Abstract
Objectives
This study aimed to investigate the epidemiology of intravenous midazolam-induced postoperative expressive aphasia (EA).
Methods
The incidence rate, risk ratio, and contributing factors to intravenous midazolam-induced postoperative EA were analyzed retrospectively in 6756 orthopedic patients. A telephone interview was conducted with patients with EA after surgery.
Results
Patients were allocated to either the midazolam group (n = 6178) or no-midazolam group (n = 578). Twelve patients developed EA in the midazolam group, with an incidence of 0.19%, and no patient developed EA in the no-midazolam group. The mean age of EA patients was 70 years, and 92% were women. Among them, 75% received general anesthesia, and the mean dose of midazolam was 1.8 mg. EA was reversed in nine of 12 (75%) patients within 4 minutes of flumazenil administration, and >60 minutes were required to reverse EA in the other three patients (25%).
Conclusion
Intravenous midazolam administration for preoperative sedation caused transient EA in 0.19% of patients, especially elderly women who received general anesthesia, and EA could be reversed by flumazenil.
Introduction
Sedation using midazolam is known to unmask focal neurological defects in brain tumor patients. 1 However, in rare cases, midazolam could induce expressive aphasia (EA), which is a reversible neurologic deficit.
EA caused by midazolam has been reported in only one case report 2 and on the internet in articles describing whether the drug―under its trade name Versed―causes aphonia. 3 The aim of this study was to investigate the incidence of EA in orthopedic patients who were administered midazolam for preoperative sedation and associations with other exposures of interest. We investigated whether EA was associated with midazolam treatment and whether these side effects were associated with age, sex, anesthesia type, American Society of Anesthesiologists (ASA) class, and/or anesthesia duration. The null hypothesis was that there is no association between midazolam medication and the occurrence of EA episodes.
Methods
After obtaining Institutional Review Board approval (DC16RISI0092) from Daejeon St. Mary’s Hospital (Daejeon, South Korea), the medical records of patients who underwent elective orthopedic surgery between March 2011 and May 2016 were reviewed. Given the retrospective nature of the study and the use of anonymized data, the requirement for informed consent was waived.
Patients
There were 6884 patients who underwent anesthesia induced by a physician anesthesiologist who were screened for entry into this study. Regardless of intravenous midazolam premedication, all patients who bypassed the recovery room and were unable to communicate in the recovery room were excluded. The final number of subjects enrolled into this study was 6756. These patients were divided into two groups: (1) exposure, midazolam treatment (n = 6178); and (2) non-exposure, no-midazolam treatment (n = 578). Moreover, patients and controls were divided based on the occurrence of EA.
Study procedures
Patients were classified as EA (+) when they had no neurological deficits and were unable to speak in the recovery room despite their intention to speak. If the recovery room nurse informed the anesthesiologist of a suspected EA patient, a brief neurological examination (including level of consciousness, motor, sensory, and reflexes) was performed by the physician anesthesiologist, and 0.2 mg of flumazenil (Bukwang Flunil®-Bukwang Pharm, Ansan, Korea) was administered to reverse the EA. If there was no immediate response to the drug, flumazenil was administered repeatedly, up to a total dose of 0.5 mg. In the absence of a response to flumazenil treatment, the patient was transferred to the ward and followed up by the physician. All EA (+) patients were referred to the neurologist to rule out possible organic neurological deficits in the brain, and they were asked to undergo brain magnetic resonance imaging (MRI) in the postoperative period.
Additionally, several days after discharge from the hospital, a telephone interview was conducted for patients who had EA to clarify the symptoms that they experienced during the episode. The telephone interview comprised several questions, including the following:
History of the four major symptoms that constitute conversion disorder: inability to speak, motor paresis of the limb, instant blindness, and numbness. History of psychiatric disorders, aphasia, and drug abuse. Where were you when you recovered consciousness in the perioperative period (operating room, recovery room, or ward)? Do you remember not being able to speak despite your intention to speak? If yes, were you panicked by the idea that you might not be able to speak again? Do you have a migraine now? If yes, did you experience migraine symptoms at the time of the EA episode?
