Abstract
Objective
The aim of this prospective randomized controlled clinical trial was to explore the relationship between
Methods
There were 82 low-risk MDS patients who were randomly divided into the following two groups: androl, thalidomide, and PND capsule (ATP group, n = 41); or androl and thalidomide (AT group, n = 41). Hemoglobin and neutrophil and platelet counts and changes in
Results
The plasma hemoglobin level increased in both groups after treatment. However, the platelet count increased only in the ATP group. Patients in the ATP group had a better platelet response than the AT group, and
Conclusion
PND can improve hematological parameters and decrease the
Keywords
Introduction
Myelodysplastic syndrome (MDS) is a heterogeneous clonal myeloid disease that is characterized by ineffective hematopoietic function, which is accompanied by abnormal maturation and developmental abnormalities in one or more blood cell lineages. 1 MDS mainly affects elderly patients. The annual incidence of MDS in the elderly population is 12 to 50 per 100,000 people, which is much higher than 3.5 to 10 per 100,000 people for the general population.2,3 MDS is associated with genetic mutations, immune disorders, chromosomal abnormalities, epigenetic abnormalities, and other factors.4,5 Therapeutic strategies for low-risk MDS include red blood cell (RBC) infusion and iron chelation therapy, the combined use of hematopoietic growth factors and immunosuppressive agents, and androgen-assisted therapy, but the response to these treatments is unsatisfactory.5–7
Numerous studies have shown that glutathione peroxidase is involved in the development and progression of many tumors.8,9 Glutathione peroxidase 3 (GPX3) is the most widely studied member of the GPX family. 10 GPX3 reduces accumulation of hydrogen peroxide in the body during cellular oxidative metabolism.11,12 The primary biochemical role of hydrogen peroxide is to modulate the characteristics of cancer cells, including proliferation, invasion, migration, angiogenesis, and apoptosis. 13 Abnormal inactivation or low GPX3 expression may result in the accumulation of excess reactive oxygen species (ROS), including hydrogen peroxide, leading to tumorigenesis.13,14
Abnormal DNA methylation at the CpG island of the promoter in tumor-associated genes is a common event in human cancer, 14 which has also been shown to be involved in the development and progression of MDS. 14 It is a biomarker for the prognosis, treatment, and survival of patients with MDS.15–19 Recently, hypomethylated drugs, such as azacitidine and decitabine, have been shown to have clinical effects in the treatment of MDS.20,21
The biologically active component in ginseng extract, the panaxadiol saponins component, was isolated from total saponins of ginsenosides using a hematopoiesis biological activity assay, and it was formulated into capsules that are called Pai-Neng-Da (panaxadiol saponins component, PND), which is a class-five new Chinese patent medicine. The composition and content of PND have been analyzed and defined as five monomers of panaxadiol saponins. The PND patent for treating a variety of pancytopenia has been authorized by the State Intellectual Property Office of China. PND was used subsequently for a series of pharmacodynamics, toxicological, and clinical studies. All research data for the 23 items were submitted to the China Food and Drug Administration (CFDA), and two certificates for the new class-five Chinese patent medicine were authorized and granted by CFDA in 2010, including both PND capsule (approval No. 2010L00856) and panaxadiol saponins component (approval No. 2010L00857). This was then successfully transferred to Ningbo Tianzhen Pharmaceutical Co. Ltd. for clinical trials and commercial production. PND was demonstrated to be safe, and six to ten tablets daily was recommended as a safe dose in a phase I clinical trial.
1
A phase II clinical trial was performed in seven hospitals with a professional advantage for treating primary immune thrombocytopenic purpura (ITP) and chronic angiogenic leukocytopenia (neutropenia).
26
The clinical results confirmed that PND was effective and had no adverse side effects.
27
Consistent with previous studies and as supported by this project, this study used PND+andriol+thalidomide or andriol+thalidomide to investigate the efficacy of PND in the treatment of low-risk MDS, and to test the association between
Methods
This study is a subanalysis of a prospective clinical trial that was registered with the China Clinical Trial Bureau (ChiCTR; http://www.chictr.org.cn/; registration number ChiCTR-IOR-15006635).
Inclusion and exclusion criteria
Low-risk MDS patients were diagnosed in accordance with the Vienna diagnostic criteria, including refractory anemia (RA), refractory anemia with multilineage dysplasia (RAEB-1), and refractory cytopenia with multilineage dysplasia (RCMD) type. On the basis of the International Prognostic Scoring System (IPSS) score, patients in the low-risk group had a score of 0 to 1.
