, 5′-ATAATGAGGAAATCTTGGGCTTG-3′ (forward) and 5′-GGAGTTGCTGATGGTGAGG-3′ (reverse); TYR, 5′-GCCAACGATCCTATCTTCCTTC-3′ (forward) and 5′-GCCAACGATCCTATCTTCCTTC-3′ (reverse); TYRP1, 5′-TAGTGACGAGCCTCTTGACAA-3′ (forward) and 5′-CTGGCAGTTAGACTGGGAGA-3′ (reverse); GAPDH, 5′-TGACTTCAACAGCGACACCCA-3′ (forward) and 5′-CACCCTGTTGCTGTAGCCAAA -3′ (reverse); U6, 5′-CTCGCTTCGGCAGCACA-3′ (forward) and 5′-AACGCTTCACGAATTTGCGT-3′ (reverse). Data analysis was performed using the 2−ΔΔCt method.
Western blot
The cells were collected and flash-frozen in liquid nitrogen; then, an ultrasonic cell-break method was adopted (2 × 5 s) in 50 mM lysis buffer (20 mM Tris at pH 7.5, 150 mM NaCl, 1 mM phenylmethylsulfonyl fluoride, 10 mM β-glycerophosphate, 1% Triton X-100, 5 mM EDTA, 0.2 mM Na3VO4, 2 µg/mL leupeptin, and 2 µg/mL pepstatin A) on ice. Homogenates were centrifuged at 12,000 × g at 4°C for 30 minutes and the supernatants were collected. Protein lysates (30 μg) were loaded onto sodium dodecyl sulfate-polyacrylamide gels for electrophoresis and transferred to polyvinyl difluoride (PVDF) membranes. The PVDF membranes were incubated overnight with the primary antibody as follows: monoclonal antibodies (1:1000, Santa Cruz Biotechnology, Santa Cruz, CA, USA) against MITF, TYR, and TYRP1 solute in PBS-Tween, followed by blocking in 5% bovine serum albumin. The membranes were then washed with Tris-buffered saline-Tween (10 minutes × 3 times) and probed with the appropriate secondary antibody (1:5000; Abcam, Cambridge, UK). Immunoreactivity was determined and observed using enhanced chemiluminescence (Millipore, Billerica, MA, USA). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a control.
Sodium hydroxide dissolution assay
Melanin contents were measured as described previously. 27 Cells in different treatment groups were dissolved in 1 M NaOH at 60°C for 2 hours and centrifuged for 10 minutes at 10,000 × g. The optical density of each supernatant was measured at 405 nm using an ELISA reader.
Plasmid construction and transfection
The following sequences were synthesized: miR-340 inhibitor, 5′-AAUCAGUCUCAUUGAUUUAUAA-3′; miR-340 mimic, 5′-UUAUAAAGCAAUGAGACUGAUU-3′; MITF-small interfering RNA (si-MITF), sense 5′-UCAUCAAACAAGAACCUGUdTdT-3′ and antisense 5′-ACAGGUUCUUGUUUGAUGAdTdT-3′; negative control (NC), 5′-UUGUACUACACAAAAGUACUG-3′. PCR was used to obtain the sequence of MITF. Expression plasmids GV268 and GV272 were then constructed and sequenced for miR-340 and MITF, respectively. Plasmids were transfected into cells using Lipofectamine 2000 (Invitrogen).
Target prediction and luciferase assay
By the means of the TargetScan (http://www.targetscan.org) website, we predicted the putative targets of miR-340. We then constructed wild-type (WT) and miR-340 binding site mutants (MUT) of MITF 3′-UTR reporter vectors with the primers 5′-GATCGCCGTGTAATTCTAGAAAGAGGAAAAGAAATCCATAC-3′ (forward) and 5′-CCGGCCGCCCCGACTCTAGAGGTTGGTATCAGTTATGTTTTC-3′ (reverse). Each sequence was subcloned into the psiCHECK2 vector, and the plasmids were designated MITF-WT and MITF-MUT after sequencing. For the luciferase assay, cells were seeded in 6-well plates 24 hours before transfection and then transiently transfected with 0.3 μg of MITF-WT or MITF-MUT and 20 pmol of miR-340 mimic or control miRNA. Cotransfections were performed using Lipofectamine 2000 (Invitrogen/Life Technologies), in accordance with the manufacturer’s protocol. Twenty-four hours after transfection, the cell culture medium was removed and cells were lysed in passive lysis buffer (Promega, Madison, WI, USA). The firefly and Renilla luciferase activities were measured consecutively using the dual-luciferase assay system (Promega) in accordance with the manufacturer’s instructions.