Anesthesia administration
For preoperative sedation, midazolam (0.5 to 2 mg; Bukwang Midazolam®, Bukwang Pharm) was administered intravenously to adults in the holding room, whereas children ≤14 years of age received 0.5 to 2 mg/kg of propofol. In the no-midazolam group, there were cases of patient refusal, no administration based on the surgeon’s request, documented allergy to midazolam, and history of delirium after sedation.
After entering the operating room, 50 to 100 µg of fentanyl were injected to facilitate induction of general or regional anesthesia in adults. General anesthesia was induced using propofol (1 to 2.5 mg/kg) with rocuronium (0.8 mg/kg) in adult patients. In pediatric patients, propofol (2 to 3 mg/kg) with rocuronium (0.5 to 1 mg/kg) was used. After endotracheal intubation, the end-tidal concentration of inhalational anesthetics was adjusted to 60% to 70% of the minimal alveolar concentration (desflurane, 4% to 4.5%; sevoflurane, 1.4% to 1.6%). 4 Desflurane and sevoflurane were the first-choice inhalation anesthetics for adult and pediatric patients, respectively. Oxygen (40%) and nitrous oxide (60%) were routinely used with inhalation anesthetics for general anesthesia. To maintain intraoperative analgesia, fentanyl (50 µg) was administered up to a maximum dose of 300 to 700 µg in adult patients during surgery. The amount of fentanyl was based on the type of surgery, preoperative general condition of the patient, patient age, changes in blood pressure, and personal sensitivity to fentanyl. Fentanyl (2 to 3 µg/kg) was injected intraoperatively in pediatric patients based on the same protocol.
For general anesthesia combined with regional block, the procedure was limited to shoulder surgery or total knee arthroplasty that was performed under general anesthesia. A parascalene or femoral block was applied using 0.75% ropivacaine (15 mL), which was performed under the guidance of a nerve stimulator immediately before the induction of general anesthesia. Pyridostigmine (10 mg) and glycopyrrolate (0.4 mg) were routinely administered to reverse muscle relaxation in the patient. In cases of delayed recovery of consciousness because of intraoperative fentanyl administration, naloxone was administered.
The following three types of regional anesthesia were induced in this study: epidural, spinal, and brachial plexus block (BPB). Epidural anesthesia was performed using 0.75% ropivacaine (15 to 20 mL), spinal anesthesia with heavy bupivacaine (13 to 15 mg), and BPB using 0.375% ropivacaine (70 mL) under the guidance of a nerve stimulator. After inducing regional anesthesia, a propofol infusion (100 to 200 mg/hour) was initiated to maintain intraoperative sedation with additional doses of midazolam (2 to 3 mg) if needed.
Another method is mask sedation combined with peripheral nerve block. The orthopedic surgeon blocked the peripheral nerves using 1% lidocaine, and the anesthesiologist performed mask sedation using sevoflurane or desflurane.
After surgery, all patients were transferred to the recovery room. If a patient exhibited EA, the nurse reported this to the anesthesiologist, as described above (all anesthetic procedures were led by one physician anesthesiologist who specialized in orthopedic anesthesia with the assistance of two anesthesia residents). EA data were collected using a “problem case record” from the first EA episode.
The dose of midazolam was categorized into five classes based on the clinical dose used (≤1 mg, 1 to 2 mg, 2 to 3 mg, 3 to 4 mg, or ≥4 mg). The ASA class was divided into four classes (1, 2, 3, or 4), and the type of anesthesia was classified into two groups, including general and regional anesthesia, with additional classification of general anesthesia into general alone and general with block.
Statistical analysis
The incidence of EA and the risk ratio were measured in the two groups. The incidence of EA was compared in terms of patient age, sex, type of anesthesia, dose of midazolam used, anesthesia duration, and ASA class between the EA (+) and EA (−) subgroups in the midazolam-treatment group. The risk ratio was calculated for the EA (+) and EA (−) subgroups with and without midazolam. Age and anesthesia duration were treated as continuous data and sex as categorical data. Means for continuous variables were compared using the unpaired Student’s t-test. The Chi-square test or Chi-square trend test was used to compare categorical variables. P < 0.05 was considered to be statistically significant. All statistical analyses were performed using SPSS version 15 (SPSS Inc., Chicago, IL, USA).
