Abstract
Objective
To assess the role of NOD-like receptor C5 (NLRC5; a major NLRC family protein that regulates immunity, inflammation and tissue fibrosis), in cerebral ischemia-reperfusion injury, characterized by inflammation and oxidative damage.
Methods
Blood NLRC5 levels were assessed in neonates with cerebral ischemia and in healthy controls. A stable PC12 cell line was established that overexpressed or knocked down NLRC5. Inflammatory responses, apoptosis rate and oxidative damage in PC12 cells under oxygen-glucose deprivation/reperfusion (OGD/R) conditions were evaluated using enzyme-linked immunosorbent assay (ELISA), terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) and reactive oxygen species (ROS) assay.
Results
Blood NLRC5 levels were suppressed in neonates with cerebral ischemia. ELISAs showed that NLRC5 suppressed levels of tumour necrosis factor-α, interleukin (IL)-6, IL-1β, ROS and superoxide dismutase in OGD/R-treated PC12 cells. Furthermore, NLRC5 overexpression was associated with reduced apoptosis rate in PC12 cells treated by OGD/R. Overexpression of NLRC5 also inhibited levels of toll-like receptor (TLR)4, myeloid differentiation primary response protein MyD88 (MyD88) and phosphorylated nuclear factor kappa B-transcription factor p65 (NF-κB p-p65) in PC12 cells, and decreased nuclear levels of NF-κB p-p65.
Conclusion
NLRC5 alleviated inflammatory responses, oxidative damage and apoptosis in PC12 cells under OGD/R conditions by suppressing activation of the TLR4/MyD88/NF-κB pathway.
Introduction
The interruption of cerebral blood flow, including supply of glucose and oxygen, can lead to many diseases, such as ischemic stroke that causes damage to the nervous system. 1 Reconstruction of the internal circulatory system may restore blood supply to the brain, however, further damage may be caused to brain tissue during the recovery of blood supply, 2 in a process known as cerebral ischemia-reperfusion injury. Neuronal cells have a high energy requirement and are sensitive to changes in energy, thus, insufficient blood supply leads to neuronal cell damage or death. 3 Furthermore, reperfusion following ischemia produces a large amount of reactive oxygen species, eventually resulting in oxidative damage. 4 The incidence rates of various diseases resulting from ischemia-reperfusion injury are rising, but there remains a shortage of effective therapies. Therefore, it is necessary to investigate the molecular mechanisms in cerebral ischemia-reperfusion injury, in order to provide new targets and strategies for further treatment.
Proteins of the NOD-like receptor (NLR) family play critical roles in innate immune responses, transcriptional activation, and autophagy. NLRC5 is an NLR family protein with three domains, including the n-terminal caspase activation and recruitment domain (CARD) region, the centrally positioned NACHT domain, and the leucine-rich repeat region. NLRC5 has been shown to regulate the inflammatory response and cell death.5–7 Previous research has also revealed that NLRC5 has an inhibitory impact on the development of inflammatory reactions, through suppression of inflammatory pathways, and in fibrosis, 8 and NLRC5 has been shown to protect liver cells from apoptosis. 9 One study indicated upregulated expression of inflammatory factors in impaired myocardial cells of NLRC5-knockout mice. 8 Furthermore, greater damage could be induced in the liver of NLRC5-knockout mice after ischemia-reperfusion, the occurrence of which is mainly due to activation of related inflammatory pathways and factors, and elevated levels of reactive oxygen species (ROS). 10 All these studies have shown that NLRC5 inhibits levels of inflammation factors, fibrosis and apoptosis in normal cells.
