Abstract
This case report describes a 43-year-old female initially diagnosed with gestational trophoblastic neoplasia that then experienced metastasis to the liver and then subsequently to the pancreas nearly 4 years after the primary diagnosis. After resection of the body and tail of the pancreas, the postoperative histopathological examination confirmed a placental site trophoblastic tumour that had developed after several cycles of chemotherapy for the original primary tumour and the liver metastases. This type of sequential recurrence of gestational trophoblastic neoplasia in the primary site or metastatic sites, such as the liver or pancreas, can be cured by a comprehensive treatment strategy involving surgery and/or salvage chemotherapy and continuous follow-up over a long period, especially for patients with a high-risk status.
Introduction
Gestational trophoblastic neoplasia (GTN) includes invasive mole, choriocarcinoma, placental site trophoblastic tumour (PSTT) and epithelioid trophoblastic tumour. 1 Choriocarcinoma is a highly malignant type of gestational trophoblastic neoplasia that secretes high levels of human chorionic gonadotropin (hCG) and it is sensitive to chemotherapy. 2 It can rapidly progress and metastasize via the haematogenous pathway to distant organs. 3 PSTT is a very rare type of GTN that only accounts for approximately 0.2% of cases and it originates from intermediate trophoblastic cells. 4 Choriocarcinomas can be subdivided into gestational and non-gestational choriocarcinomas. Non-gestational choriocarcinoma is also known as extragonadal choriocarcinoma or primary choriocarcinoma. 5 A review of the literature showed that GTN metastases to the pancreas are rarely mentioned. This current report describes a very rare case of GTN showing sequential metastasis to the liver and pancreas.
Case report
A 43-year-old female presented to the Department of Hepatobiliary and Transplantation Surgery, The First Affiliated Hospital of University of Science and Technology of China, Hefei, Anhui Province, China because of a mass in the body of the pancreas that was detected by enhanced computed tomography (CT) in May 2018 (Figures 1A–1C). The patient also demonstrated rising serum levels of the β-subunit of hCG (β-hCG), which had been monitored since her earlier hospitalization (see below) (Figure 2).

Abdominal computed tomography scans of a 43-year-old female showing a mass in the body of the pancreas (arrow) that was highly suspected to be a pancreatic metastasis from the original primary gestational trophoblastic neoplasia (a–c); and the abdomen again at 2 months after distal pancreatectomy (d–f).

Serial assessment of a 43-year-old female patient’s serum levels of the β-subunit of human chorionic gonadotropin (β-hCG) during the course of her disease from her earlier diagnosis with gestational trophoblastic neoplasia to her most recent medical assessment. FAV, floxuridine + actinomycin-D + vincristine; FAVE, floxuridine + actinomycin-D + vincristine + etoposide; EMP, etoposide +methotrexate + platinum; EMA-CO, etoposide + methotrexate + actinomycin-D + cyclophosphamide + vincristine; TP, paclitaxel + cisplatin. The colour version of this figure is available at: http://imr.sagepub.com