Results
Among the 6884 patients who underwent surgery, 6756 patients who met the study entry criteria were enrolled into the study. Patients were excluded if they bypassed the recovery room and were directly transferred to the intensive care unit (n = 65), if they were generally unable to communicate (e.g., cerebrovascular incident, dementia, delirium; n = 58), if they underwent minor surgery under sedation with peripheral nerve block (n = 28), and in cases of intraoperative death (n = 5; Figure 1). Among the 6756 included patients, 6178 received midazolam as a sedative and 578 did not receive midazolam. Among the 6178 patients who received midazolam, 12 exhibited EA, corresponding to an EA incidence of approximately 0.19% (Figure 1). There were no EA patients in the no-midazolam group. The risk ratio for EA was found to be infinite (Table 1). However, the P value for the association of midazolam with EA occurrence was not significant, and therefore, the null hypothesis could not be rejected.

Flow chart for the study, including the number of patients who were excluded, exposed to midazolam, or who developed expressive aphasia.
Result of expressive aphasia based on midazolam treatment.

Prevalence = 0.19%, Risk ratio = infinite; P=N.S.
EA, expressive aphasia; Mida, midazolam; N.S., not significant.
Overall, the average age was 57 years (men/women, 51/62 years) and 57% were women. The distribution of age and midazolam treatment in the cohort is illustrated in Figure 2. The types of anesthesia the patients received are summarized in Table 2. The types of surgery included lower extremity (32%), shoulder (21%), arthroplasty (19%), upper extremity (19%), spine (5%), pelvis (1%), and other (3%).