The toll-like receptor (TLR)2/myeloid differentiation primary response protein MyD88 (MyD88)/ nuclear factor kappa B (NF‑κb) signalling pathway has been shown to play a vital role in regulating inflammatory responses and oxidative stress, and in mediating cell injury. 11 In terms of molecular mechanisms involved in ischemia-reperfusion injury, previous research has revealed that TLR4/MyD88/NF-κB signals are abnormally enhanced in renal ischemia/reperfusion, the inhibition of which ameliorates renal injury. 12
Cerebral ischemia-reperfusion injury is a type of neurological injury disease characterized by inflammatory injury, oxidative damage and apoptosis. The objective of the present study was to explore the role of NLRC5 in nerve-cell injury caused by ischemia-reperfusion. Levels of NLRC5 were compared between healthy neonates and neonates with cerebral ischemia-reperfusion injury, followed by in vitro experiments using PC12 cells. The effects of cerebral ischemia-reperfusion were simulated through culture of PC12 cells under oxygen–glucose deprivation/reperfusion (OGD/R) conditions. PC12 cells were utilized to construct NLRC5 overexpression or silencing in order to evaluate inflammatory reactions, oxidative stress and apoptosis under normal and OGD/R incubation conditions. Finally, the role of NLRC5 in cerebral ischemia-reperfusion injury, and the related mechanism, was described.
Patients and methods
Study population
This study included blood samples that were collected from neonates with cerebral ischemia-reperfusion injury, or healthy neonate controls at the Department of Paediatrics, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China between June 2018 and October 2019. Inclusion criteria for the study were: (1) <24 h from time of hospital admission; (2) birth weight ≥2 500g; (3) gestational age > 37 weeks; and (4) written informed consent from family members of the children. Exclusion criteria were: (1) severe obstructive respiratory disease; and (2) anaemia.
The study was approved by the ethics committee of the First Affiliated Hospital of Bengbu Medical College, and written informed consent for providing blood samples for the study was obtained from each patient’s legal proxy.
Cell culture
The PC12 cells (ATCC; Manassas, VA, USA) were cultured in Dulbecco’s modified Eagle’s medium (DMEM [Gibco, Thermo Fisher Scientific; Waltham, MA, USA]) supplemented with 10% Gibco fetal bovine serum (Thermo Fisher Scientific), in a 37°C humid atmosphere with 5% CO2. To simulate cerebral ischemia-reperfusion in vitro, the PC12 cells were first cultured using a glucose-free balanced salt solution in 95% N2, 5% CO2, and 37°C environment for 2 h. Following these conditions, the cells were cultured with normal serum-containing DMEM for 12 h to mimic the reperfusion process.
Cell transfection
PC12 cells that overexpressed NLRC5 were established using overexpression and negative control lentivirus vectors for NLRC5 (Genechem; Shanghai, China), and all procedures were performed according to the manufacturer’s instructions. Briefly, 24 h before transfection, cells were seeded at 1 × 105 cells/well into 24-well plates. The original medium was replaced with 2 ml fresh medium containing 6 µg/ml polybrene (Beijing Solarbio Science and Technology Co. Ltd., Beijing, China) and virus suspension, and incubated at 37°C for 24 h. The transfection efficiency was then evaluated by Western blot and RT-qPCR. Small interfering (si)-RNA was used to establish PC12 cell lines that were silenced for NLRC5. siRNA targeting NLRC5 and the corresponding negative scrambled si-RNA were provided by GenePharma Corporation (Shanghai, China). siRNA and Lipofectamine™ 2000 (Invitrogen, Thermo Fisher Scientific) were both diluted using DMEM with no serum or penicillin-streptomycin. The diluted siRNA was incubated with Lipofectamine™ 2000 (Invitrogen, America) for 20 min. Cells were seeded into a 24-well plate 24 h before transfection. Then, siRNA was transfected into PC12 cells in accordance with the manufacturer’s instructions. Both Western blot and RT-qPCR were performed to assess the transfection efficiency after 24 h.
RT-PCR
Blood samples (10 ml) from healthy control neonates and from neonates with cerebral ischemia-reperfusion injury were collected into blood RNA storage tubes (BioTeke; Beijing, China), and stored at –20°C prior to use. Total RNA was extracted from blood samples and from PC12 cells by the Trizol method, and was then reverse transcribed into cDNA using a PrimeScript™ RT Reagent Kit (TAKARA Bio Inc. Shiga, Japan). After reverse transcription, expression of the target gene was quantified by real-time PCR using the following NLRC5 primers: forward, 5′-CTGCCCAGGGAATTATGCTA-3′; and reverse, 5′-ATCCTGTGCTGCTCCTCAGT-3′, with a SYBR™ Green PCR Master Mix (Thermo Fisher Scientific), according to the manufacturer’s instructions. GAPDH was amplified as the endogenous control. Each reaction contained 2 µl cDNA sample, 2 µl of each primer and 10 µl master mix in a 20 µl reaction volume. The cycling programme involved initial denaturation of cDNA at 94°C for 10 min, followed by 40 cycles of 94°C for 30s and 60°C for 1 min. Real-time PCR was performed using an Applied Biosystems™ real time PCR system (Thermo Fisher Scientific), and relative mRNA levels were quantified using the 2–ΔΔCt method.
