Abstract
Objective
This study aimed to investigate whether coenzyme Q10 is effective in preventing exercise-induced oxidative stress and muscle damage.
Methods
Fourteen randomized controlled studies examining the effects of supplementation with coenzyme Q10 analogs on postexercise oxidative stress and muscle damage were identified through searches in literature databases, including PubMed, the Cochrane Library, Embase, and Web of Science, and then the quality of the included studies was assessed. Quantitative and qualitative analyses were performed.
Results
The study screened 14 randomized controlled trials that included a total of 433 subjects. The results demonstrated that oral coenzyme Q10 elevated blood coenzyme Q10 concentration (standardized mean difference: 2.710, 95% confidence interval: 1.57–3.85, p < 0.00001) and reduced blood malondialdehyde concentration (standardized mean difference: −0.289, 95% confidence interval: −0.541 to −0.038, p = 0.024). Additionally, oral coenzyme Q10 was found to reduce blood creatine kinase values (standardized mean difference: −1.532, 95% confidence interval: −2.856 to −0.209, p = 0.023), suggesting a potential protective effect on skeletal muscle. The metabolism-related blood lactate and maximal oxygen uptake levels were not affected by coenzyme Q10 (standardized mean difference: −0.68, 95% confidence interval: −1.89 to 0.53, p = 0.271; standardized mean difference: −0.156, 95% confidence interval: −0.79 to 0.478, p = 0.630).
Conclusions
Coenzyme Q10 may reduce exercise-induced oxidative stress on blood malondialdehyde and exert a protective effect on muscle; however, no effect was observed from the anaerobic and aerobic metabolism of the organism.
Introduction
Appropriate exercise helps maintain redox balance in the body; however, high-intensity exercise may cause redox imbalance in muscle tissue, leading to muscle fatigue and damage. Some studies suggest that reducing oxidative stress in the body may enhance exercise performance, 1 but an appropriate level of oxidative stress over the long term may be beneficial for enhanced exercise capacity and performance. 2
The oxygen species and free radicals generated during oxidative stress not only block electron transfer but also disrupt energy metabolism and biosynthesis. These processes interfere with the ion channel transportation on the cell membrane, promote lipid peroxidation of the cell membrane, and ultimately result in cell damage. To mitigate the adverse effects of exercise-induced oxidative stress on the body, a variety of antioxidant supplements, including vitamin C, vitamin E, and other antioxidant vitamins, are available in the market for use. Among these antioxidants, coenzyme Q10 (CoQ10) acts directly on the inner mitochondrial membrane, where it increases the efficiency of electron transfer and stabilizes redox balance and energy metabolism. Moreover, it maintains the cell membrane stability and facilitates the regeneration and recycling of vitamins C and E.3–5 CoQ10-related supplements are primarily used in patients with cardiomyopathy and mitochondrial dysfunction. Some studies have also suggested that CoQ10 supplementation may reduce fatigue, improve physical performance, and enhance the aerobic capacity of skeletal muscle cells.6,7
In this study, CoQ10 analogs, including oxidized CoQ10 (ubiquinone), reduced CoQ10 (ubiquinol), and mitochondria-targeted antioxidant MitoQ (mitoquinone), were investigated—all of which are capable of localizing to the inner mitochondrial membrane and play a role in facilitating electron transfer. Among these compounds, ubiquinol and mitoquinone exhibit high water solubility. Mitoquinone, a CoQ10 analog derived from idebenone, possesses antioxidant capacity, which is consistent with natural CoQ10. Therefore, this study aimed to investigate the effects of different CoQ10 types on exercise-induced oxidative stress and muscle damage.
Methods
Search strategy
The meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement. 8 Two researchers searched for data in the following electronic databases in 2025: PubMed, Embase, the Cochrane Library, and Web of Science. Also, we searched for reference lists of the included literature and other possible articles using Google Scholar. The following subject terms and keywords constitute the search strategy (using PubMed as an example): ((“Ubiquinone” [Mesh]) OR ((((Coenzyme Q10[Title/Abstract]) OR (ubiquinol[Title/Abstract])) OR (mitoquinone[Title/Abstract])) OR (MitoQ[Title/Abstract])) AND (exercise or train*) AND (randomized controlled trial). Document management was performed using EndNote software. The literature screening is presented in Figure 1.
