Abstract
Objective
The pathogenesis of chronic obstructive pulmonary disease (COPD) remains elusive. Here, we assessed the correlation between CD8+ T cell frequencies and autophagy in COPD patients.
Methods
Subjects were divided into three groups (n = 30 patients/group): (1) COPD patients in the stable phase; (2) smokers with normal lung function; and (3) non-smokers with normal lung function. Flow cytometry was used to enumerate CD8+ T cell subsets (CD8+, CD8+ effector, and CD8+ memory T cells) and quantitate T-cell apoptosis. RT-PCR and western blotting were used to measure levels of LC3 and p62.
Results
Frequencies of CD8+ T cell subsets and expression of p62 and LC3 II/I were significantly higher in COPD patients compared with the other two groups, while the rate of apoptosis was lower. In COPD patients, LC3 II/I and p62 expression were positively correlated with CD8+ T cell subset frequencies. Moreover, a significant correlation was observed between LC3 II/I and p62 expression and T cell subset frequencies.
Conclusion
Autophagy level is positively correlated with the frequencies of CD8+ T cells, suggesting that autophagy might be involved in COPD pathogenesis.
Introduction
Chronic obstructive pulmonary disease (COPD), characterized by recurrent airway inflammation and progressive exacerbation of airflow, is a common chronic airway inflammatory disease. COPD can result in severe disability and is the third leading cause of death globally. In China, the overall prevalence of COPD among adults 40 years or older is high (13.6%). Long-term treatment of COPD is costly and the condition is irreversible, placing a huge economic burden on families and public health systems. 1 Clinically, airway inflammation and destructive emphysema are two major pathological features of COPD. 2 However, the pathogenesis of COPD is still poorly understood, and there are no definitive and effective cures. It is therefore critically important to better understand the pathogenic mechanisms involved in COPD. Recent studies have shown that smoking is a significant cause of COPD because airway inflammation in lung tissue caused by smoking persist even after smoking cessation.3,4 Recently, several studies suggested that destruction of the alveolar wall in smokers is mainly related to T-cell function.5–6 CD8+ T cells are the key subpopulation of lymphocytes among infiltrating cells responsible for chronic airway inflammation in COPD.7–12 These cells infiltrate the airways of patients with stable COPD. However, the underlying mechanisms of CD8+ T cell infiltration and function in COPD are poorly understood.
It was reported that tobacco extract induces multiple autophagy mechanisms13,14 and leads to emphysema.15–18 Previous studies found that levels of specific autophagy biomarkers were increased in the lung tissues of COPD patients.15,19 Autophagy promotes the survival of CD8+ effector T cells and the formation of CD8+ memory T cells.20,21 These studies indicated that there might be an association between autophagy and CD8+ T cells in COPD patients. However, the mechanism through which CD8+ T cell frequencies increase and the role of autophagy remain unclear. In this study, we assessed the expression of the autophagy related genes, LC3 and p62, in the peripheral blood of patients with stable COPD. We also assessed LC3 II/I and p62 protein levels, frequencies of CD8+ T cell subsets (CD8+ effector and CD8+ memory cells), and the rate of CD8+ T cell apoptosis to investigate the relationship between autophagy and CD8+ T cell frequency. Together, these data provide insight into potential pathogenic mechanisms underlying COPD.
Materials and methods
Patients and sample collection
This study included patients with stable COPD at the Respiratory Outpatient Center of Shenzhen Longgang Central Hospital from January 2017 to August 2017. The study was approved by the ethics committee of Shenzhen Longgang Central Hospital, and was performed according to the principles laid out in the Helsinki Declaration.
All cases were diagnosed according to the standard diagnostic guidelines for chronic obstructive pulmonary disease (2013 edition, Chinese version). All patients were investigated in the stable state without acute exacerbation within the preceding 2 weeks. None of the participants had chronic hunger, malnutrition, acute infection, chronic viral hepatitis, herpes simplex virus infection, autoimmune disease, coronary artery sclerosing heart disease, diabetes, other organ failure, pulmonary encephalopathy, psychosis, acute hypoxia or mental stress. Patients were of both genders and were 50 to 70 years old. Participants were randomly selected Han nationals. Their participation was voluntary and all participants provided written informed consent.
Age, sex, and ethnicity-matched healthy controls were recruited during the same period. These controls were stratified according to their smoking history. The healthy controls were divided into smokers with normal lung function (SN group) and non-smokers with normal lung function (NSN group). A smoker was defined as an individual who currently smoked more than one cigarette per day without attempts at cessation for at least 1 year before enrollment.