Western blotting
Total protein was extracted from PC12 cells using RIPA buffer, and protein concentration was determined using the BCA method. Equal quantities of protein were then separated using 10% SDS-PAGE and transferred to PVDF membranes (Millipore; Burlington, MA, USA). Subsequently, the membranes were blocked with 5% skimmed milk powder solution, and then incubated with the following primary antibodies overnight at 4°C: rabbit anti-NLRC5 (1: 1 000 dilution; ab105411; Abcam, Cambridge, UK), rabbit anti-Bcl-2 (1: 1 000 dilution; ab32124; Abcam), rabbit anti-Bax (1: 1 000 dilution; ab32503; Abcam), rabbit anti-Caspase3 (1: 500; ab13847; Abcam), rabbit anti-Cleaved-caspase3 (1: 500 dilution; ab32042; Abcam), rabbit anti-TLR-4 (1: 500 dilution; ab13556; Abcam), rabbit anti-MyD88 (1: 1 000 dilution; ab133739; Abcam), rabbit anti-NF-κB p65 (1: 1 000 dilution; ab16502; Abcam), mouse anti-NF-κB p-p65 (1: 1 000 dilution; sc-166748; Santa Cruz, Dallas, TX, USA), mouse anti-GAPDH endogenous control (1: 5 000 dilution; ab8245; Abcam). After incubation, the membranes were washed three times with phosphate buffered saline-Tween 20 (PBST) and further incubated with horse-radish peroxidase (HRP)-conjugated secondary antibody (goat anti-rabbit IgG, 1:10 000 dilution, ab6721; rabbit anti-mouse IgG, 1:10 000 dilution, ab6728; both Abcam) for 2 h at room temperature. Membranes were then washed three times with PBST and exposed using an enhanced chemiluminescence detection system and Amersham imager (Amersham, Shanghai, China). Relative protein levels were calculated by the results normalised to GAPDH.
Cytoplasmic and nuclear detection of phosphorylated p65
Total protein was extracted from PC12 cells using cytoplasmic and nuclear protein extraction kits (CWBIO; Beijing, China). After harvesting the PC12 cells, the cell pellet was resuspended in cytoplasmic protein extraction reagent A and PMSF, and incubated for 10 min on ice. Cytoplasmic protein extraction reagent B and PMSF was added to the cells and mixed, then the tube was centrifuged at 12 000 g for 5 min at 4°C to collect the supernatant comprising the cytoplasmic extract, which was then transferred to a clean pre-chilled tube. The insoluble pellet fraction was resuspended in nuclear protein extraction reagent, and incubated on ice for 30 min. The mixture was then centrifuged at 12 000 g for 10 min at 4°C to collect the supernatant comprising nuclear extract. Cytoplasmic and nuclear protein extracts were separated on SDS-PAGE gels for Western blotting as described above, using anti-p-NFκB p65 (Santa Cruz) to detect phosphorylated p65. Mouse anti-Lamin B (1: 500 dilution, ab16048, Abcam) was used as a nuclear endogenous control and GAPDH was used as the cytoplasmic endogenous control.
ELISA assays
Levels of tumour necrosis factor (TNF)-α, interleukin (IL)-1β and IL-6 in the culture medium of PC12 cells, were detected using enzyme-linked immunosorbent assay (ELISA) kits: Abcam human TNF-α ELISA kit (ab181421); human IL-1β ELISA kit (ab214025); and human IL-6 ELISA kit (ab178013). All ELISAs were performed according to the manufacturer’s instructions. ELISA results were detected at 420 nm using a microplate reader (BioRad, Hercules, CA, USA).