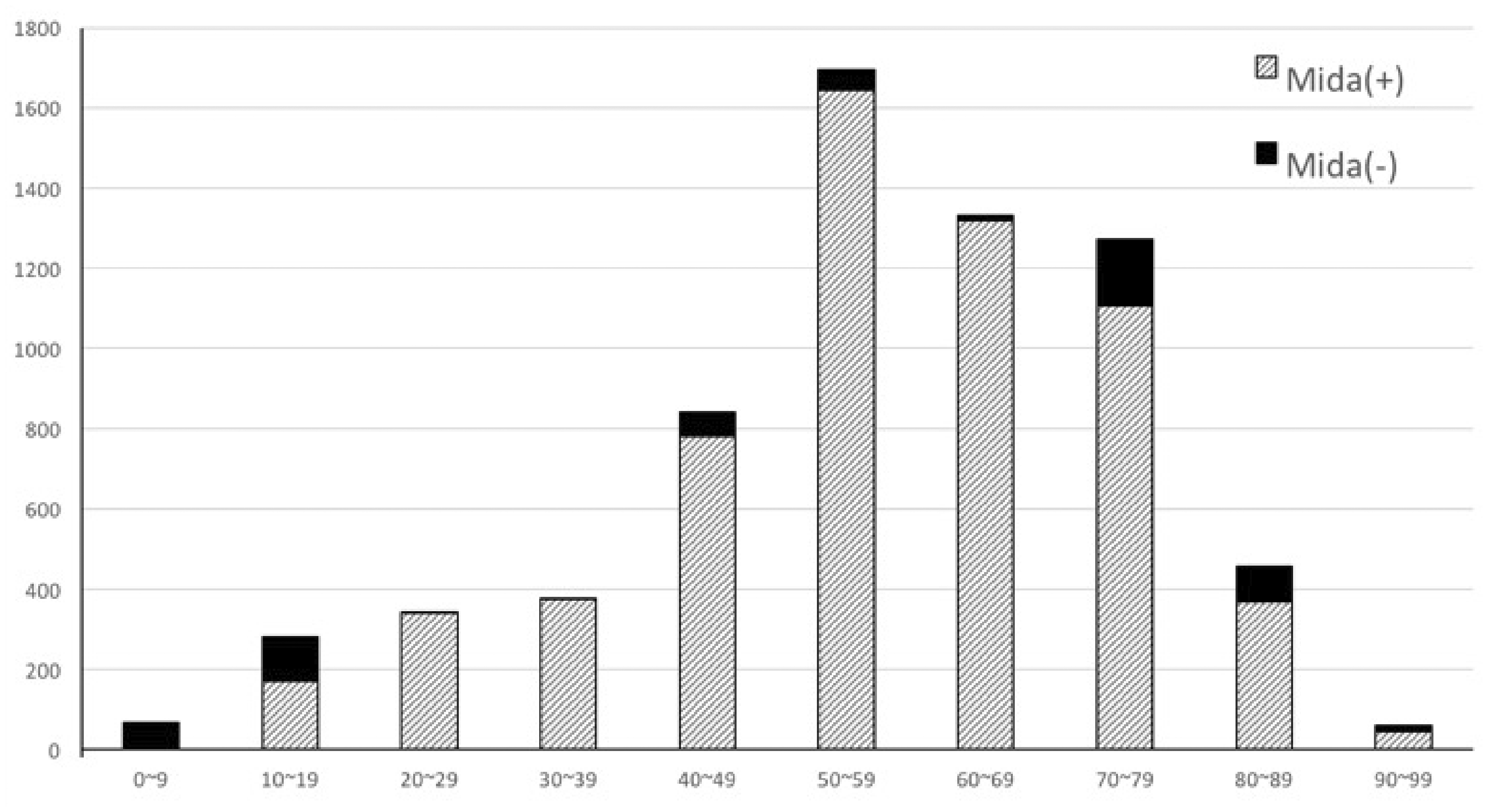
Age distribution of the patients with midazolam treatment.
Type of anesthesia.
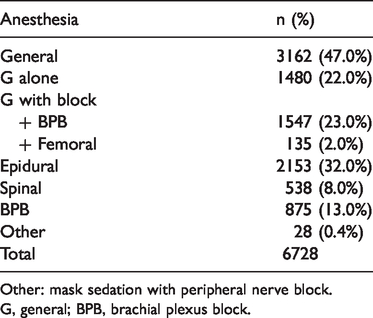
Other: mask sedation with peripheral nerve block.
G, general; BPB, brachial plexus block.
Table 3 lists factors that contribute to the development of EA. The mean age in the EA and non-EA groups was 70 and 58 years, respectively. There were 11 women and 1 man in the EA group. There were more patients who underwent general anesthesia (n = 9) than regional anesthesia (n = 3) in the EA group than in the non-EA group. Age, sex, and type of anesthesia had a significant influence on the occurrence of EA. However, the amount of midazolam used, duration of anesthesia, and patient ASA class did not appear to affect the development of EA. The mean dose of midazolam in the EA group was 1.8 mg compared with 2.3 mg in the non-EA group. The mean duration of anesthesia and ASA score were 153 minutes compared with 137 minutes and 1.9 minutes compared with 1.8 minutes in the EA and non-EA groups, respectively. In a brief neurological examination of the EA patients, those who underwent general anesthesia were able to lift their head for >5 s and their muscle strength was 5/5 for all extremities. The patients’ sensory and deep tendon reflexes were normal, and their pupils were reactive to light.
Comparison between the expressive aphasia and non-expressive aphasia groups.
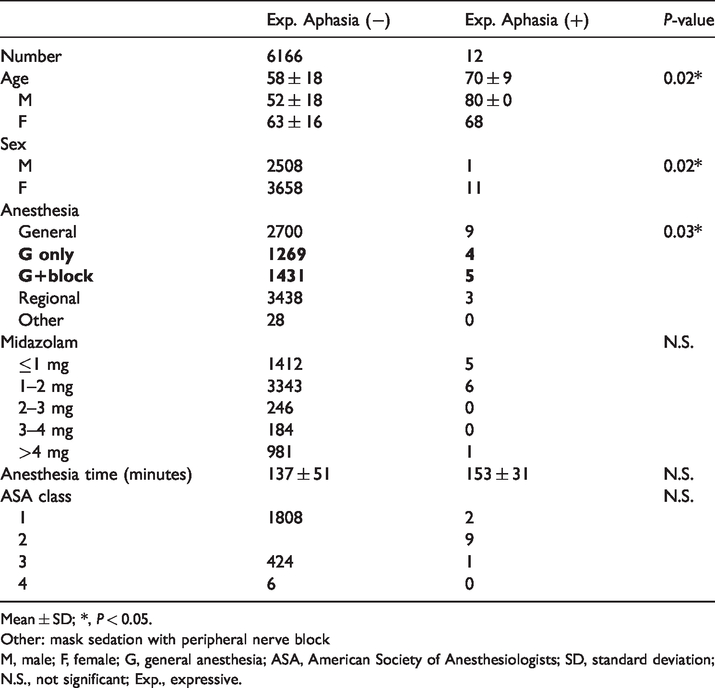
Mean ± SD; *, P < 0.05.