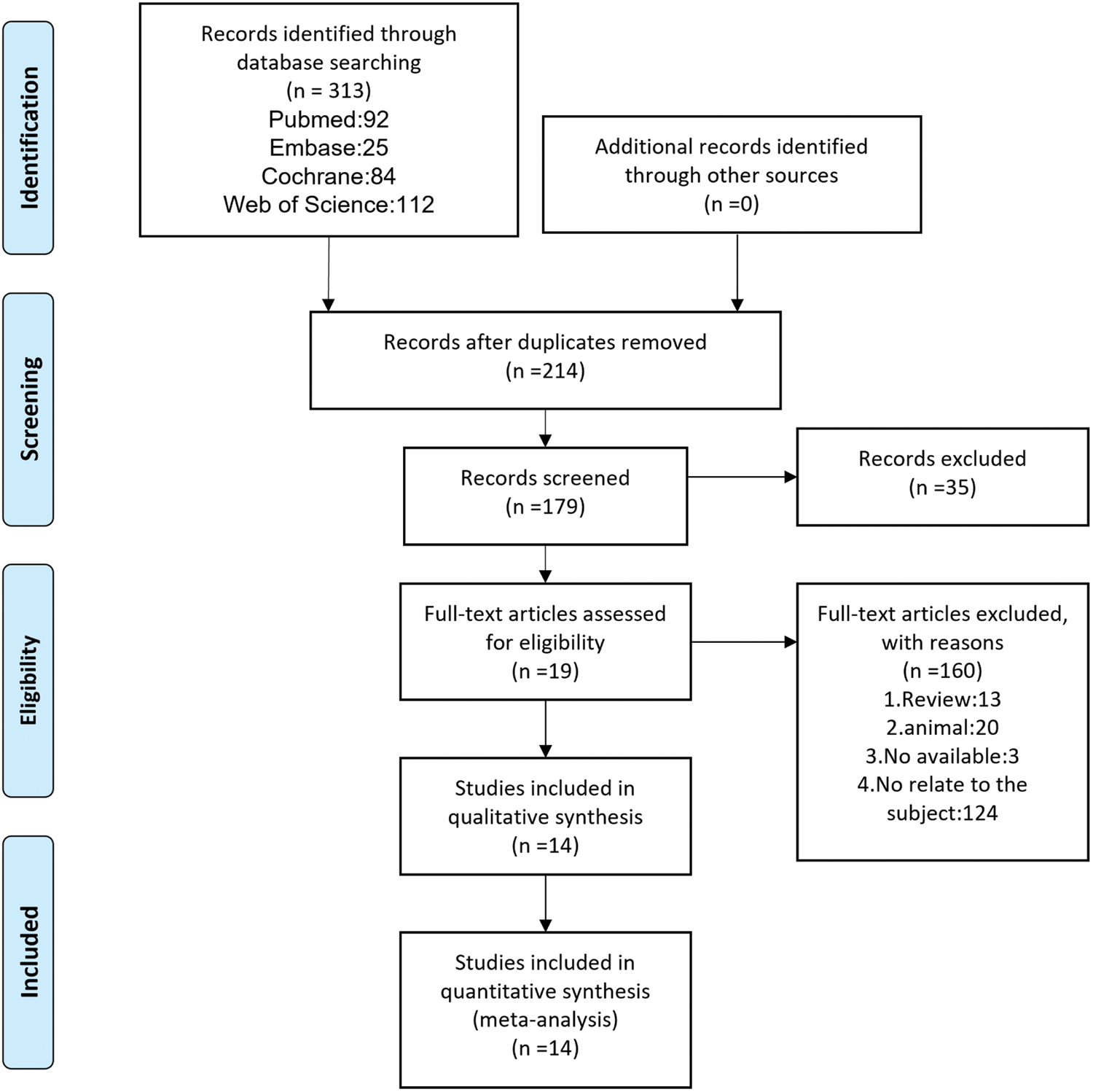
The PRISMA flow chart of literature screening. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (a) randomized controlled or crossover controlled trials; (b) administration of safe doses of CoQ10-related analogs; (c) subjects aged 18 years or older who were able to tolerate strong exercise and had not been taking any other antioxidant for more than 1 month; and (d) the study outcome indicators including blood CoQ10 concentrations, blood and cellular oxidative stress indicators of blood malondialdehyde (MDA), muscle damage indicators of blood creatine kinase (CK), aerobic metabolism indicators of maximal oxygen uptake (VO2max), and anaerobic metabolism indicators of blood lactate.
