Abstract
Objective
Patients with non-small-cell lung cancer (NSCLC) and primary or acquired resistance do not respond to targeted drugs. We explored whether cancer cells can be cultured from liquid biopsies from patients with primary resistance to tyrosine kinase inhibitors (TKIs). We aimed to predict patients’ responses to drugs according to in vitro drug testing results.
Methods
Cancer cell cultures were established from the pleural effusion of a patient with TKI-resistant NSCLC using a conditional reprogramming technique. Phenotypic drug sensitivity tests were performed using the Cell Counting Kit-8 assay. We tested individual drugs and compared the synergistic and inhibitory effects of drug combinations.
Results
The results of our in vitro sensitivity test using the combination of cisplatin and pemetrexed were correlated with the patient’s response.
Conclusion
This represents the first successful report of predictive testing for combination therapy in patients with epidermal growth factor receptor-mutant NSCLC and primary TKI resistance. This strategy should be applicable to both chemotherapies and targeted therapies, and it will significantly improve the clinical treatment and management of patients with NSCLC and primary or acquired resistance to targeted therapies, as well as patients lacking targetable mutations.
Keywords
Introduction
In total, 70% to 80% of patients with non-small-cell lung cancer (NSCLC) carrying epidermal growth factor receptor (EGFR) mutations respond to first-generation tyrosine kinase inhibitors (TKIs), whereas a small percentage of patients with mutant EGFR have primary resistance to TKIs. 1 Patients with EGFR-mutant NSCLC initially respond to first-line treatment, but most patients repeatedly experience relapse because of acquired drug resistance. Although the mechanisms of acquired resistance to EGFR inhibitors in EGFR-mutant NSCLC have been identified, little is known about the evolution of resistant clones during drug therapy. 2
The standard therapies for advanced NSCLC include chemotherapy, continuous EGFR-TKI therapy, and combination therapies. Patients with advanced NSCLC who are treated with platinum-based doublet chemotherapy have response rates of less than 30%. 3 An overall response rate of approximately 70% has been reported for patients who received targeted therapies against EGFR mutations or anaplastic lymphoma kinase gene fusion, 4 compared with less than 30% for patients treated with regular chemotherapy. 3 Currently, less than 10% of patients benefit from genomics-based precision oncology, and functional or biological analyses using patients’ cells are urgently needed for precision oncology.5–7
Conditional reprogramming (CR) is a new technique for rapidly expanding normal and tumor cells from surgical specimens or needle biopsies.8,9 CR technology has displayed promise in basic and clinical cancer research, 10 especially for the rapid screening of candidate drugs and personalized medicine.11–13 The genomic and histological features of the original tumor are maintained in cultured CR cells.14–16 CR cell culture makes it possible to directly assess drug sensitivity and conduct molecular analysis in individual patients with cancer. It has been reported that CR cells were useful for identifying effective treatments for respiratory papillomatosis, 17 adenoid cystic carcinoma, 11 and pancreatic and prostate cancers. 13 For example, the results of drug testing using CR cells from patients with lung cancer were consistent with the clinical findings. 18
In this study, we established cancer cell cultures from the pleural effusion of a patient with NSCLC and primary resistance to TKIs using CR technology. Then, we performed drug testing using these cancer cells and determined the optimal combination regimens for the patient. Interestingly, the in vitro drug response was correlated with the patient’s response to clinical treatment. To our knowledge, this is the first described use of CR technology to culture cancer cells from pleural effusion samples and assessment of drug efficacy in a patient with EGFR-mutant NSCLC.
Materials and methods
Cancer cell culture from pleural effusion
Pleural effusion samples were collected from a patient with NSCLC. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Peking University Shenzhen Hospital. Written informed consent was obtained from the patient. To culture cancer cells, 10 mL of pleural effusion were mixed with a specific Sample Medium (YongTech, Shenzhen, China). Red blood cell lysis buffer (Beyotime Biotechnology, Shanghai, China) was then added to eliminate red blood cells from the pleural effusion. Cancer cells in the pleural effusion from the patient were cultured under a previously described co-culture CR condition.9,19 Cell cultures were differentially trypsinized with 0.05% trypsin-EDTA for further passage.