Pulmonary function test
Pulmonary function tests were carried out for all subjects. Subjects inhaled a bronchodiastolic agent (salbutamol 400 µg), followed by lung function measurement. The percentage of forced expiratory volume occupied by vital capacity in the first second and FEVl1 in the first second were recorded. The FEV1 ratio of the first second forced expiratory volume to the predicted value was assessed. Pulmonary function was measured using a Medisoft HypAir type pulmonary function instrument (Leeds, UK).
Autophagy and CD8+T cell assessments
A FACSAria III flow cytometer (BD, San Jose, CA, USA) was used for identification and sorting of CD8+ T cells. Real-time quantitative reverse transcription–polymerase chain reaction (qRT-PCR) was performed to assess expression levels of the autophagy associated genes LC3 and p62 using the MAX™ System (BD). Western blotting was used to assess protein expression levels of LC3Iǁ/LC3I and p62. Flow cytometry was used to assess the distribution of CD8+ T cell subsets, including the ratio of CD8+ memory to effector T cells, and to quantitate apoptosis in CD8+ T cells.
Statistical methods
Data were plotted and analyzed using GraphPad Prism (GraphPad Corp., San Diego, CA, USA). Data were presented as means ± standard deviations. All statistical analyses were performed using SPSS 21 software (SPSS, Chicago, IL, USA). Pearson’s method or Spearman’s nonparametric method were used to assess the correlation between autophagy level (expression levels of LC3II/I and P62) and frequencies of CD8+ T cells. The Student’s t test was used to assess differences between two groups. Values of P < 0.05 were considered statistically significant.
Results
CD8+ T cells are increased in COPD patients
Patients sample were collected and divided into three groups: the COPD group, the SN group and the NSN (n = 30 participants per group). The characteristics of participants in each group are summarized in Table 1. Flow cytometry showed that total CD8+ T cells were significantly more numerous in the peripheral blood of COPD patients compared with the SN and NSN groups (32.33 ± 4.23% vs 27.92 ± 4.18% and 25.82 ± 4.62%, respectively) (Figure 1a). CD8+ effector and CD8+ memory T cells in COPD patients were also significantly more numerous in the peripheral blood of COPD patients compared with the SN and NSN groups (Figure 1b). CD8+ T cells, CD8+ effector and CD8+ memory T cells were also significantly more numerous in the SN group compared with the NSN group (Figure 1a, b). In contrast, the average rate of CD8+ T cell apoptosis of COPD patients (23.51 ±6.11%) was lower than the rates in the SN and NSN groups (SN :27.39 ±7.21%; NSN: 31.61 ±4.98%) (P < 0.05) (Figure 1c). These results indicated that impaired lung function was responsible for accumulation of CD8+ T cells in the lung.
Gender and age distribution of participants in the COPD, SN and NSN groups.
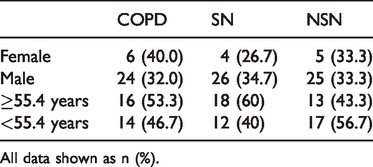
All data shown as n (%).

CD8+ T cells are increased in COPD patients. (a) Total CD8+ T cells were more numerous in COPD patients compared with the SN and NSN groups. (b) Memory T cells and effector T cells were more numerous in CPOD patients compared with the SN and NSN groups. (c) The T-cell apoptosis rate was decreased in COPD patients compared with the SN and NSN groups.
Expression of the autophagy related genes LC3 and p62 is increased in CD8+ T cells of COPD patients
To address the relationship between autophagy and CD8+ T cells, we evaluated the expression of the autophagy related genes LC3 and p62 in CD8+ T cells using qRT-PCR. There was no difference in LC3 expression between COPD patients and the other groups, whereas expression of p62 in COPD patients was significantly higher compared with the other two groups (Figure 2) At the protein level, expression of LC3 in COPD patients also showed no difference compared with the other two groups, while p62 expression was higher in COPD patients compared with the other two groups (Figure 3a). The LC3II/LC3I ratio was significantly increased in COPD patients compared with the other two groups (Figure 3b). Interestingly, the LC3II/LC3I ratio and p62 expression was higher in the SN group compared with the NSN group (Figure 3a&b). These observations indicated a potential association between autophagy and COPD, with smoking acting as a driver of autophagy.