Other: mask sedation with peripheral nerve block
M, male; F, female; G, general anesthesia; ASA, American Society of Anesthesiologists; SD, standard deviation; N.S., not significant; Exp., expressive.
Table 4 presents detailed information on the EA patients. The mean flumazenil dose that was used to reverse EA symptoms was 0.3 mg. Nine of 12 (75%) patients with EA experienced relief of their EA within 4 minutes of flumazenil administration; for three (25%) patients, >60 minutes were required for the EA symptoms to resolve. Six of these 12 patients (50%; mean age, 75 years) could not recall being unable to speak at the time of the EA episode. With the exception of one patient who was unable to communicate because of a perioperative cerebral vascular incident, the remaining five patients (mean age, 61 years) recalled the EA episode. Four of the five patients remembered being panicked by the idea that they would never be able to speak again. Three of the 12 patients had difficulty opening their eyes during the EA episode. No patient had a history of conversion disorder, aphasia, migraine headache, or psychiatric illness, except for a 56-year-old woman who was using antidepressants. All EA (+) patients showed negative results on neurological examination by the neurologist, and the brain MRI study results were also negative.
Perioperative medical records for the 12 expressive aphasia patients.

*, Time for complete recovery from expressive aphasia.
**, No communication by perioperative cerebrovascular attack sequela.
ASA, American Society of Anesthesiologists; OP time, operation time; Fx, fracture; RCT, rotator cuff tearing; A/S, arthroscopic surgery; MM tear, medial meniscus tear; ORIF, open reduction and internal fixation; G, general anesthesia; BPB, brachial plexus block; Epi, epidural anesthesia; HTN, hypertension; Dexa, dexamethasone; HNP, herniated nucleus pulposus, pethidine for shivering control.
Discussion
EA induced by midazolam is uncommon and mostly responsive to therapy with flumazenil. It is associated with older women and general anesthesia, but not with the dose of midazolam that was administered.
EA, which is also known as Broca’s aphasia, is characterized by a partial loss of the ability to produce language, although comprehension generally remains intact. It is caused by acquired damage to the anterior regions of the brain, such as Broca’s area. It is one subset of a larger family of disorders that are known as aphasia. 5
Broca’s aphasia patients exhibit EA and moderate impairment in language comprehension because it is very difficult for patients with organic brain disorders such as Broca’s aphasia to understand complex grammar. 6 Additionally, Broca’s aphasia is often accompanied by definite neurological deficits.
When an alert patient complains of an inability to speak using body language in the recovery room, the anesthesiologist usually considers the possibility of an organic brain disease such as Broca’s aphasia. In our patients, we performed a brief neurological examination immediately. However, there were no other neurological deficits except for the inability to speak. Based on these unusual findings, we considered the possibility of temporary brain motor dysfunction or transient EA that was caused by intraoperative medication(s). We compiled a list of drugs from the anesthesia record that have specific antagonists, and we administered these antagonists to the EA patients. Naloxone was the first candidate drug, but it failed to reverse EA. Flumazenil relieved EA symptoms in some patients; this led us to suspect that the EA was caused by midazolam.
