Abstract
Objective
To explore the usefulness of highly sensitive nucleic acid detection for assisting with the accurate antiviral treatment of patients with cirrhosis that was caused by hepatitis.
Methods
There were 377 patients with hepatitis B with cirrhosis and 119 patients with hepatitis C with cirrhosis, either as hospitalized patients and outpatients, who were enrolled into the study. Among them, 299 were men and 197 were women between 23 and 82 years of age. All patients were examined using a domestic HBV DNA/HCV RNA test, which was negative in 162 cirrhosis with hepatitis B and 54 cirrhosis with hepatitis C patients (HBV DNA/HCV RNA <500 IU/mL). Prediction and analysis of the HBV DNA load using alanine aminotransferase (ALT) level was based on receiver operating characteristics (ROC) curve analysis.
Results
For patients with hepatitis C with cirrhosis, after the antiviral therapy, ALT, HCV RNA, and Child–Pugh grade were significantly improved compared with before treatment. ROC analysis results showed that an ALT level of 29 IU/mL was the most sensitive cutoff value to judge a positive HBV DNA load (sensitivity 1.0, specificity 0.237, Youden index 0.763).
Conclusion
Precise detection for patients with cirrhosis caused by hepatitis is required for accurate therapy.
Keywords
Introduction
In cirrhosis that develops from chronic hepatitis B, C, and D, the liver gradually changes its shape and becomes hardened, eventually resulting in cirrhosis. 1 In the end stage, complications such as upper gastrointestinal hemorrhage, hepatic encephalopathy, secondary infection, hypersplenism, ascites, and cancer often occur. 1 Thus, its timely diagnosis is critical to provide the best treatment. 1
Detection of virus in serum from patients with cirrhosis often shows low-level replication, which may be correlated with the significant reduction in residual hepatic tissue and a corresponding reduction in the amount of virus that enters the blood. 1 Currently, the lower limit of quantitative detection for HBV DNA using the domestic test is mostly below 100 to 500 IU/mL, while the lower limit using highly sensitive viral nucleic acid detection is below 15 to 20 IU/mL. However, the sensitivity of both methods is not high enough. 2 Therefore, for patients with cirrhosis who show negative nucleic acid quantitative detection results using a domestic kit and have low-level replication using the highly sensitive nucleic acid detection, timely and effective antiviral treatment is often delayed due to false-negative results. Thus, more sensitive detection methods are required.
The present study aimed to explore the guiding significance of highly sensitive nucleic acid detection in the accurate antiviral treatment of patients with cirrhosis that is caused by hepatitis, and to evaluate the value of transaminase levels in highly sensitive nucleic acid detection.
Methods
Study subjects
From May 2013 to April 2018, cirrhosis patients with hepatitis B and cirrhosis patients with hepatitis C who were hospitalized or outpatients were enrolled into this study. Inclusion criteria for cirrhosis patients with hepatitis B and cirrhosis patients with hepatitis C was a negative result using a domestic HBV DNA/HCV RNA test (HBV DNA/HCV RNA <500 IU/mL) or a result that was than the lower limit of detection (the normal value was <500 IU/mL; Hunan Shengxiang Reagent Co., Ltd., Changsha City, China) in two consecutive tests within 3 months. Thus, 216 patients with cirrhosis caused by hepatitis were enrolled into this prospective self-controlled study. The hepatitis B and C diagnoses and cirrhosis diagnosis were made in accordance with the guidelines for the prevention and treatment of chronic hepatitis B and the guidelines for the prevention and treatment of chronic hepatitis C, which were jointly formulated by the Hepatology Branch and Infectious Diseases Branch of the Chinese Medical Association in 2015. The exclusion criteria were as follows: concurrent hepatitis D infection, HIV infection, fatty liver, alcoholic fatty liver disease, or primary hepatoma.
The present study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee at the First People’s Hospital of Lanzhou on 14 April 2013. All patients provided written informed consent.
Study methods
Routine blood tests, biochemical indicator tests, coagulation tests, serum virus marker detection, and computed tomography (CT) or magnetic resonance imaging (MRI) examinations of the upper abdomen, domestic HBV DNA/HCV RNA tests (normal range lower limit was 500 IU/mL; Hunan Shengxiang Reagent Co., Ltd.) and Cobas HBV DNA and Cobas HCV RNA detection (RT-PCR was performed at the Golden Domain Detection Center, normal range lower limit was 15 to 20 IU/mL, Cobas AmpliPrep/COBAS TaqMan v 2.0, Roche, Basel, Switzerland) were performed for all patients.