The following patients are excluded, including pregnant women or women who planned to become pregnant, and breastfeeding women; patients with severe cardiovascular, hepatic, renal system or mental disease; patients with the complication of severe infection; patients with some types of MDS including refractory anemia with increased blasts, 5q-syndrome, MDS that were not classified, or IPSS score ≥1.5 points; or MDS patients who needed more active treatment such as demethylation chemotherapy, chemotherapy, or bone marrow transplantation.
Drugs
PND capsule was provided by Ningbo Tianzhen Pharmaceutical Co. Ltd. (Ningbo, China; batch No. 20120101). Specifications were as follows: size 4 gelatin capsules and 40 mg per capsule. Each capsule contained five panaxadiol saponins monomers with a purity of 92.44% as analyzed and defined by high-performance liquid chromatography (HPLC) using specific monomers of ginsenosides as the reference standards. Androl was purchased from Catalent France Beinheim S.A. (Beinheim, France), and thalidomide was purchased from Changzhou Pharmaceutical Factory Co., Ltd. (Changzhou, China).
Study design
Low-risk MDS patients from Ningbo First Hospital (from October 2015 to March 2019) were included in this prospective randomized controlled clinical trial. Patients were randomly divided into two groups. The ATP group comprised patients who received oral androl, thalidomide and PND (PND capsule, ginsengdiol group saponin extract; 40 mg, three capsules at a time twice per day). Andriol Testocaps capsule (80 mg) were taken twice daily, and thalidomide tablets 100 mg were taken before going to bed. Patients in the AT group received oral androl and thalidomide, as follows: Andriol Testocaps capsule 80 mg twice daily; and thalidomide tablets 100 mg before going to bed for a 4-week course of treatment, and there were five courses of medication. The neutrophil count, hemoglobin level, platelet count, hepatic function, and renal function of patients were assessed at the beginning of the experiment and at follow-up. Bone marrow samples from the two groups were collected before and after treatment for 3 months and stored at −80°C until methylation detection. This study was approved by the Human Research Ethics Committees of Ningbo First Hospital (Ethical approval number, 2015-R001, Approval Date, 23 January 2015). All patients provided written informed consent for the bone marrow collection and immunophenotyping research.
Efficacy evaluation
Therapeutic efficacy was evaluated in accordance with the efficacy standard of the MDS International Working Group. 28 In this study, the hematologic improvement including platelet response (HI-P), neutrophil response (HI-N), and erythroid response (HI-E) was our focus, especially HI-P. The major HI-P response was defined as follows: for patients with a pretreatment platelet count that was less than 100,000/mm3, it was defined as an absolute increase of 30,000/mm3 or more; and for platelet transfusion-dependent patients, it was defined as stabilization of the platelet count and platelet transfusion independence. The minor HI-P response was defined as follows: for patients with a pretreatment platelet count less than 100,000/mm3, it was defined as a 50% or more increase in platelet count with a net increase greater than 10,000/mm3 but less than 30,000/mm3. The improvements must last at least 2 months in the absence of ongoing cytotoxic therapy.
DNA methylation detection for GPX3
The Omega DNA Mini Kit (Omega Bio-Tek, Norcross, GA, USA) was used to extract genomic DNA from bone marrow and blood. The bisulfite conversion process of the DNA template was as described in our previous work.
29
The quantitative methylation-specific polymerase chain reaction (PCR) (qMSP) technology, which was based on fluorescent dye (SYBR-Green), was used to quantitatively detect the
Statistical analysis
SPSS version 17.0 software (SPSS Inc., Chicago IL, USA) was used for statistical analysis. Measurement data were presented as the mean ± standard deviation (x ± s). Paired-sample
Results
There were 101 MDS patients included in the main study, and 19 were excluded here because of incomplete data. Thus, 82 MDS patients were included in this subanalysis. There were 26 men and 56 women who were enrolled into this study. Among the enrolled patients, 41 were enrolled into the ATP group and 41 were enrolled into the AT group. The average age of patients in the ATP and AT groups was 53.66 ± 10.86 years and 52.63 ± 13.46 years, respectively. The clinical pathology and biochemical data from patients in the ATP and AT groups were matched for parameters such as age, gender, neutrophils, hemoglobin, and platelets (Table 1).