This patient was originally admitted to The First Affiliated Hospital of University of Science and Technology of China with persistent elevation of β-hCG and was diagnosed with GTN (I:7, according to the International Federation of Gynaecology and Obstetrics [FIGO 2000 scoring system]) in December 2013. 6 She had a history of drug-induced abortion in 2007. Subsequently, she attended The First Affiliated Hospital of University of Science and Technology of China for further treatment in January 2014, which included fertility-sparing therapy and six sequential cycles of chemotherapy; five times with the floxuridine + actinomycin-D + vincristine (FAV) regimen (1.75 g floxuridine intravenous [i.v.] drip per day [days 1–6] + 350 µg actinomycin-D i.v. drip per day [days 1–6] + 2 mg vincristine i.v. drip once on day 1) followed by one cycle of the floxuridine + actinomycin-D +vincristine + etoposide (FAVE) regimen (1.25 g floxuridine i.v. drip per day [days 1–5] + 300 µg actinomycin-D i.v. drip per day [days 1–5] + 2 mg vincristine i.v. drip once on day 1 + 0.16 g etoposide i.v. drip per day [days 1–5]). The last two cycles of chemotherapy were for consolidation. The patient achieved a stable, normal level of serum β-hCG (<3.1 IU/l) over the following 30 months (until June 2016; Figure 2) and was then lost to follow-up. In December 2016, the patient attended the outpatient clinic for a routine follow-up appointment and her β-hCG was 6.59 IU/l, but she decided not to see the gynaecological oncologist. However, in May 2017, she was sent to the Emergency Department of The First Affiliated Hospital of University of Science and Technology of China because of recurrent syncope and a continuous dull pain in the epigastric abdominal area for the previous 12 h. Physical examination of the patient revealed pale appearance, tachycardia, hypotension and tenderness in the right abdomen. Subsequently, CT and B-ultrasound examinations suggested multiple liver masses and intra-abdominal fluid accumulation. A haematoma measuring 2.2 cm ×1.76 cm in the anteroposterior and transverse dimensions was suspected (Figures 3A–3C). Laboratory examinations showed a highly elevated level of serum β-hCG (148 IU/l) and decreased levels of haemoglobin (77 g/l) and haematocrit (0.24 l/l). She was diagnosed with rupture and bleeding from a mass suspected to be a liver metastasis from the original GTN. She was treated with embolization of a hepatic artery branch identified to be the feeder of the active haemorrhage. One month later (in June 2017), she was treated with four cycles of chemotherapy with the etoposide +methotrexate + platinum (EMP) regimen (160 mg etoposide i.v. drip per day [days 1–3] + 40 mg methotrexate i.v. drip once