Statistical analysis
All data are presented as mean ± standard deviations. All experiments were performed independently at least 3 times. We calculated the statistical significance using one-way analysis of variance followed by Duncan’s multiple-comparison test using SPSS version 19.0 (IBM Corp., Armonk, NY, USA). P < 0.05, P < 0.01, or P < 0.001 were regarded as statistically significant.
Results
Melanin content variation after UVB irradiation
To determine the functional role of miR-340 on UVB-induced pigmentation, Pig-1 cells were exposed to 100 mJ/cm2 UVB irradiation and cells were observed at different time intervals. Six hours after irradiation, melanocytes were significantly shrunken compared with cells in the control group, the number of dendrites was reduced, and their length was shortened (Figure 1a). Sixteen hours after irradiation, morphological changes induced by UVB irradiation within Pig-1 cells were evident, with significant elongation of dendrites and a marked increase in the number of dendrites compared with controls and cells at 6 hours, as well as an increase in the number of pigment particles in the cells (Figure 1a). More and longer dendrites were observed 24 hours after irradiation (Figure 1a). We also measured melanin content using the sodium hydroxide dissolution assay. A marked increase of melanin content was observed after irradiation, with 1.4 times as much melanin in irradiated cells as in the control group at 24 hours (Figure 1b). Together, these results suggested that UVB irradiation promoted melanocyte dendricity and increased melanin content.

Variation in melanin content after ultraviolet B (UVB) irradiation of immortalized human epidermal melanocytes (Pig-1 cells). (a) Morphological changes induced by UVB irradiation within Pig-1 cells. Black arrows show typical cell morphology. (b) Assessment of melanin content after UVB irradiation at each time point. *P < 0.05, **P < 0.01.
miR-340 and MITF are negatively correlated after UVB irradiation
We then determined expression levels of miR-340 in Pig-1 cells subjected to UVB irradiation using quantitative real-time PCR (qRT-PCR). Upregulation of miR-340 was detected 3 hours after irradiation and peaked at 6 hours compared with that in untreated cells; expression of miR-340 decreased significantly at 12 hours and continued to decrease from 12 to 24 hours (P < 0.05) (Figure 2a). Expression of MITF was also evaluated by qRT-PCR (Figure 2b) and western blot analysis (Figure 2c). Expression of MITF mRNA decreased 3 hours after irradiation, reached its lowest level at 6 hours compared with untreated cells, and then increased from 12 hours, in the opposite pattern to miR-340 (P < 0.05) (Figure 2b). Similar results were obtained by western blot (Figure 2c). Thus, a negative correlation between miR-340 and MITF was found in Pig-1 cells following UVB irradiation.

Negative correlation between miR-340 and expression of MITF (melanocyte inducing transcription factor) after ultraviolet B (UVB) irradiation. (a) Change in expression of miR-340 in Pig-1 cells after UVB irradiation. (b) Change in expression of MITF mRNA in Pig-1 cells after UVB irradiation. (c) Change in MITF protein in Pig-1 cells after UVB irradiation (GAPDH used as control). *P < 0.05.
Overexpression of miRNA-340 inhibited the protein level of MITF
Given that MITF functions as a crucial transcription factor involved in regulation of melanocytes, we utilized computational prediction in TargetScan to identify potential binding sites for miR-340 on MITF. We found that the 3′-UTR of MITF mRNA bears the miR-340 binding site (Figure 3a). We then designed a dual-luciferase assay to verify the effect of miR-340 on translation of MITF mRNA. Co-transfection of MITF with wild-type 3′-UTR and miR-340 showed that expression of luciferase decreased significantly compared with that of the NC group (P < 0.05) (Figure 3b), whereas co-transfection MITF with mutant 3′-UTR and miR-340 showed similar expression to the NC group (Figure 3b). These results confirmed that miR-340 may inhibit the expression of MITF through specific complementary base pairing to the predicted binding site in the 3′-UTR of MITF.