Basic clinical characteristics of patients.

N.S., not significant.
ATP promotes hematological improvement in patients with MDS
After 3 months of treatment, 62.64% (21 of 33) of patients showed a hematologic improvement (platelet response, HI-P) in patients receiving ATP compared with 36.67% (11 of 30) of patients in the AT group. As shown in Figure 1, platelet and hemoglobin increased significantly after 3 months of ATP treatment compared with before treatment (n = 41; platelets: 73.24 ± 54.54 × 109/L vs. 98.76 ± 75.46 × 109/L,

Routine blood test results for MDS patients changed significantly after 3 months and 5 months of treatment compared with before treatment in the ATP and AT groups. A. Change in the platelet level before and after treatment. B and C. Change in hemoglobin level before and after treatment in the two groups.
GPX3 demethylation induced by PND in patients with MDS was related to gender and could promote the recovery of PLT hematopoietic function
As shown in Figure 2, we selected three CpG sites from the CpG island in the

Drawing of the amplified fragment (chr5: 150399986-150400083) at GPX3 CpG island that was obtained from the UCSC genome browser (GRCh37/hg19). The qMSP primers were underlined, and three CpG sites were in red. The targeted region was located at the CpG island of GPX3.
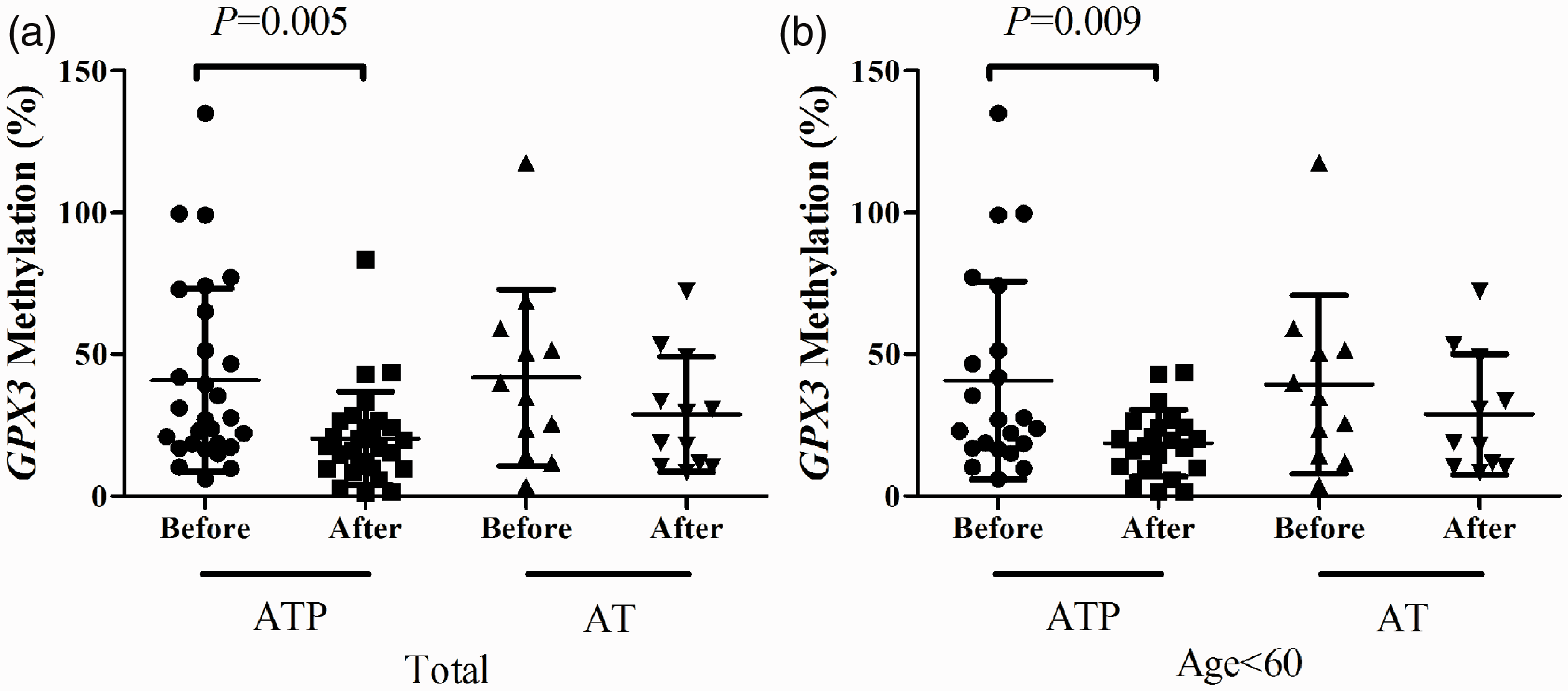
Comparison of the difference in GPX3 methylation between the ATP and AT groups before and after treatment. a. After 3 months of treatment, GPX3 methylation was significantly reduced in the ATP group compared with before treatment, but there was no difference in the AT group; b. GPX3 methylation in patients who were younger than 60 years in the ATP and AT groups was investigated. GPX3 methylation was significantly lower in the ATP group after compared with before treatment. There was no difference in the AT group after 3 months of treatment compared with before treatment.