Drug sensitivity testing
CR cancer cells derived from pleural effusion were plated in 96-well plates at a density of approximately 1 × 104 cells per well. Twenty-four hours later, the cells were treated with monotherapy (cisplatin, gemcitabine, pemetrexed, or docetaxel) or combinations of these drugs (cisplatin plus gemcitabine, cisplatin plus pemetrexed, or cisplatin plus docetaxel) at the indicated concentrations for 48 hours. The viability of CR cancer cells from pleural effusion was measured using the Cell Counting Kit-8 assay. To compare the synergistic and inhibitory effects of the aforementioned drugs, a concentration versus response curve was plotted according to the data using Calcusyn version 2.0 (Biosoft, Cambridge, UK).
Results
Clinical history of the patient with NSCLC
Figure 1 presents the clinical history of the patient. In October 2016, a 62-year-old woman was admitted to the Department of Respiratory Medicine of Peking University Shenzhen Hospital because of a constant cough that had persisted for approximately 2 months. After examination via computed tomography (CT) and pathologic and molecular biological analyses, she was diagnosed with pulmonary adenocarcinoma with systemic multiple lymphatic tumor extension. An EGFR mutation (L858R in exon 21) was detected in both lung biopsy and blood samples. EGFR-TKI therapy (icotinib, 125 mg, p.o. three times/day) was prescribed. However, the patient’s symptoms worsened after 2 weeks. She coughed constantly and exhibited shortness of breath. She displayed bilateral lower extremity edema. These features indicated a failure of EGFR-TKI therapy. In November 2016, CT revealed atelectasis, obstructive pneumonia, multiple pulmonary nodules, and malignant pleural effusion with identifiable primary lesions in both lungs of the patient. Brain CT did not identify an intracranial lump and swelling, indicating that her cancer had not metastasized to the brain. Thereafter, cisplatin plus gemcitabine chemotherapy, a standard first-line treatment for patients with progressive NSCLC without the EGFR-T790M mutation, was prescribed to the patient. During the first therapy cycle, the patient displayed chemotherapy-induced myelosuppression (leukopenia). Thus, treatment was halted, and subcutaneous recombinant human interferon α2b was prescribed to the patient. Based on the in vitro chemosensitivity test result, cisplatin plus pemetrexed therapy (cisplatin 75 mg/m2 and pemetrexed 800 mg/m2, D1), which was associated with fewer side effects and greater efficacy in clinical trials, was recommended to this patient 2 weeks later. The patient reported no obvious discomfort during cisplatin plus pemetrexed therapy, and a second cycle of this regimen was administered during her next admission. According to the American Joint Committee on Cancer, the overall response to cisplatin plus pemetrexed therapy was classified as “stable disease.”

Clinical history and disease status of the patient. The patient was initially hospitalized on October 18, 2016. Based on the in vitro sensitivity findings, the combination of cisplatin plus pemetrexed was recommended to the patient 10 weeks after her confirmed diagnosis. According to the American Joint Committee on Cancer, the overall response was classified as “stable disease” during this entire chemotherapy cycle (between December 22, 2016 and January 16, 2017).
Generation of cancer cells from pleural effusion
Cancer cells were cultured from pleural effusion using the CR technique as presented in Figure 2a. The morphology of cancer cells was captured 24 hours after plating (Figure 2b upper panel), and continued proliferation was noted at passage 4 (15 days, Figure 2b lower panel). Cancer cells cultured from pleural effusion exhibited the typical features of epithelial cells, including a polygon shape and clear arrangement.