ROS detection
Potential changes in ROS levels after overexpression or silencing of NLRC5 in PC12 cells were detected using ROS assay kits (S0033M; Beyotime, Shanghai, China). Briefly, the PC12 cells were harvested and added to DCFH-DA solution diluted with serum-free medium. The cell suspension was incubated with the DCFH-DA for 20 min in the dark, shaking every 5 min to ensure the complete mixture of cells with DCFH-DA, which is oxidised by ROS into a highly fluorescent compound. At the end of the incubation period, the fluorescence intensity was detected using a fluorescence microplate reader (Infinite F500; Tecan, Mannedorf, Switzerland) at 525 nm.
Apoptosis assay
Apoptosis was detected in PC12 cells by the terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) assay using a TUNEL apoptosis assay kit (Keygen BioTECH, Nanjing, China), according to the manufacturer’s instructions. The PC12 cells were seeded onto sterilized glass and allowed to adhere for 24 h at 37°C. Cells were then fixed using 4% paraformaldehyde and washed three times with cold PBS. Terminal deoxynucleotidyl transferase (TdT) reaction mix was added to the cells, and incubated at 37°C for 1 h. Cells were then rinsed with PBS, and streptavidin-HRP was added and incubated for 1 h in the dark. Finally, DAB solution was added to develop the colour reaction. Cells were observed under light microscopy and photographed. Levels of apoptotic cells were calculated as the mean proportion of positive cells out of total cells in six fields of view per slide.
Oxidative stress assays
Activities of malondialdehyde (MDA) and superoxide dismutase (SOD) in PC12 cells were measured using assay kits (MDA, Cat. No. A003-1-2; and SOD, Cat. No. A001-3-2; both Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s instructions.
Statistical analyses
Data are presented as mean ± SD values of triplicate experiments. Between-group comparisons were determined by Student’s t-test and all statistical analyses were performed using GraphPad Prism software, version 7.0 (GraphPad Software, La Jolla, CA, USA). A P value < 0.05 was considered statistically significant.
Results
NLRC5 suppressed expression of the inflammation factors TNF-α, IL-6 and IL-1β