The exclusion criteria were as follows: (a) intervention supplements mixed with antioxidants other than CoQ10, (b) studies without exercise interventions or other study types, and (c) duplicate publications, unavailability of the original article, incomplete experiments, or incomplete data in the article.
Data extraction
Two reviewers (Yangqi Zhang and Yilang He) independently extracted data from the literature according to the predefined inclusion and exclusion criteria. Disagreements were resolved through discussion or consultation with a third reviewer (Mingling Ma). The extracted data included title, authors, year, type of study design (randomized controlled or crossover controlled), sample size (experimental and control groups), age, intervention characteristics (type of CoQ10 used, dosage, and duration of the intervention), type of exercise (aerobic, anaerobic, or other mixed patterns of exercise), and outcome indicators such as blood-related (CoQ10, MDA, CK, and lactate) and cardiorespiratory function (VO2max). Data types were converted when the statistical types of the collected data were inconsistent, such as standard error of the mean to standard deviation conversion.
Risk of bias assessment
Risk of bias was assessed by two reviewers according to the Cochrane Handbook guidelines, evaluating for randomization, allocation concealment, blinding of participants and researchers, blinding of outcome assessments, incomplete outcome data, selective reporting of outcomes, and other potential sources of bias. Risk of bias was assessed using the Cochrane risk of bias tool, and each outcome was assessed at the level of low risk, high risk, or uncertain risk. Studies with at least four low-risk domain outcomes were considered to be of relatively good quality. Studies with one or more domains rated as high risk were categorized as poor quality, while the others were considered to be of fair quality. A third reviewer was consulted when necessary. The overall risk of bias is illustrated in Figure 2.

Risk of bias summary.
Grading of Recommendations Assessment, Development, and Evaluation (GRADE) assessment
The quality of evidence for each outcome measure was assessed using the GRADE system. This system evaluates evidence across five dimensions: risk of bias, inconsistency, indirectness, imprecision, and publication bias. Given that all included studies were randomized controlled trials, the quality of initial evidence grade was rated as “high” and subsequently downgraded based on the assessment results across the aforementioned dimensions. The evaluation process was conducted independently by two reviewers, with disagreements resolved through consensus.
Statistical analysis
Statistical analyses were performed using Stata (version 12.0) and Reviewer Manager (version 5.3.0). A random-effects model analysis was performed first to account for the heterogeneity among studies. Continuous variables were assessed using standardized mean difference (SMD) and 95% confidence intervals (CI), while binary variables were assessed using odds ratio (OR) and 95% CI. Data transformations were performed as estimates when data types were not uniform. Interstudy heterogeneity was analyzed qualitatively and quantitatively using χ2 and I2 statistics. Pooled p values < 0.05 were considered statistically significant.
Results
Selection process
A total of 313 publications were identified through electronic databases. After excluding 134 duplicates, reviews, animal experiments, and unavailable literature, 14 randomized controlled trials were included in the meta-analysis; among these, two articles investigated ubiquinol, four examined MitoQ (mitoquinone), and eight focused on CoQ10 (ubiquinone).