Transcript abundance of autophagy related genes. Expression of LC3 was not significantly different in COPD patients while p62 expression was significantly higher in COPD patients compared with the SN and NSN groups.

Protein expression of autophagy related genes. (a) LC3 expression was unchanged in COPD patients, but the LC3II/LC3I ratio was significantly increased. Consistent with analysis of mRNA expression, p62 expression was also higher in COPD patients. (b) Statistical analysis of LC3 and p62 expression.
Autophagy gene expression is positively correlated with CD8+ T cell frequency in COPD patients
To further address the association between autophagy and CD8+ T cell frequency, we performed a correlational analysis. The LC3II/LC3I ratio was positively correlated with frequencies of total CD8+ T cells (r = 0.572, P < 0.05), CD8+ effector T cells (r = 0.672, P < 0.05) and CD8 memory T cells (r = 0.739, P < 0.05) (Table 2). Moreover, positive correlations between p62 expression and frequencies of total CD8+ T cells (r = 0.613, P < 0.05), CD8+ effector T cells (r = 0.681, P < 0.05) and CD8 memory T cells (r = 0.745, P < 0.05) were observed (Table 2). These results indicated that there is a positive correlation between the LC3II/LC3I ratio, p62 expression and the number of CD8+ T cells.
Pearson correlation analysis of CD8+ T cell frequencies, LC3 II/I ratio and p62 expression.
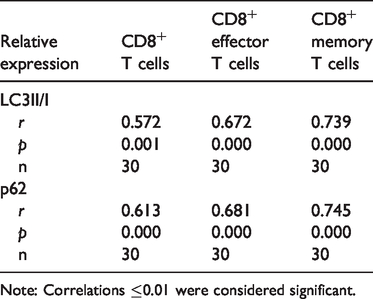
Note: Correlations ≤0.01 were considered significant.
Discussion
The pathogenic mechanisms underlying COPD remain elusive. Viral infections 13 and increased production of autoantibodies22,23 may contribute to COPD. However, no specific causative virus or immune mechanism has been identified to date. In this study, we found that LC3 gene expression was not significantly different between COPD patients and healthy controls. However, the LC3II/LC3I ratio and p62 expression in COPD patients were significantly higher than in healthy controls, suggesting that increased autophagy was occurring in the COPD group. Pearson correlation analysis showed that autophagy level and frequencies of CD8+ T cells in COPD patients were positively correlated.
LC3 is an autophagy biomarker gene in mammals and is homologous to autophagy-related protein 8 in yeast. LC3 has two isoforms, LC3I and LC3II. The cellular LC3II/I ratio reflects the degree of autophagy. 24 In this study, LC3II/I ratios in COPD patients were significantly increased, indicating that autophagy was occurring in COPD patients even when LC3 gene expression was normal, consistent with a previous study. 25 The p62 protein, also known as sequestosome 1, is an ubiquitin-binding protein encoded by SQSTM1 and acts as an autophagic substrate during autophagy. 26 P62 can guide LC3 to the autophagosomal membrane to form autolysosomes. Typically, the production and degradation of p62 is in equilibrium at normal levels of autophagy. Studies revealed that p62 expression is increased when autophagy is elevated.27–29 P62 also activates the caspase8 apoptotic pathway, 30 which may explain why CD8+ T cells had reduced apoptosis rates in COPD patients in our study.
COPD is mainly caused by inflammation. In our study, we found that frequencies of CD8+ T cells were increased in COPD patients, which may lead to increased inflammation in the lung. Moreover, we found that increased autophagy was correlated with increased frequencies of CD8+ T cells, suggesting that autophagy is the main cause of pulmonary inflammation. Interestingly, we also found autophagy and frequencies of CD8+ T cells were significantly higher in the SN group compared with the NSN group, indicating that smoking alone can drive the autophagy to increase pulmonary inflammation. One limitation of our study is that we did pursue further analyses because of limitations in our lab.
In conclusion, these results demonstrated that frequencies of CD8+ T cells in patients with stable COPD were increased, and were associated with increased autophagy levels.
Footnotes
Acknowledgements
The authors gratefully acknowledge financial support from the Science Foundation of ShenZhen (China).
Authors' contributions
HZ and SMZ participated in data collection and drafted the manuscript. WGZ, YS and XQX performed the statistical analysis and participated in study design. NL and SDC helped to draft the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
The datasets used and analyzed in this study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the basic research project of the Shenzhen Science and Technology program (JCYJ20160427100015608).