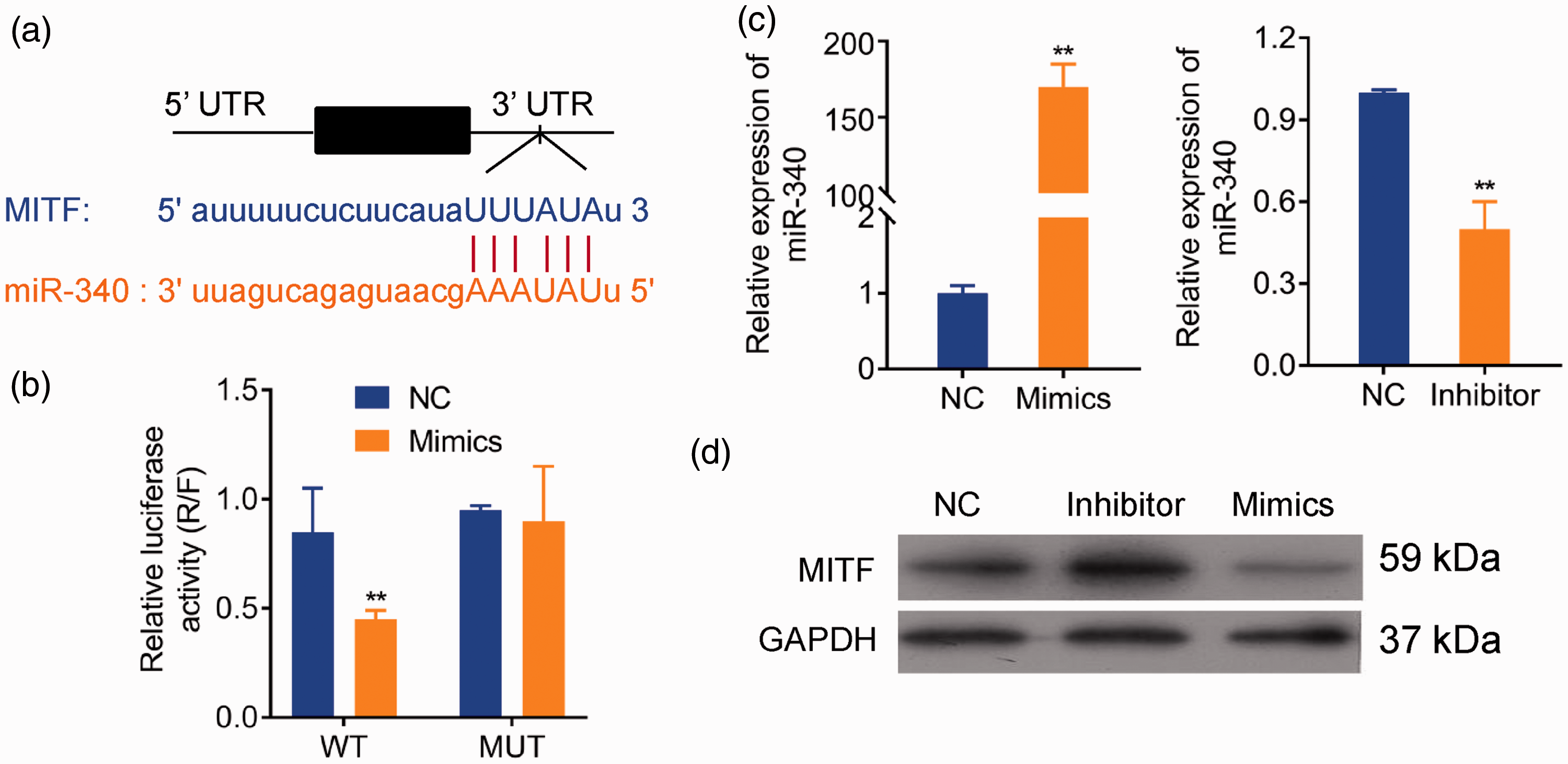
Overexpression of miRNA-340 inhibited the protein level of MITF (melanocyte inducing transcription factor). The time point of Pig-1 cells induced by ultraviolet B (UVB) is 24 hours. (a) Predicted binding sites for miR-340 in the 3′-untranslated region (UTR) of MITF. (b) Dual-luciferase reporter (R = Renilla; F = firefly) assay of MITF 3′-UTR-wildtype (WT) and mutant (MUT) with miR-340 in Pig-1 cells. (c) Transfection efficiency of miR-340 mimics and inhibitor determined by real-time quantitative PCR (qRT-PCR). (d) Western blot analysis of MITF transfected with miR-340 mimics and inhibitor plasmids. *P < 0.05, **P < 0.01.