Patients who were positive for HBV DNA were treated with 0.5 mg of entecavir once daily for 24 weeks. Patients who were positive for HCV DNA were treated with 400 mg of sofosbuvir in combination with 60 mg of daclatasvir for 24 weeks. Changes in the related indicators between these two groups before and after treatment were compared.
Statistical analysis
The SPSS 19.0 software (Armonk, NY, USA) package was used for data processing. A t-test was used to compare the mean between two groups, and the χ 2 test was used to compare the rate. The relationship was analyzed using receiver operating characteristics (ROC) curves. The ROC curves were created using SPSS 19.0. P < 0.05 was considered to be statistically significant.
Results
There were 377 cirrhosis patients with hepatitis B and 119 cirrhosis patients with hepatitis C in who were hospitalized or outpatients who were enrolled into this study from May 2013 to April 2018. Among these patients, 162 cirrhosis patients with hepatitis B and 54 cirrhosis patients with hepatitis C had negative results using a domestic HBV DNA/HCV RNA test (HBV DNA/HCV RNA <500 IU/mL), and these 216 patients with cirrhosis caused by hepatitis were enrolled. Among these patients, 162 patients had cirrhosis caused by hepatitis B and 54 patients had cirrhosis caused by hepatitis C. Among these patients, 133 were men and 83 were women. Their average age was 54.7 years and their age ranged from 27 to 78 years. Additionally, 145 patients were classified as Child–Pugh grade A for liver function and 71 patients were classified as Child–Pugh grade B.
Therapeutic effects of patients who were HBV DNA/HCV RNA-positive after 24 weeks of antiviral therapy
As illustrated in Table 1 and Figures 1a and 1b, after 24 weeks of treatment with entecavir, the alanine aminotransferase (ALT) level, HBV DNA level, and Child–Pugh grade of liver function for patients with cirrhosis caused by hepatitis B significantly improved compared with before the treatment (P < 0.05). After 24 weeks of treatment with direct-acting antiviral (DAA) therapy, the ALT level, HCV RNA, and Child–Pugh grade significantly improved in patients with cirrhosis caused by hepatitis C (P < 0.05). Additionally, 64.2% of HBV patients who were negative for HBV DNA using a domestic kit had a low level of replication using the Cobas kit.
Comparison of HBV DNA/HCV RNA-positive rate and other data between the two groups.
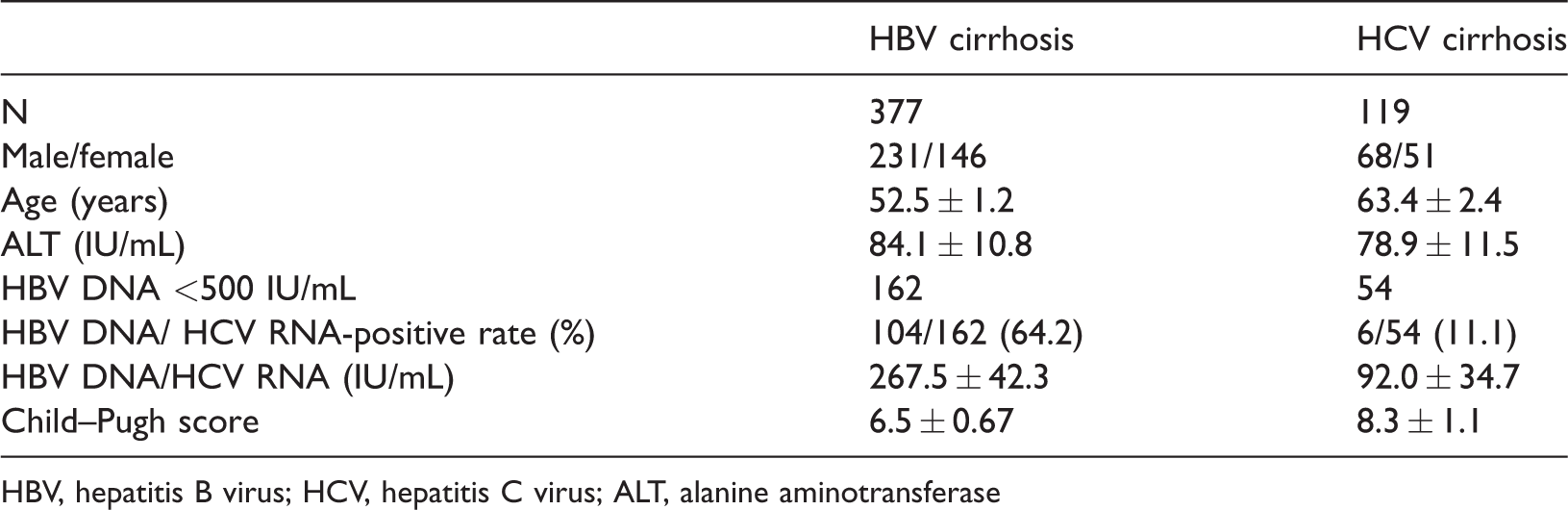
HBV, hepatitis B virus; HCV, hepatitis C virus; ALT, alanine aminotransferase

Study results. (a) The therapeutic effects after 24 weeks of anti-viral therapy. (b) The therapeutic effects in HCV RNA-positive patients with cirrhosis after 24 weeks of anti-viral therapy. (c) The ROC curve of the correlation between 500 IU/mL of HBV DNA and ALT. The cutoff value is HBV DNA 500 IU/mL, ALT optimum decision-making value is 33.5 IU/mL, sen is 0.87, and spec is 0.227 (Youden index, 0.643). (d) The ROC curve for the correlation between 20 IU/mL of HBV DNA and ALT. The cutoff value is HBV DNA 20 IU/mL, ALT optimum decision-making value is 29 IU/mL, sen is 1, and spec is 0.237 (Youden index, 0.763).
In HBV patients, 89 (85.6%) showed no detectable HBV DNA and their liver function improved after 24 weeks of treatment. In HCV patients, all patients showed no detectable HCV RNA and their liver function improved after treatment.