Description of the included studies
The analysis included 14 eligible studies,9–22 comprising a total of 433 subjects, published between 1995 and 2023. The basic information of the included studies is described in Table 1, including author names, sample sizes of the experimental and control groups, participant ages, study design, type of CoQ10intervention, intervention duration, and exercise modality. Outcome indicators included blood CoQ10 concentrations, lipid peroxidation (blood MDA), muscle damage (blood CK), and indices of anaerobic and aerobic metabolism (blood lactate and VO2max). The relevant data used for performing quantitative and qualitative analyses are presented in Table 2.
Basic characteristics of the studies included.

C: control group; T: treatment group.
Data on the outcome indicator of the study included.

C: control group; d: day; F: female; M: male; m: month; T: treatment group; VO2max: maximal oxygen uptake.
Outcome of meta-analysis
Blood CoQ10 concentration
Seven studies9,10,12,14–16,18 reporting blood CoQ10 concentrations after oral CoQ10 are presented in Figure 3. This meta-analysis demonstrated that oral CoQ10 elevated plasma CoQ10 concentrations (SMD: 2.710, 95% CI: 1.57–3.85, p < 0.00001, I2 = 90%). However, subgroup analysis stratified by age partially reduced the observed heterogeneity. Furthermore, subgroup analyses indicated that CoQ10 supplementation elevated plasma CoQ10 concentrations (Table 3). The primary sources of heterogeneity were explored using Galbraith’s star plot (Figure S1). The primary contributors of heterogeneity were the studies conducted by Cooke et al., Gokbel et al., and Bloomer et al. Sensitivity analyses revealed that although heterogeneity existed, the heterogeneity of the individual studies did not affect the combined results. No evidence of publication bias was detected using quantitative Egger’s test (p = 0.112) (Figure S2).

Forest plot of the effect of CoQ10 supplementation on blood CoQ10. CoQ10: coenzyme Q10.
Subgroup analysis of CoQ10 supplementation on Serum CoQ10 and CK.

CI: confidence interval; CK: creatine kinase; CoQ10: coenzyme Q10; SMD: standardized mean difference; VO2max: maximal oxygen uptake.
Blood MDA
In this meta-analysis,9,10,12,13,15,16,21,22 blood MDA concentrations were used to measure the effect of CoQ10 supplementation on lipid peroxidation associated with exercise-induced oxidative stress. Lower blood MDA concentration was observed in the experimental group relative to the control group across all included randomized controlled trials, although several of these studies have concluded that the difference was not statistically significant. However, the results of this meta-analysis showed that CoQ10 resulted in a better response to lipid peroxidation, which means that the difference was statistically significant (Figure 4). The results of heterogeneity were satisfactory (SMD: −0.289, 95% CI: −0.541 to −0.038, p = 0.024, I2 = 0%). However, at the same time, there was a publication bias (p = 0.007) in the quantitative detection of Egger’s test (Figure S3). No literature could be trimmed using the cut-and-paste method. This was explained by the fact that despite the presence of publication bias, the overall results of the meta-analysis remained relatively stable and reliable.

Forest plot of the effect of CoQ10 supplementation on blood MDA. MDA: malondialdehyde; CoQ10: coenzyme Q10.
Blood CK
High expectations were placed on the meta-analysis results for blood CK concentrations.11,14,16,17 The initial meta-analysis revealed no statistically significant difference between the experimental group and the control group (p = 0.082, I2 = 89.4%), which was unexpected. To increase the effective sample size and strengthen the evidence, we considered the specificity of studies conducted by Emami and Bazargani-Gilani. This study explored the effects of CoQ10 combined with precooling on exercise performance using four parallel groups: placebo, CoQ10, precooling, and CoQ10+precooling. The initial meta-analysis included only the placebo and CoQ10 groups, while the second meta-analysis included placebo versus CoQ10 and precooling versus CoQ10+precooling as the results of two independent studies (Emami and Bazargani-Gilani a and b) and obtained different results (SMD: −1.532, 95% CI: −2.856 to −0.209, p = 0.023, I2 = 86.7%) (Figure 5). CoQ10 supplementation appears to be beneficial in preventing muscle damage. Subgroup analysis stratified by dose and duration failed to identify potential sources of heterogeneity (Table 3). Further subgroup analyses revealed that the reductions in plasma CK levels following CoQ10 supplementation were observed only under specific conditions: short-term supplementation (within 2 weeks) and high-dose administration (≥300 mg/day). However, the second meta-analysis was similarly heterogeneous, with Galbraith star plots indicating that heterogeneity originated from Emami and Bazargani-Gilani a and b and Ostman et al. studies. Sensitivity analyses demonstrated that the exclusion of individual studies did not affect the results of meta-analysis (Figure S4). Publication bias was also detected quantitatively using Egger’s test (p = 0.008), a result that was expected (Figure S5). This indicates that although publication bias was present, it had no significant effect on the results of meta-analysis, suggesting that the results of meta-analysis are relatively stable and reliable.