We then constructed miR-340 mimics and miR-340 inhibitor and detected the efficiency of their transfection in Pig-1 cells, with upregulation for miR-340 mimics and downregulation for miR-340 inhibitor (P < 0.05) (Figure 3c). Western blot analysis further confirmed that the protein level of MITF was upregulated by miR-340 inhibitor transfection and downregulated by mimics (Figure 3d). These results showed that MITF was the target for miR-340 by binding to the 3′-UTR.
MITF promotes UVB-induced pigmentation
MITF expression in pcDNA-MITF and si-MITF, respectively, was evaluated by qRT-PCR analysis (Figure 4a) and western blot (Figure 4b). MITF expression in Pig-1 cells transfected with pcDNA-MITF and si-MITF was significantly increased and decreased, respectively. The melanin contents of irradiated Pig-1 cells were determined by sodium hydroxide dissolution assay; melanin content in MITF siRNA transfected cells was significantly increased compared with control (P < 0.05) (Figure 4c), whereas melanin content was dramatically decreased in the MITF overexpression group compared with controls (P < 0.05) (Figure 4c). These results confirmed the inhibition of UVB-induced pigmentation by MITF.

MITF (melanocyte inducing transcription factor) promotes UVB-induced pigmentation. The time point of Pig-1 cells induced by ultraviolet B (UVB) is 24 hours. (a) Transfection efficiency of MITF overexpression (OE) and knockdown (small interfering MITF, si-MITF) were determined by qRT-PCR. (b) Transfection efficiency of MITF overexpression and knockdown were determined by western blotting. (c) Detect of melanin content changes by MITF overexpression and knockdown after UVB irradiation. *P < 0.05, **P < 0.01.
miR-340 inhibited UVB-induced pigmentation by targeting MITF
No significant changes were detected for TYR mRNA (Figure 5a) and protein (Figure 5b) at each time point following UVB irradiation. TYR may be involved in multiple signaling pathways and be affected by many molecules, and its effect of inducing increased pigment synthesis may be mainly related to increased activity of tyrosinase rather than an increased quantity of tyrosinase. Therefore, expression changes in TYR were not particularly noticeable after irradiation. The mRNA (Figure 5a) and protein (Figure 5b) levels of TYRP1 began to increase 3 hours after UVB irradiation, peaked at 16 hours, and decreased at 24 hours, similar to the trend observed for MITF, further suggesting that TYRP1 was regulated by MITF during the signal pathway of pigment synthesis. Melanin content data (Figure 5c) demonstrated that after 24 hours of UVB irradiation, Pig-1 cells transfected with miR-340 inhibitor had significantly increased melanin content compared with the UVB irradiated cells transfected with miRNA inhibitor control, whereas miR-340 mimics had decreased melanin content, suggesting that miR-340 inhibited UVB-induced pigmentation. The mRNA (Figure 5d) and protein (Figure 5e) expression of TYR and TYRP1 were significantly decreased in the miR-340 mimic group and increased in the miR-340 inhibitor group.

miR-340 inhibited ultraviolet B (UVB)-induced pigmentation by targeting MITF (melanocyte inducing transcription factor). The time point of Pig-1 cells induced by UVB is 24 hours. (a) Change of TYR and TYRP1 mRNA in Pig-1 cells after UVB irradiation. (b) Change of tyrosinase (TYR) and tyrosine related protein 1 (TYRP1) protein in Pig-1 cells after UVB irradiation. (c) Effects of miR-340 mimics and inhibitor on melanin content after UVB irradiation. (d) Effects of miR-340 mimics and inhibitor on TYR and TYRP1 mRNA after UVB irradiation. (e) Effects of miR-340 mimics and inhibitor on TYR and TYRP1 protein expression after UVB irradiation. *P < 0.05, **P < 0.01, ##P < 0.01.
Discussion
The miRNAs are a class of endogenous, small, noncoding single-stranded RNAs that are highly conserved during evolution. A complex network of growth factors, transcription factors, and other factors regulates a variety of biological processes in cells and inhibits the expression of genes at the post-transcriptional level.28–30 miRNAs can bind specifically to the 3′-UTR of target genes, regulate about 30% of human genes, reduce the stability of mRNA and promote its degradation, inhibit corresponding protein translation, and affect the functional expression of genes.31,32 To date, studies have shown a close connection between miRNA, UVB irradiation, 33 and skin.15–17 Although several studies have examined functional roles for miRNAs in the regulation of pigmentation of different species,34–36 to our knowledge, such a role for miRNAs in the regulation of genes critical to pigmentation has not been demonstrated in normal adult melanocytes. Moreover, in melanoma cells, miR-340 can bind to the 3′-UTR of MITF mRNA and mediate the degradation of its mRNA, reducing the expression and activity of MITF protein. 26 No research has yet reported the effects of the binding of miR-340 to the 3′-UTR of MITF on pigment synthesis and UVB-induced pigmentation. UVB irradiation is believed to be the main stimulation for skin pigmentation,37,38 and hyperpigmentation skin diseases related to UV irradiation are very common in clinical practice. 39 Therefore, an in-depth understanding of the pathogenesis of UVB-induced pigmentation at the molecular level will help in the search for new targets for prognosis evaluation and therapy.