In addition, the
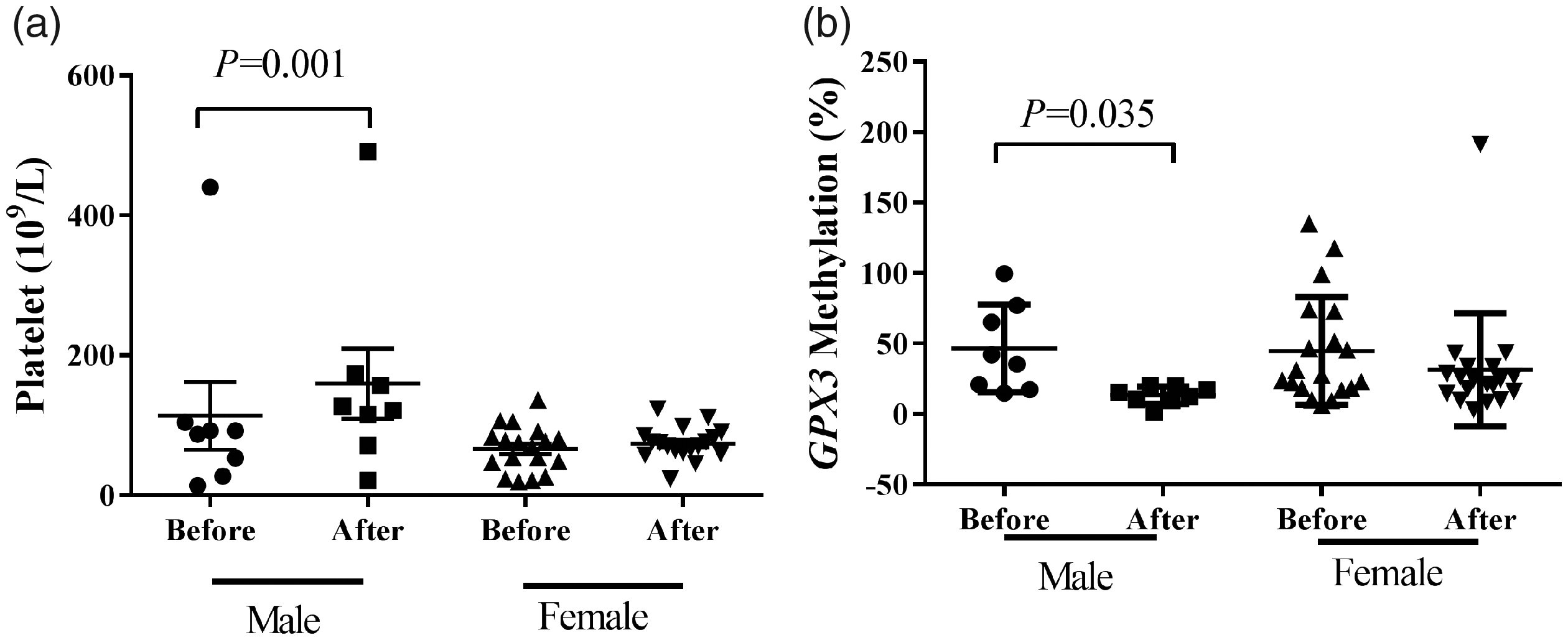
In patients with low-risk MDS, GPX3 demethylation by ATP may be related to gender, and platelet elevation was more pronounced in male patients with stronger GPX3 demethylation. a. GPX3 demethylation was more pronounced in male MDS patients compared with female MDS patients after 3 months of ATP treatment. b. The platelet count increased significantly in low-risk male compared with low-risk female MDS patients with more significant GPX3 demethylation.