Blood samples from 50 neonates with cerebral ischemia-reperfusion injury and 50 healthy control neonates were included in this study. RT-PCR analysis showed that blood levels of NLRC5 mRNA were significantly higher in the cerebral ischemia group than in the healthy controls (P < 0.001; Figure 1a).
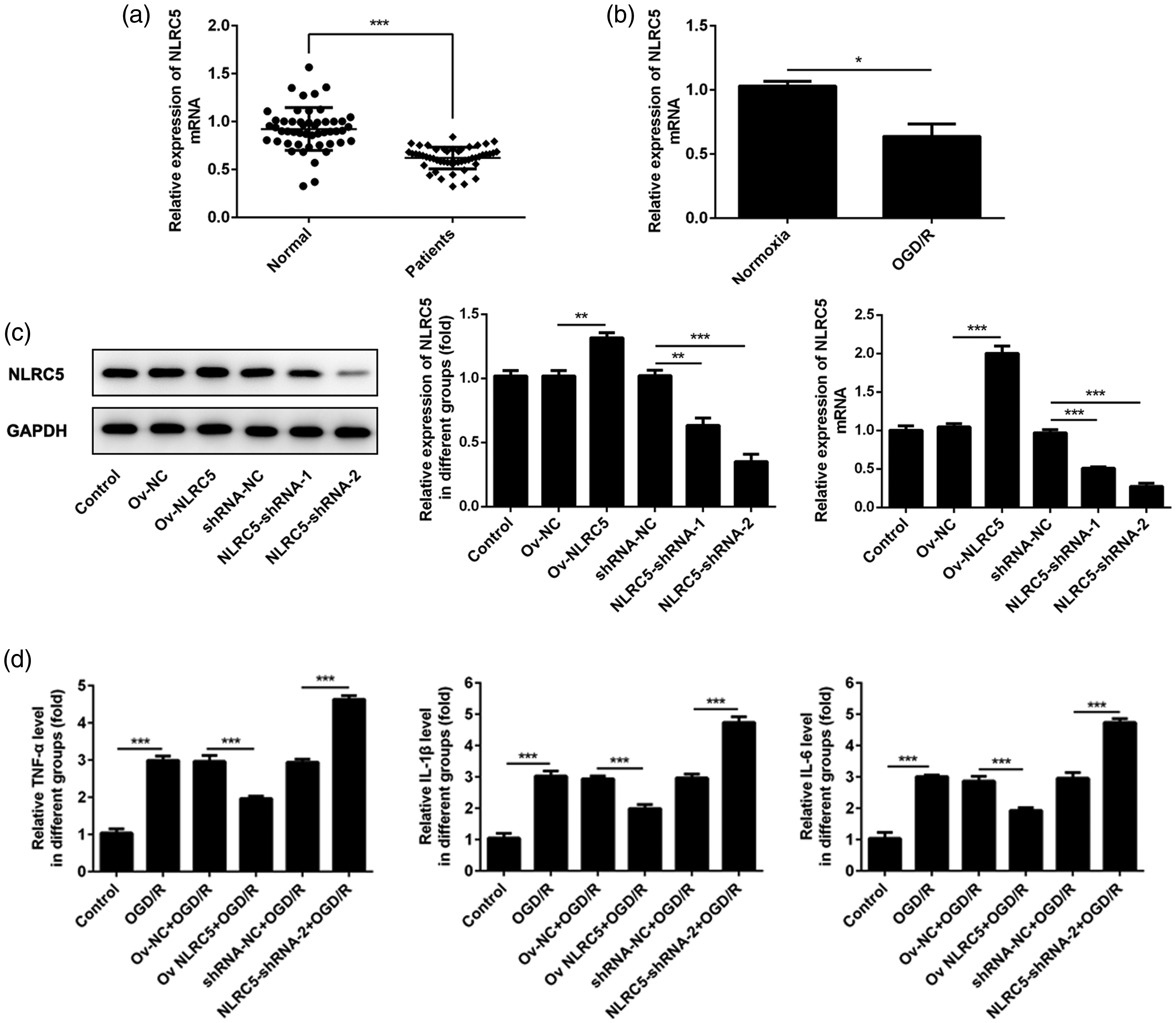
NOD-like receptor C5 (NLRC5) inhibited the expression of tumour necrosis factor (TNF)-α, interleukin (IL)-1β and IL-6: (a) Vertical scatter plots showing blood NLRC5 mRNA levels in neonates with cerebral ischemia (patients) and healthy control neonates (normal), determined by reverse transcription (RT)-polymerase chain reaction (PCR); (b) Levels of NLRC mRNA in PC12 cells, detected by RT-PCR; (c) Protein and mRNA levels of NLRC5 levels in PC12 cells following overexpression and silencing of NLRC5, determined by Western blot and RT-PCR, respectively; (d) Levels of TNF-α, IL-1β and IL-6 in PC12 culture supernatants of PC12 cells under oxygen-glucose deprivation/reperfusion (OGD/R) conditions, detected by enzyme-linked immunosorbent assays. PC12 data presented as mean ± SD of triplicate experiments; *P < 0.05, **P < 0.01, ***P < 0.001 (Student’s t-test). Ov-NC, overexpression negative control; Ov-NLRC5, overexpression of NLRC5; shRNA-NC, scrambled siRNA negative control; NLRC5-shRNA-1 and NLRC5-shRNA-2, NLRC5 silenced with siRNA.
To evaluate NLRC5 mRNA levels in vitro, PC12 cells were cultured under OGD/R and normal conditions. Levels of NLRC5 mRNA were found to be significantly lower in cells cultured under OGD/R conditions (P < 0.05; Figure 1b). Next, NLRC5 upregulation or downregulation was established in PC12 cells to investigate the influence of NLRC5 in OGD/R and normoxia conditions. Western blots and RT-PCR to assess protein and mRNA levels, respectively, showed that NLRC5 overexpression or silencing was successfully established in PC12 cells (Figure 1c). OGD/R conditions commonly induce inflammatory responses, hence the inflammation factors TNF-α, IL-6 and IL-1β were analysed using ELISAs. All three factors were found to be inhibited under OGD/R conditions in cells overexpressing NLRC5, and all were found to be increased under OGD/R conditions in cells silencing NLRC5 (P < 0.001; Figure 1d).