Generation of cancer cells from pleural effusion. (a) Concise summary of the experimental protocol, including the patient’s tumor cells derived from pleural effusion and potentially individualized tumor cells that were conserved for other experimental purposes if necessary. (b) The morphological features of tumor cells from the patient’s pleural effusion after 24 hours of culture and continued proliferation for four passages (15 days). The circles in the upper panel indicate the colonies of cancer cells isolated from the patient’s pleural effusion. The tumor cells exhibit typical morphological features of epithelial cells including a polygon shape and clear arrangement between cells.
Identification of a combinative chemotherapy for the patient
Currently, there is no available genome-based method for predicting patient response to drug combinations. CR technology can be used to establish cultures of primary cancer cells and propagate the cells rapidly. This new technique has been reported to predict patient response to chemotherapy or targeted therapy for precision oncology.11–13,20,21 We selected chemotherapeutic regimens according to an Eastern Cooperative Oncology Group performance status of 0 to 1. 9 Our in vitro sensitivity test illustrated that the inhibitory effects of combinations were concentration-dependent (Figure 3a). Cisplatin plus pemetrexed had inferior effects on tumor cells from pleural effusion than cisplatin plus gemcitabine (95% confidence interval [CI] = −0.05 to 0.37; P = 0.0538). However, cisplatin plus pemetrexed had superior growth-inhibitory effects than cisplatin plus docetaxel (95% CI = 0.01 to 0.44; P = 0.0003). Based on the drug combination indices, cisplatin had the strongest synergistic effects with pemetrexed (Figure 3b, 3c, 3d).

Sensitivity of cancer cells from pleural effusion to anti-tumor drugs. (a) Inhibitory effects of cisplatin, cisplatin plus gemcitabine, cisplatin plus pemetrexed, and cisplatin plus docetaxel at the indicated concentrations on tumor cells isolated from the patient’s pleural effusion. The combination index plot of cisplatin was generated in compliance with gemcitabine (b), pemetrexed (c), and docetaxel (d).
Meanwhile, combinations featuring carboplatin and other drugs had weaker inhibitory effects on the growth of tumor cells from pleural effusion than combinations featuring cisplatin (data not shown). Four days after the first cycle of cisplatin plus pemetrexed, routine inspection of this patient’s blood revealed no obvious hematotoxicity (Table 1). Cisplatin plus pemetrexed chemotherapy was less myelosuppressive than cisplatin plus gemcitabine according to the patient’s white blood cell and platelet counts. Our in vitro sensitivity test using the combination of cisplatin plus pemetrexed matched the patient’s response.
Comparison of safety data after treatment with different chemotherapy regimens.

Discussion
EGFR mutations are present in a large subset of patients with NSCLC, 22 especially East Asian women with adenocarcinoma. This patient with advanced EGFR-mutated NSCLC was diagnosed via molecular biological analysis of biopsy and blood samples. Icotinib (an oral EGFR-TKI) was administered to the patient because it is the first-line treatment for patients with EGFR mutations in China. 23 However, the patient’s clinical symptoms worsened rapidly approximately 2 weeks after icotinib therapy. Thereafter, the assessment of the EGFR mutation status was repeated using ARMS-PCR. EGFR exons 18, 19, 20, and 21 were identified as wild-type using the patient’s pleural effusion samples. It has been reported that EGFR mutations from different sources of specimens are diverse, including bronchoscopy biopsy, surgery, lung puncture, lymph node biopsy, bone marrow biopsy, and pleural effusion. 24 Concerning the heterogeneity in NSCLC, EGFR mutation testing results have been inconsistent in the same patients because of tumor heterogeneity and sampling. 25 The findings of no EGFR mutations may be related to the aforementioned factors because of the different sources of testing materials. Thus, we cannot deny the possibility of false-negative results in ARMS-PCR. Liu found that TKIs may have efficacy in patients who are negative for EGFR mutations. 