on day 1 + 140 mg nedaplatin i.v. drip once on day 2); followed by two cycles of the etoposide + methotrexate + actinomycin-D + cyclophosphamide + vincristine (EMA-CO) regimen (180 mg etoposide i.v. drip per day [days 1–2] + 540 mg methotrexate i.v. drip once on day 1 + 500 µg actinomycin-D i.v. drip per day [days 1– 2] + 1.2 g cyclophosphamide i.v. drip once on day 8 + 2 mg vincristine i.v. drip once on day 8) as consolidation chemotherapy with the patient having a stage/score of IV:13. Gradual decreases in the serum β-hCG levels and a shrinking liver mass in the CT scan indicated considerable sensitivity of the tumour to the chemotherapy regimens that were used (Figures 3D–3F).
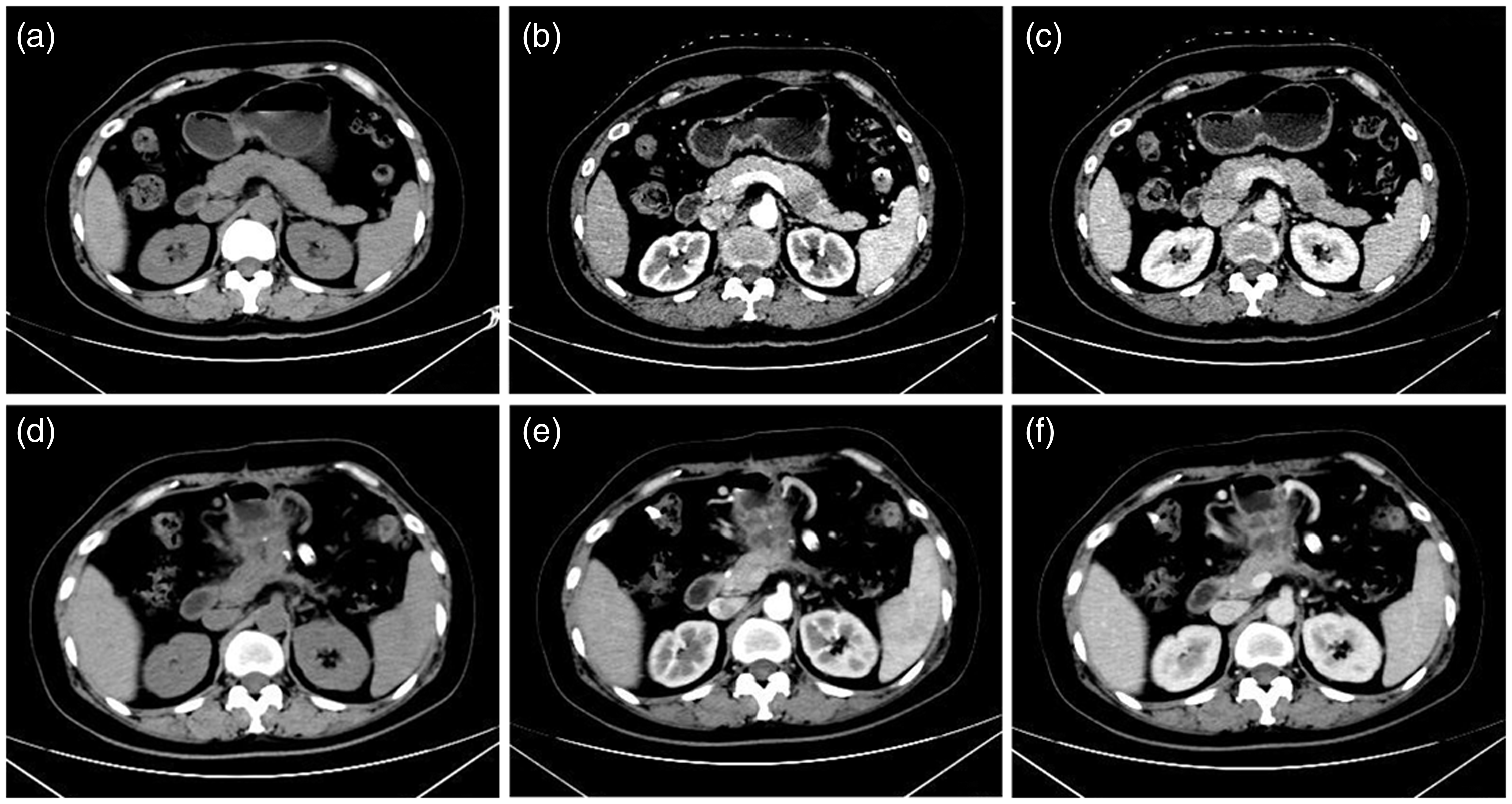
Abdominal computed tomography scans of the same patient from May 2017 showing multiple liver masses and intra-abdominal fluid accumulation. A haematoma (arrow) measuring 2.2 cm × 1.76 cm in the anteroposterior and transverse dimensions was suspected. The patient was diagnosed with rupture and bleeding from a mass suspected to be a liver metastasis from the original primary gestational trophoblastic neoplasia (a–c); and the abdomen again at 1 year after the hepatic artery branch embolization operation showing shrinkage of the metastatic lesions (d–f).
Based on the patient’s medical history and the serum β-hCG level (14.62 IU/l), the mass in the body of the pancreas was suspected to be a GTN metastasis. The patient was treated with two cycles of the EMA-CO regimen and achieved a level of 3.33 IU/l of β-hCG (Figure 2). Following chemotherapy, the patient still presented with a visible mass in the distal part of the pancreas. Examination by the multidisciplinary team resulted in the decision to resect the body and tail of the pancreas with preservation of the spleen and the procedure was undertaken in the Department of Hepatobiliary and Transplantation Surgery, The First Affiliated Hospital of University of Science and Technology of China (Figures 1A–1C). The postoperative pathological report of the mass that measured 1.2 cm × 1.6 cm × 1.6 cm indicated a metastatic PSTT that had the following immunohistochemical phenotype: human placental lactogen-positive; cytokeratin-positive; CD10-positive; Ki-67-positive (proliferation index of 22%); focal hCG-positive; P63-negative; CD34-negative; and vimentin-negative (Figure 4). Following a rapid recovery from the operation (normal laboratory values and no signs of diabetes mellitus), the patient underwent five cycles of the docetaxel and cisplatin (TP) chemotherapy regime (260 mg paclitaxel i.v. drip once on day 1 + 110 mg cisplatin i.v. drip once on day 1, every 28 days) for consolidation chemotherapy starting on postoperative day 20. Routine follow-up of the serum β-hCG level and abdominal and pelvic CT scans at 2, 6, 9, 12 and 18 months did not reveal any evidence of recurrence of the PSTT (Figures 1D–1F). The patient provided written informed consent for publication of this case report.