NLRC5 alleviated oxidative stress under OGD/R conditions
Damage due to oxidative stress occurs during the process of cerebral ischemia-reperfusion injury. Analysis of ROS levels in PC12 cells under OGD/R conditions showed that overexpression of NLRC5 supressed ROS production (P < 0.001) and NLRC5 silencing was associated with increased ROS levels (P < 0.01; Figure 2a). In addition, oxidative stress assays showed that SOD, which can act as an antioxidant, was upregulated following overexpression of NLRC5 and inhibited after NLRC5 silencing in OGD/R-treated cells (P < 0.01; Figure 2b). In PC12 cells under OGD/R conditions, MDA, a critical biomarker of oxidative stress, was significantly increased in the NLRC5 silencing group and downregulated in the NLRC5 overexpression group (P < 0.001; Figure 2b).
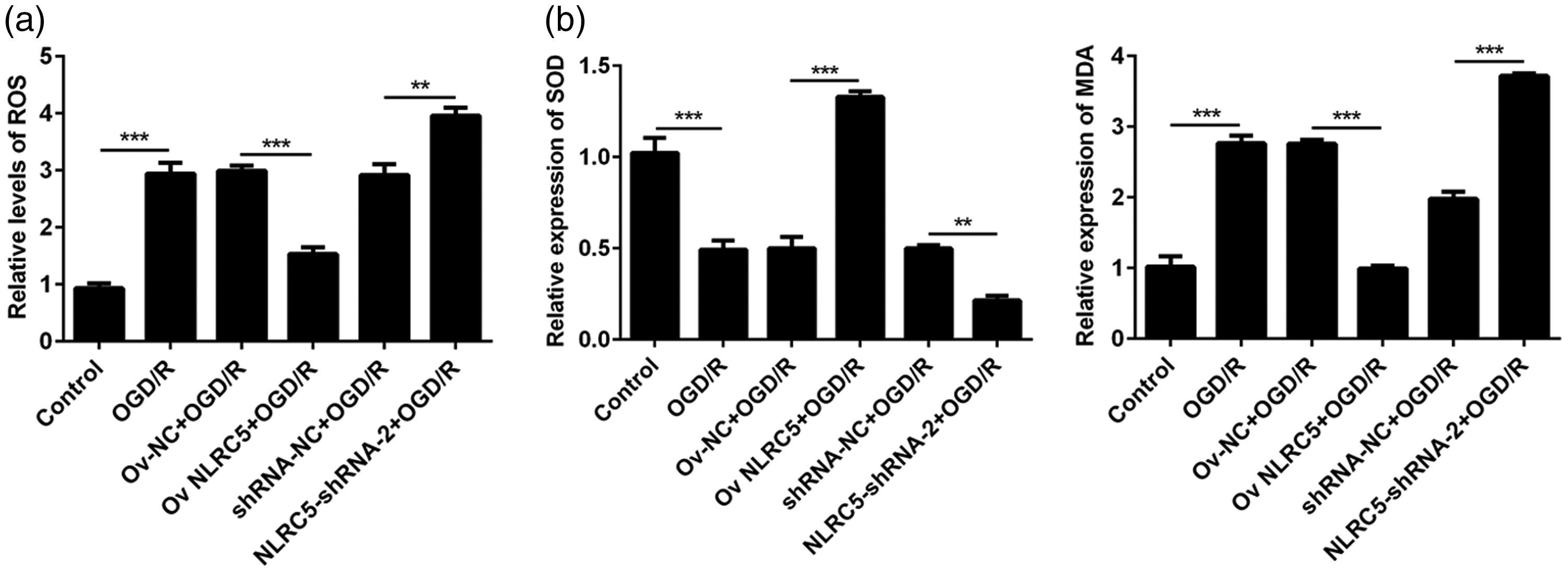
NOD-like receptor C5 (NLRC5) alleviated oxygen-glucose deprivation/reperfusion (OGD/R)-induced oxidative damage in PC12 cells: (a) Reactive oxygen species (ROS) levels in PC12 cells were analysed using a DCFH-DA fluorogenic dye assay; (b) superoxide dismutase (SOD) and malondialdehyde (MDA) levels were detected with specific kits. Data presented as mean ± SD of triplicate experiments; *P < 0.05, **P < 0.01, ***P < 0.001 (Student’s t-test). Ov-NC, overexpression negative control; Ov-NLRC5, overexpression of NLRC5; shRNA-NC, scrambled siRNA negative control; NLRC5-shRNA-2, NLRC5 silenced with siRNA.