In this study, we first confirmed the appearance of morphological changes induced by UVB irradiation in Pig-1 cells; these included significant elongation of dendrites and a marked increase in the number of dendrites, consistent with previous reports that UVB irradiation promotes melanocyte dendricity. 20 Moreover, melanocyte dendrites contribute to skin darkening; 40 in the present study, UVB irradiation increased the number of pigment particles in Pig-1 cells. As a key regulator of dendrite formation, miR-340 was upregulated 3 hours after UVB irradiation. 20 We also showed that UVB irradiation could increase miR-340 expression. Interestingly, the increased miR-340 expression decreased 6 hours after irradiation, opposite to the expression of MITF, which increased 6 hours after irradiation. Considering that MITF participates in melanin synthesis, 41 the negative correlation between miR-340 and MITF was suggestive of a potential important functional role for miR-340 in regulation of UVB-induced pigmentation.
Notably, by computational prediction, we found that the 3′-UTR of MITF mRNA bears the miR-340 binding site. A dual-luciferase assay confirmed the inhibiting ability of miR-340 on translation of MITF mRNA, similar to a previous report. 26 Although a functional role for miRNA in the regulation of MITF expression has been previously reported,42,43 expression of miR-25 and miR-137 in melanoma cell lines downregulated MITF expression, in a similar manner to miR-340 in this study. miR-340 was reported to involve UVB-induced dendrite formation; 20 however, to our knowledge, a potential functional role for miR-340 in UVB-induced pigmentation has not been documented previously.
MITF has been found to stimulate transcriptional activity of Rab27A to restore melanosome transport, facilitating pigmentation. 44 Inhibition of MITF results in suppression of melanin synthesis. 45 The present study also showed that knockdown of MITF reduced the melanin content, whereas overexpression of MITF dramatically increased melanin content. In a previous study, expression of TYR (a downstream target of MITF) was increased, whereas TYRP1 and DCT were not significantly changed by sarsasapogenin treatment to increase melanin synthesis. 41 Downregulation of TYR and TYRP1 was accompanied by a decrease in melanin synthesis. 46 However, in the present study, we showed that downregulation of miR-340 could relieve the degradation and inhibition of MITF and strongly promote the expression of TYR and TYRP1 mRNA to promote melanin synthesis. The inconsistency between TYRP1 expression level might be caused by TYR enzyme activity. Therefore, measurement of TYR activity is needed in future studies. These findings revealed mechanistic insights for miR-340 as well as a novel regulatory role for MITF in UVB-induced pigmentation. However, this mechanism needs further study because of the lack of in vivo data in patients with UVB-induced pigmentation skin disease. Future studies to evaluate the role of the miR-340/MITF axis in a clinical context are warranted.
To the best of our knowledge, this is the first study to show that miR-340 inhibited UVB-induced pigmentation by specifically targeting MITF. We showed that miR-340 could combine with the 3′-UTR of MITF to induce degradation and inhibit the expression of MITF. miR-340 also downregulated proteins related to pigment synthesis signaling pathways and reduced pigment synthesis induced by UVB. Understanding the regulatory mechanism of miR-340 in UVB-induced pigmentation could help identify useful clinical biomarkers. In addition, miR-340 had an efficient protective effect against UVB-induced pigmentation, indicating that miR-340 might be a potential therapeutic target for developing a novel approach to treat skin pigmentation diseases.
Footnotes
Author contributions
Study design/planning: YY and CL; data collection/entry: YY and YH; data analysis/statistics: YY; data interpretation: XW, JB, MH, TH, YH, and CL; preparation of manuscript: YY, XW, JB, MH, TH, YH, YW, and CL; literature analysis/search: XW, JB, MH, TH, YW, and CL; manuscript revision: YY and CL; funding collection: CL. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