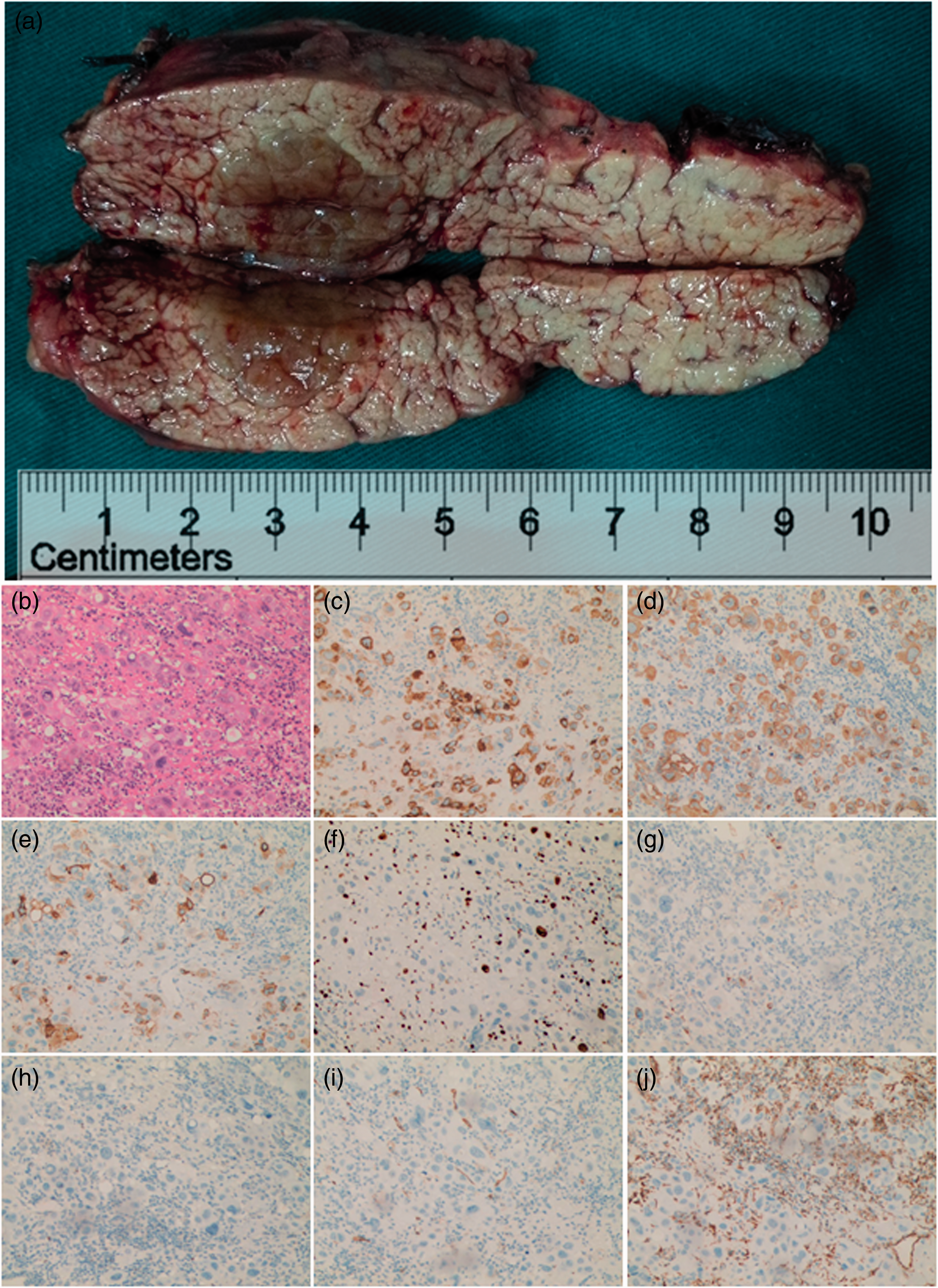
Histopathological examination of the resected specimen from the distal pancreatectomy. (a) Gross specimen showing a soft, tan-coloured lesion with necrosis (arrow) located in the body of the pancreas, which measured 1.2 cm × 1.6 cm × 1.6 cm. (b) Haematoxylin and eosin stained sample of the metastatic placental site trophoblastic tumour (PSST). Immunochemical phenotype of the metastatic PSST tumour: (c) human placental lactogen-positive; (d) cytokeratin-positive; (e) CD10-positive; (f) Ki-67-positive (proliferation index of 22%); (g) focal human chorionic gonadotropin-positive; (h) P63-negative; (i) CD34-negative; (j) vimentin-negative. Scale bar 100 µm. The colour version of this figure is available at: http://imr.sagepub.com
Discussion
Choriocarcinoma is a highly malignant type of trophoblastic neoplasia that is sensitive to chemotherapy, with almost all low-risk lesions and 80–90% of high-risk lesions eventually being cured. 7 PSTT is a rare pathological subtype of GTN that does not present with typical symptoms and diagnosis mostly depends on detailed histological examination. 4 According to the medical history and the postoperative results, this current patient was re-evaluated and diagnosed with PSTT. In this current case, after so many cycles of chemotherapy and based on the pathology report after resection of the pancreatic mass, the patient was diagnosed with PSTT.