26 EGFR mutation-positive samples according to direct DNA sequencing were identified as mutation-negative using ARMS-PCR. The inconsistency may be explained by the low abundance of EGFR mutations. 27 Regardless of the method used, the detected results must be explained by the patient’s clinical response. Targeted therapies for cancers with specific genetic mutations have displayed promising efficacy in patients. However, primary and acquired resistance can arise, and the mechanisms of resistance vary among patients. Crystal et al. 28 cultured cancer cells obtained from CT-guided needle biopsies of patients who developed resistance to targeted therapies using CR technology. Then, they performed genetic analysis and drug screening using the cultured cells. They were able to identify monotherapies and drug combinations with efficacy against resistant lung cancers. This team also used the cultured cancer cells to study the mechanisms of primary resistance to targeted therapies.29,30
According to the patient’s clinical symptoms and income level, a first-line targeted therapy (TKI) was prescribed. After icotinib resistance and deteriorated progression were observed, a platinum-based regimen was recommended. From a pharmaceutical viewpoint, it is rational that combination regimens have stronger inhibitory effects than monotherapies. 31 Currently, the first-line chemotherapy regimen for advanced NSCLC in some studies included cisplatin or carboplatin combined with other drugs such as docetaxel, pemetrexed, or gemcitabine. 32
Cisplatin plus gemcitabine chemotherapy had severe side effects in this patient. Myelosuppression was detected in a regular blood test, necessitating urgent treatment withdrawal. Thus, the physicians seriously considered the toxicity and side effects of chemotherapies that cannot be tolerated by patients with advanced NSCLC who have poor physical conditions. Obviously, during the treatment of this advanced case of NSCLC, the standard therapeutic guidance was not appropriate in terms of safety. Considering the tumor heterogeneity and differences in chemosensitivity among patients, the efficacy of chemotherapies for advanced NSCLC should vary among patients. 15 Therefore, it is of great importance to establish an optimal treatment model that can predict chemosensitivity in vitro and identify “tailored” individualized treatments for specific patients. This in vitro individualized tumor model both mimics the treatment, which can be used to confirm the known clinical information before the application of therapy, and prevents drug resistance. 33
In this study, we designed an in vitro pulmonary adenocarcinoma treatment model by reprogramming pleural effusion tumor cells to imitate the clinical conditions. The reprogrammed cells maintained continuous proliferation, tumorigenicity, and heterogeneity in vitro, thereby matching the physical and clinical situation with predictive utility for clinical treatment. The EGFR mutation status of cancer cells developed using CR technology matched that of the patient’s lung biopsy. Establishing a long-term culture of pleural effusion tumor cells will permit the screening of large numbers of potential drugs for clinical reference. The experimental results obtained using CR technology were objective and highly reproducible. This, this strategy represents an important step toward more tailored treatments for patients with advanced NSCLC, and it may be used as a quantitative model for analyzing the mechanism of action and testing new targeted or compound drugs in the future (Figure 4). The screening of pleural effusion tumor cells from our patient is advantageous for identifying the appropriate combination therapies and reducing side effects. In terms of individual differences and drug safety, the combination of cisplatin and pemetrexed was the most promising therapeutic regimen, and it was previously selected for patients with advanced NSCLC who did not undergo surgery. Our study found that cisplatin and pemetrexed had stronger inhibitory and synergistic effects.