NLRC5 reduced PC12-cell apoptosis under OGD/R conditions
Cerebral ischemia-reperfusion injury may induce apoptosis in nerve cells. Therefore, TUNEL assays were used to analyse the effect of NLRC5 overexpression or silencing on apoptosis in OGD/R-treated PC12 cells. TUNEL-positive PC12 cells were increased under OGD/R conditions, and were decreased in cells overexpressing NLRC5, however, NLRC5 silencing enhanced the breakage of DNA double strands, which further increased TUNEL-positive PC12 cells under OGD/R conditions (P < 0.001; Figure 3a). Apoptosis-related proteins were assessed using Western blots, and the results showed that levels of Bax and cleaved-caspase3 were upregulated, while the level of Bcl-2 was reduced under OGD/R conditions after NLRC5 silencing. In contrast, the NLRC5 overexpression group displayed inhibited levels of Bax and cleaved-caspase3 and increased levels of Bcl-2 after OGD/R treatment (Figure 3b).

NOD-like receptor C5 (NLRC5) alleviated PC12-cell apoptosis caused by oxygen-glucose deprivation/reperfusion (OGD/R): (a) Representative photomicrographs showing apoptotic cells stained using terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) with bar graph showing the proportion of apoptotic cells per group; (b) Representative Western blot images for Bcl2, Bax, cleaved caspase3 and caspase3 with relative levels normalised to GAPDH loading control (cleaved caspase3 further normalised to caspase3). Data presented as mean ± SD of triplicate experiments; *P < 0.05, **P < 0.01, ***P < 0.001 (Student’s t-test). Ov-NC, overexpression negative control; Ov-NLRC5, overexpression of NLRC5; shRNA-NC, scrambled siRNA negative control; NLRC5-shRNA-2, NLRC5 silenced with siRNA.
NLRC5 protected PC12 cells from cerebral ischemia-reperfusion injury by targeting the TLR4/MyD88/NF-κB pathway
The TLR4/MyD88/NF-κB pathway is associated with the occurrence and development of inflammation, and knockout of NLRC5 has been shown to promote fibrosis and inflammation in mice fed a high fat diet, through elevating TLR4 and MyD88 expression levels. 8 Thus, it was hypothesized that NLRC5 may target the TLR4/MyD88/NF-κB axis to alleviate cerebral ischemia-reperfusion injury. Levels of TLR4, MyD88 and NF-κB p-p65 were evaluated by Western blot in PC12 cells under OGD/R conditions. Levels of TLR4, MyD88 and NF-κB p-p65 were found to be enhanced under OGD/R conditions and further enhanced with NLRC5 silencing, but the increased levels under OGD/R were suppressed with NLRC5 overexpression (P < 0.05; Figure 4a). Similar trends in the different groups were seen in terms of NF-κB p-p65 levels in both the nucleus and cytoplasm of PC12 cells in response to OGD/R treatment (P < 0.05; Figure 4b).

NOD-like receptor C5 (NLRC5) alleviated oxygen-glucose deprivation/reperfusion (OGD/R)-induced damage of PC12 cells by suppressing the toll-like receptor 4 (TLR4)/myeloid differentiation primary response protein MyD88/nuclear factor kappa B (NF-κB) pathway: (a) Representative Western blot images for TLR4, MyD88, NF-κB p65 and phosphorylated NF-κB (NF-κB p-p65) with relative levels normalised to GAPDH (NF-κB p-p65 further normalised to NF-κB p65 levels); (b) Representative Western blot images for nuclear and cytoplasmic NF-κB p-p65 levels with relative levels of nuclear NF-κB p-p65 (normalised to Lamin B) and cytoplasmic NF-κB p-p65 (normalised to GAPDH). Data presented as mean ± SD of triplicate experiments; *P < 0.05, **P < 0.01, ***P < 0.001 (Student’s t-test). Ov-NC, overexpression negative control; Ov-NLRC5, overexpression of NLRC5; shRNA-NC, scrambled siRNA negative control; NLRC5-shRNA-2, NLRC5 silenced with siRNA.