The organs most commonly affected by metastasis from GTN are, in decreasing order of frequency, the lung (50%), liver (5–20%), lower genital tract (cervix, vagina, vulva), brain, kidney and gastrointestinal tract. 8 Treatment of GTN metastases includes chemotherapy, surgery or a combination of both. To the best of our knowledge, this is the first reported case demonstrating metastasis to the body of the pancreas, following a spontaneous rupture of a liver metastasis that was treated with arterial embolization and chemotherapy.
At the beginning of the most recent examination of this current patient, abdominal CT scans revealed a mass in the body of the pancreas that was highly suspected to be a metastatic lesion from the original primary GTN. Metastatic choriocarcinoma to organs such as the pancreas, lung or brain has been previously reported.7,9 This current patient was a rare case of sequential GTN metastasis to the liver and subsequently to the pancreas, which was shown by histopathological examinations to be a PSTT after a long period of disease quiescence. The mass in the body of the pancreas was successfully resected and an additional five cycles of monthly chemotherapy were administered. Routine follow-up with measurements of the serum β-hCG (<3 IU/l) and abdominal CT scans did not indicate any new relapse beyond 12 months. It is noteworthy that, following the rupture of the liver metastasis, no other metastases were noted. In addition, during the operation, careful inspection of all four abdominal quadrants did not reveal signs of peritoneal metastasis. The relapse of the disease could have been due to the following reasons: (i) chemoresistant tumour cells becoming quiescent tumour cells that did not produce β-hCG; (ii) chemoresistant and quiescent tumour cells producing modified types of β-hCG, such as hyperglycosylated hCG, although this was not specifically detected in this case; 10 (iii) PSTT is commonly resistant to chemotherapy and secretes hCG at significantly lower levels compared with other types of GTN. 11
The serum levels of β-hCG after recurrence were less than 20 IU/l in the current case. The immunohistochemical analysis of the metastatic lesion removed from the pancreas after several rounds of chemotherapy showed positive staining for human placental lactogen and cytokeratin, focally positive staining for hCG and a Ki-67 proliferation index of 22%. The lesion was negative for vimentin and P63. This immunohistochemical phenotype was consistent with a diagnosis of PSTT.4,12
The occurrence of PSTT can follow term pregnancy, premature delivery, hydatidiform mole and choriocarcinoma, with an interval between the occurrence and previous pregnancy ranging from months to several years. 4 PSTT is relatively resistant to chemotherapy compared with other GTNs. 4 The treatment recommended by the National Comprehensive Cancer Network was surgery and chemotherapy with a platinum/etoposide-containing regimen, such as EMA/etoposide +cisplatin (EP) or other regimens such as TP/paclitaxel + etoposide (TE), bleomycin +etoposide + cisplatin (BEP), etoposide +ifosfamide + cisplatin (VIP) or ifosfamide + carboplatin + etoposide (ICE). 13 Therefore, this patient received five cycles of chemotherapy with the TP regimen after surgical resection of the metastatic lesion in the pancreas.
Based on the experience with this current case, this type of sequential recurrence of GTN in either the primary site or metastatic sites, such as the pancreas or liver, can be cured using a comprehensive treatment strategy involving surgery and/or salvage chemotherapy. Moreover, continuous follow-up (serum β-hCG levels and imaging) over a long period of time (up to 10 years) should be undertaken for patients with a high-risk status.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Anhui Provincial Organ Transplantation Innovation Programme (no. S20183400001), the Fundamental Research Funds for the Central Universities (no. WK9110000055), the National Natural Science Foundation of China (no. 81872110) and the National Key Research and Development Programme of China (no. 2018YFC1003900). The funders played no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript.