The demethylation function of PND on GPX3 might be related to the overall HI-P of patients with low-risk MDS
As described above, in the ATP group,

Effect of the change in GPX3 methylation on the hematological response to low-risk MDS patients with ATP treatment. A. Compared with the Non HI-P group, platelet counts of patients in the HI-P group increased significantly after treatment. B. GPX3 methylation decreased significantly in patients with a hematological improvement (HI-P), and those without hematological improvement (Non HI-P) had the negative results after treatment compared with before treatment.

Correlation between the changes in GPX3 methylation and PLT counts before and after ATP treatment.
Discussion
A series of preclinical studies of PND31–33 were preformed including pharmacodynamic studies in animal models with hemocytopenia, cell biology, molecular biology experiments, and toxicological studies to test the biologically active panaxadiol saponins component. These studies demonstrated that PND possessed dual activity, i.e., promoting proliferation and differentiation of hematopoietic progenitor cells and regulating the immune function. PND reduced peripheral blood CD8+ cells, and increased CD4+ cells, returning the CD4+-to-CD8+ cell ratio to normal. It reduced the myelosuppression of bone marrow that was caused by chemotherapy, accelerated bone marrow recovery, and increased the peripheral WBC and platelet count, as shown by mice treated with hemocytopenia that was caused by cyclophosphamide chemotherapy. PND was effective in promoting proliferation for the three lineages of hematopoietic progenitor cells, increasing erythroid, myeloid, and megakaryocytic progenitor cell colony formation in bone marrow culture. A pharmacodynamic study in CD34+ hematopoietic stem/progenitor cells suggested that PND was effective in promoting hematopoiesis and inducing differentiation, which suggests that its activity is similar to that of hematopoietic growth factors.
34
PND promotes hematopoiesis using an intracellular signal pathway, and it induced the up-regulation of multiple transcription factors such as GATA-1, GATA-2, c-Fos, c-Jun, and NF-E2, and their protein expression level, phosphorylation status, and DNA binding activities were increased dramatically by PND treatment in hematopoietic cells, thereby promoting hematopoiesis and blood cell formation.26,35–37 However, in this study, PND improved hematopoietic function in low-risk MDS patients by promoting
In this study, after 3 months of treatment with ATP, the platelet count was significantly elevated in patients with low-risk MDS, and patients achieving HI-P. However, the
In our study, the hemoglobin level was significantly increased in both groups. After 3 months of treatment, 62.64% of ATP patients and 36.67% of AT patients achieved HI-P, indicating that PND as an auxiliary drug provided a better clinical outcome in low-risk MDS. Thrombocytopenia is common in patients with MDS and the estimated prevalence is 40% to 65%. 38 The average platelet component mean platelet volume is a useful screening marker for MDS. 39 MDS patients with thrombocytopenia have a poor prognosis. 40 Bleeding complications are a major cause of morbidity and mortality, and thrombocytopenia is also an independent factor in reducing survival. This has been included in the updated prognostic scoring system. 41 Platelet transfusion is a necessary support for clinically significant thrombocytopenia.42,43 MDS patients with severe thrombocytopenia may benefit from the recovery of platelet activity and further allo-hematopoietic stem cell transplantation (HSCT) after decitabine treatment. 44 In this study, the platelet count was significantly increased after 3 months of treatment in the ATP group compared with before treatment, indicating that PND capsule has a positive effect on enhancing hematopoietic function and improving symptoms in low-risk MDS patients.
Promoter DNA hypermethylation and down-regulation of the
In this study, we enrolled 82 patients who had low-risk MDS, which did not reach the expected number of cases, and this may affect the statistical significance of the results. However, in conclusion, the study shows, to a certain extent, that PND capsule is able to increase the platelet count to obtain HI-P in low-risk MDS patients, which showed better results than the group without PND. Decreasing the
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was supported by the traditional Chinese medicine administration of Zhejiang Province (Grant No. 2015ZZ018), the National Science Foundation of Zhejiang Province (Grant No. LY17H160005), the Zhejiang Medical and Health Science and Technology Project (Grant No. 2019KY158), the National Science Foundation of Ningbo (Grant No. 2018A610390), and grants from K. C. Wong Magna Fund at Ningbo University.