The sedative, anxiolytic, anticonvulsant, muscle relaxant, and amnestic effects of midazolam are produced by acting on the gamma-aminobutyric acid type A (GABAA) receptor. 7 GABA is the major inhibitory amino acid transmitter in the mammalian central nervous system, and it is present in approximately 40% of all neurons. 8 Numerous GABAA receptors are found in the neurons of Broca’s speech region, over Brodmann areas 44 and 45. 9 Considering that EA is closely related to the Broca’s speech areas, it is reasonable to consider that EA could be caused by the GABA receptor effects. Thus, midazolam could be a causative agent for EA given that the EA was reversed by flumazenil, which is a centrally acting midazolam antagonist. 10
Flumazenil, which is a competitive antagonist at the benzodiazepine binding site of the GABAA receptor, has been used to successfully treat postoperative emergence delirium associated with midazolam exposure. 11 , 12
Propofol, sevoflurane, and desflurane are also GABAergic agents, but there is no report of EA as a drug side effect.13,14
Drobish et al. 2 reported the case of emergent delirium with transient associative agnosia and EA that was reversed by flumazenil in a pediatric patient. A 12-year-old girl who received preoperative midazolam underwent general anesthetic, and then exhibited postoperative delirium, consisting of a transient associative agnosia and EA. Administration of flumazenil led to an immediate resolution of her symptoms. Postanesthetic emergence delirium with transient associative agnosia and EA may occur with the atypical offset of midazolam, and it is amenable to flumazenil.
EA occurred more frequently in elderly female patients. From a pharmacokinetic perspective, women have a relatively larger volume of distribution, lower rate of clearance, and longer elimination time for benzodiazepines than men. 15 Additionally, there is a pharmacodynamic interaction between midazolam and female hormones. There is some evidence that supports a periodic alteration of specific GABAA receptor subunits by changes in estrogen levels in mice. 16 Additionally, there have been reports on the pharmacodynamic changes in the binding of benzodiazepines to the GABAA receptor during the ovarian cycle.17–19 Based on our EA data, the mean age of the 11 EA female patients was 68 years, which closely approximates the overall mean age of female patients, which was >50 years (67.5 years), in our study cohort.
The results of this study showed that EA was more common in patients who underwent general anesthesia than in those who underwent regional anesthesia. Considering that midazolam and inhalational anesthetics act on the GABA receptor simultaneously, 20 the synergistic effects of the two drugs may have contributed to the development of EA in general anesthesia patients.
There was no correlation between the amount of midazolam that was administered and the incidence of EA in this study. Moreover, EA did not occur in patients who received 2 to 3 mg or 3 to 4 mg of midazolam, presumably because the total number of patients in both groups was too low compared with the total number of patients who received other doses.
Administration of intraoperative fentanyl may have no effect on the incidence of EA. Although a mean fentanyl dose of 288 µg was used in the general anesthesia alone group, compared with 87 µg of fentanyl in the combined regional–general anesthesia group, there were no differences in the incidence of EA. There was no correlation between ASA class and the incidence of EA.
There were some limitations to this study. First, 9 of the 12 EA patients showed complete recovery with flumazenil within 4 minutes, which strongly supports that their EA symptoms were midazolam side effects. However, we cannot exclude the possibility of a transient ischemic attack (TIA) in the three patients who did not respond to flumazenil. Second, the composition of the patient cohort was skewed toward elderly women, and the prevalence of EA was zero in the midazolam (−) group. Therefore, it was not possible to calculate the odds ratios for EA and proceed with further analysis using a logistic regression model. Third, the size of the midazolam (−) group was small compared with the size of the midazolam (+) group. This resulted in a non-significant association between midazolam and EA. Fourth, there were three patients whose EA was not reversed by 0.5 mg of flumazenil. Generally, midazolam side effects are reversed with an initial dose of flumazenil (0.2 mg) to a maximum total of 3.0 mg. 21 If the three EA patients had been treated with a full dose of flumazenil (3.0 mg) instead of 0.5 mg, the outcome may have been different. Fifth, all EA patients should have undergone a thorough neurological examination by a neurologist before flumazenil treatment by a physician anesthesiologist to rule out other possible diagnoses, including TIA. Thus, the incidence of EA was 0.19%, and it occurred only in the midazolam group. EA symptoms were immediately reversed using flumazenil in 75% of patients. The incidence of EA was affected by age, sex, and general anesthesia.
In conclusion, administration of midazolam may result in the occurrence of transient EA, primarily in elderly women who undergo general anesthesia, which may be reversed using flumazenil. Rapid diagnosis and proper treatment can minimize the duration of this terrifying state. Further work is needed to determine which patients are predisposed to this phenomenon so that they can be identified and managed appropriately.