Discussion
This research aimed to investigate the effect and underlying mechanism of NLRC5 in cerebral ischemia-reperfusion injury. Mounting evidence suggests that NLRC5 exerts anti-inflammatory effects under impaired conditions in a variety of tissues.13,14 Cerebral ischemia-reperfusion injury is a disease characterised by inflammation and apoptosis of nerve cells induced by hypoxia and hypoglycaemia. 2 The results of the present study revealed that NLRC5 expression was reduced in the blood of neonates with cerebral ischemia compared with healthy controls. Furthermore, higher levels of NLRC5 were shown to alleviate the in vitro damage induced by cerebral ischemia-reperfusion in PC12 cells.
Cerebral ischemia may cause lack of oxygen and glucose in the brain, which could result in effects on normal brain function. Additionally, reperfusion of blood following insufficient blood supply may produce excessive ROS and free radicals, further impairing the brain tissues. 2 The mechanism underlying the damage induced by cerebral ischemia-reperfusion is complicated, but two factors, inflammation and ROS, are still recognized to be crucial parts in the process.15–17 NLRC5 has been shown to relieve allergic airway inflammation, 18 and suppress the NF-κB pathway to alleviate the inflammatory reaction. 19 In the present paper, levels of TNF-α, IL-6 and IL-1β were found to be reduced by overexpression of NLRC5, and together with published data, these results indicate that NLRC5 may inhibit the inflammatory reaction in cerebral ischemia-reperfusion injury.
Excessive production of ROS is shown to be closely related to the injury induced by cerebral ischemia-reperfusion,20–22 which may contribute to brain damage. Moreover, oxidative stress-induced damage is the main cause of myocardial ischemia-reperfusion injury, and ROS levels are promoted in cardiac muscle tissue following ischemia reperfusion. 23 In the present study, the level of ROS was inversely related to NLRC5 expression in OGD/R-treated cells, suggesting that NLRC5 may suppress ROS production. SOD, a powerful antioxidant enzyme, is decreased in renal tissue following ischemia reperfusion, 24 and levels of MDA, the critical product of lipid peroxidation, are enhanced after ischemia reperfusion in renal tissue.25,26 In the present research, the inhibition of NLRC5 was found to be associated with increased MDA and reduced SOD under cerebral ischemia-reperfusion conditions. The combined present and published results discussed above imply that NLRC5 may alleviate oxidative stress in cerebral ischemia-reperfusion. Furthermore, various studies have maintained that nerve cell apoptosis in cerebral ischemia-reperfusion is also critical for brain damage.27–29 Results of the present study revealed that NLRC5 alleviated the cell apoptosis induced by OGD/R conditions.
Toll-like receptor (TLR)4 plays a pivotal role in the occurrence and development of the inflammatory response. 30 NF-κB, a downstream protein of TLR4, is activated during the inflammatory reaction and may promote the expression of various proinflammatory cytokines and chemokines. 31 MyD88 is a major adapter and exerts a vital role in delivering the signals from TLR4 to NF-κB, thus, the TLR4/MyD88/NF-κB pathway is related with the occurrence and development of the inflammation reaction. 32 NLRC5 has been reported to exert suppressive effect of NF-κB and the inflammation pathway. 8 Some research has demonstrated the existence of an inverse correlation between the NLRC5 and NF-κB pathways.13,33,34 NLRC5 silencing enhanced TLR4/MyD88/NF-κB signals in mice with high fat-induced myocardial damage. 13 Collectively, NLRC5 silencing has been shown to promote NF-κB activation, possibly through inducing the formation of phosphorylated p65 and promoting the translocation of p65 from the cytoplasm into the nucleus, while TLR4 activation has been shown to recruit MyD88 to induce NF-κB activation. The present results also indicated that levels of TLR4, MyD88 and NF-κB were negatively related with the expression of NLRC5.
In conclusion, results of the present study, combined with previously published data, provides supportive evidence that NLRC5 lowers oxidative damage, apoptosis and the inflammatory response by suppressing the TLR4/MyD88/NF-κB pathway in cerebral ischemia-reperfusion injury.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the funding project of Bengbu Medical College of Science and Technology Development (No. BYKF1867) and key project of natural science of colleges and universities in Anhui province (No. KJ2019A0378).