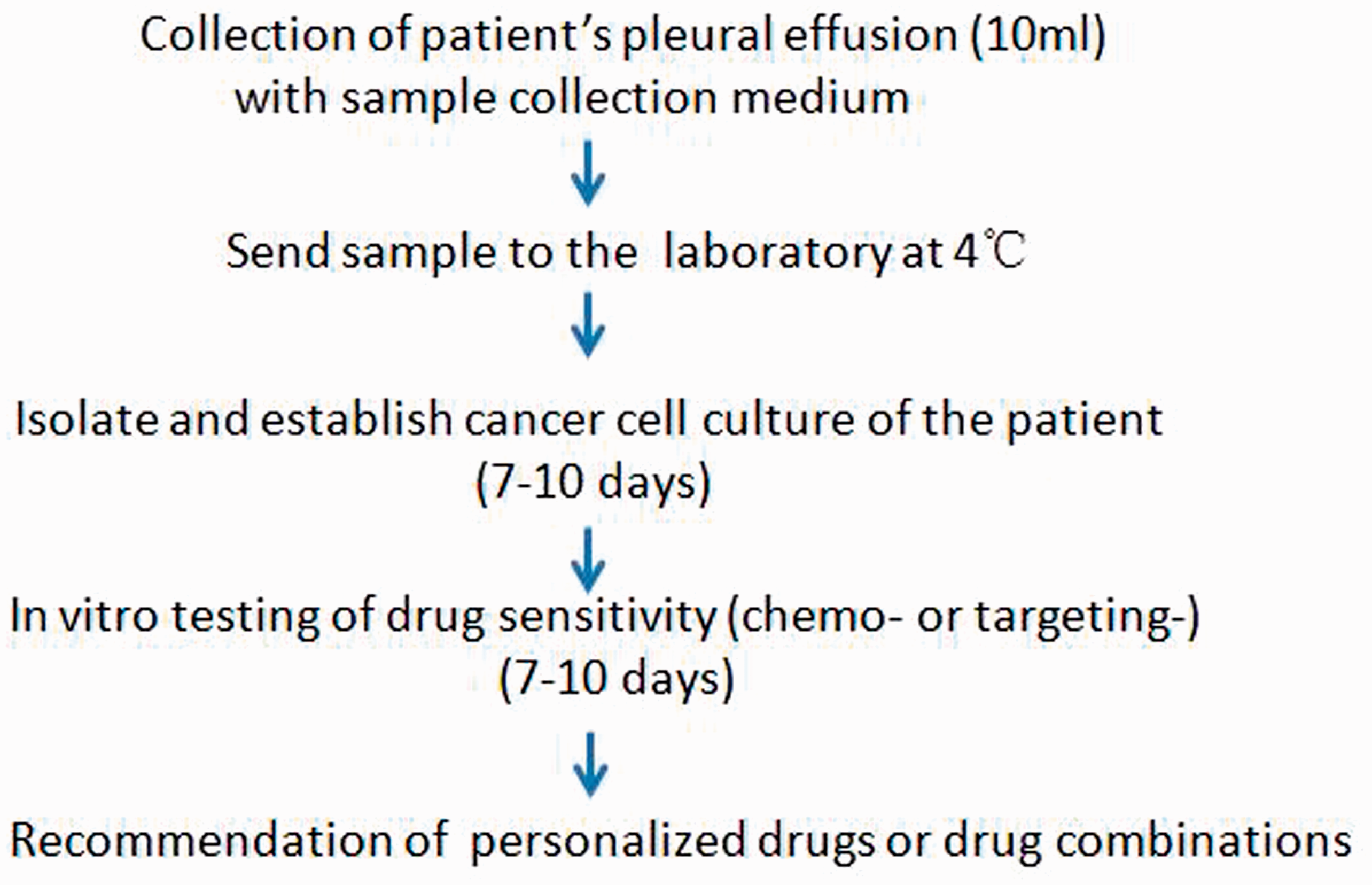
Protocol for drug testing for non-small-cell lung cancer cells derived from the pleural effusion of a patient.
In this study, we developed a pre-clinical model that can propagate cells that are derived directly from human tumors or healthy tissues in vitro. With this model, in vitro drug sensitivity testing can be performed using a personalized cell model. However, it is necessary to further extend the number of cases and experiments in vitro to improve credibility. Additionally, the present study did not reveal resistance-causing genes that are functionally associated with relevant proteins and drug susceptibility.