Forest plot of the effect of CoQ10 supplementation on blood CK. CK: creatine kinase; CoQ10: coenzyme Q10.
Blood lactate
Four studies9,11,18,22 assessed anaerobic metabolism during exercise based on blood lactate. The results of the meta-analysis (Figure 6) indicated that CoQ10 did not have a positive effect on plasma lactate (SMD: −0.68, 95% CI: −1.89 to 0.53, p = 0.271, I2 = 89.7%). Forest plot suggested that the heterogeneity was primarily driven from Emami and Bazargani-Gilani study, likely due to substantial differences in participant age compared with the other studies. After exclusion of the study by Emami and Bazargani-Gilani, heterogeneity was markedly reduced (I2 = 18.4%), and there was no change in the overall results (p = 0.421).

Forest plot of the effect of CoQ10 supplementation on blood lactate. CoQ10: coenzyme Q10.
VO2max
Four studies12,15,17,22 used VO2max to assess aerobic capacity and endurance exercise performance (Figure 7). The results of the meta-analysis demonstrated no statistically significant difference between CoQ10 supplementation and the control group (SMD: −0.156, 95% CI: −0.79 to 0.478, p = 0.630, I2 = 66.5%). The forest plot indicated that the heterogeneity was primarily derived from Ostman et al. study. This heterogeneity was likely due to differences in aerobic and anaerobic exercise protocols, while the exercise pattern of the other three studies was aerobic. After exclusion of the study by Ostman et al., heterogeneity was significantly less, and the results of the meta-analysis remained unchanged (p = 0.461, I2 = 0%).

Forest plot of the effect of CoQ10 supplementation on VO2max. VO2max: maximal oxygen uptake; CoQ10: coenzyme Q10.
GRADE assessment
The quality of evidence for assessment results was generally low to very low (Table 4). Evidence quality for blood CoQ10 and MDA was downgraded to low due to risks of imprecision and inconsistency. Evidence quality for the muscle damage biomarker CK was very low due to the presence of publication bias. Evidence quality for metabolic-related indicators, blood lactate and VO2max, was very low.
Summary assessment of study findings and evidence quality using the GRADE approach.

CI: confidence interval; CK: creatine kinase; CoQ10: coenzyme Q10; GRADE: grading of recommendations, assessment, development, and evaluation; MDA: malondialdehyde; No: number; VO2max: maximal oxygen uptake.
Assessing risk of bias using the Cochrane risk of bias tool.
Crossing the 95% CI line or having a small sample size.
I2 > 50% or unexplained heterogeneity.
Factors related to participants, interventions, or outcomes were not observed to limit the generalizability of the results.
Small sample size or asymmetric funnel chart.
Discussion
This study included 14 randomized controlled studies and analyzed the effects of CoQ10 on exercise-induced oxidative stress, muscle damage, and metabolism. Our results demonstrated that CoQ10 supplementation effectively elevated blood CoQ10 concentrations and attenuated lipid peroxidation, as indicated by blood MDA levels. In addition, CoQ10 appeared to exert a potential protective effect on muscle and did not seem to affect the metabolism and exercise endurance of the subjects during exercise.