The relationship between HBV DNA and ALT in cirrhosis caused by hepatitis B
In Figure 1c and d, a correlation was shown between ALT and HBV DNA. When the HBV DNA viral load of 500 IU/mL was the cutoff value, the area under the ROC curve (AUC) was 0.830, with a 95% confidence interval of 0.703 to 0.953. When the ALT level was 33.5 IU/L, the sensitivity for predicting a HBV DNA level lower than 500 IU/mL was 0.87, and the specificity was 0.227 (Youden index 0.643). When the HBV DNA viral load of 20 IU/mL was the cutoff value, the AUC was 0.904, with a 95% confidence interval of 0.810 to 0.998. When the ALT level was 29 U/L, the sensitivity for predicting a HBV DNA level lower than 20 IU/mL was 1.0, and the specificity was 0.237 (Youden index 0.763).
Discussion
In the present study, the results showed that the prevalence of low-level replication in patients with cirrhosis caused by hepatitis B was significantly higher than that in patients with cirrhosis caused by hepatitis C. This may be due to the differences in viral nucleic acids. Thus, it is important to remind clinicians to perform Cobas HBV DNA detection for patients with cirrhosis caused by hepatitis B, which provides good guidance upon which to base decisions for starting treatment, regardless of whether the goal is a complete response or to evaluate drug resistance. 3
In the present study, most patients with very low levels of viral replication still had inflammatory activity in the liver. Thus, effective and timely antiviral therapy could be of great value for preventing disease progression, thereby improving the patient’s prognosis. A study that was conducted by Kim et al. 4 suggested that the risk of hepatoma in patients with low-level replication who were treated with entecavir is higher than that in patients with negative viral transformation.
ALT is an important index that reflects hepatic disease. 5 Because highly sensitive nucleic acid detection is expensive, if patients with a high risk of cirrhosis because they are HBV DNA-positive are initially screened for their ALT level and highly sensitive HBV DNA detection is used, individualized, precise detection, and accurate therapy in a hospital can be realized, and the cost of detection can be reduced. Therefore, the correlation between the ALT level and viral replication was explored. The difference was that the subjects in the present study were patients with cirrhosis, and no gender grouping analysis was conducted. Therefore, on the basis of these above results and the relevant literature, the ALT level was suggested to be correlated with the HBV DNA load in patients with cirrhosis. Thus, the ALT level can be used to predict the presence of viral replication in patients with cirrhosis. This guidance and decision-making role are applicable to hospitals and medical institutions that cannot perform highly sensitive HBV DNA detection. If conditions permit, relevant and accurate detection is recommended for patients with cirrhosis. The limitation of this study is its small sample size, and the results of this study need to be verified with a larger sample size in the future.
In summary, precise detection for patients with cirrhosis caused by hepatitis is required for accurate therapy.