In the present study, CoQ10 supplementation was confirmed to be effective in elevating blood CoQ10 concentrations. Although these findings were favorable, the primary objective was to explore whether there was a link between this type of antioxidant and the physiological and physical performance of muscles during exercise. With the exception of the study by Cooke et al., 10 which reported that CoQ10 supplementation during acute exercise elevated CoQ10 concentrations in skeletal muscle, the other included studies mention that oral CoQ10 does not elevate CoQ10 concentrations in muscle tissue. The results of subgroup analysis based on age and duration were consistent with the overall results; however, the heterogeneity within each subgroup remained high, which must be interpreted with caution. The CoQ10 types included in this study were ubiquinone, ubiquinol, and MitoQ. Among these, MitoQ appears the most promising, as the other two types of CoQ10 are less stable and bioavailable. However, there is still insufficient clinical evidence to prove that MitoQ can elevate CoQ10 concentration in muscle tissue, which is attributed to various aspects such as oral dosage and intestinal absorption function. This seems to be a common question in many other clinical trials investigating antioxidant supplementation; hence, many scholars have suggested that future studies should place a greater emphasis on the accumulation of CoQ10 within muscle tissue.
We used blood MDA as evidence of lipid peroxidation. The results of the meta-analysis reflect the acquisition of a certain resistance to oxidative stress with CoQ10 supplementation. Several included studies15,16,21,22 did not find any effect of CoQ10 supplementation on MDA. Moreover, Cooke et al. 10 reported that MDA level was significantly higher in the CoQ10 group than in the placebo group after aerobic and anaerobic exercise, which was explained by the fact that the method of plasma MDA measurement was influenced by other components of blood. These findings indicate that measurements of blood samples were not sufficient to account for physiology in the tissues, and it has also been previously demonstrated that anaerobic exercise did not affect MDA in the blood of all subjects. 23
In addition to evaluating the redox state in the body, the primary objective of this study was to investigate the effects of CoQ10 on muscle after exercise. It has been suggested that after exercise, inflammatory cells penetrate into damaged skeletal muscle to generate reactive oxygen species (ROS), leading to muscle cell membrane damage. 24 CoQ10 stabilizes phospholipid structures within the cell membrane while facilitating the clearance of ROS and free radicals. After muscle damage, CK leaks into the bloodstream through the muscle cell membranes; therefore, muscle damage was assessed using plasma CK values. Among the included studies, only the study by Kon et al. 14 reported statistically significant differences in blood CK levels between the two groups. No differences were observed in other studies. Okudan et al. 16 suggested that different exercise patterns influence the degree of muscle damage. Rapid centrifugal exercise elicits more CK release than slow centrifugal exercise. 25 Other studies have also indicated that plasma CK levels are influenced not only by the intensity and duration of exercise but also by an individual’s training status.26,27 It should be noted that skeletal muscle is considered the preferred tissue for assessing ubiquinone levels. 28 Duncan et al. 29 study reported that the coenzyme Q concentrations in monocytes correlate with those in skeletal muscle. These findings suggest that plasma CK levels are not a gold standard marker of muscle damage and that greater emphasis should be placed on muscle tissue–based assessments. The meta-analysis procedures have been described in detail in the “Results” section, and subgroup analyses were performed based on duration and dose. Subgroups with a duration of 2 weeks or less, or dosages exceeding 200 mg/day, demonstrated significant reductions in plasma CK levels. These findings provide valuable reference for individualized CoQ10 supplementation protocols. Although the final results were consistent with the overall results, heterogeneity remained high, indicating the need for further clinical trials to provide relevant data.
In addition, we used blood lactate as an indicator of anaerobic metabolism and VO2max as an indicator of aerobic metabolism. The meta-analysis results showed that CoQ10 supplementation had no significant effect on these two indicators. High metabolic rates and reduced cellular pH are known to accelerate ROS production during exercise. 30 Bloomer et al. 9 reported that CoQ10 supplementation did not improve training performance during either anaerobic or aerobic exercise. Emami and Bazargani-Gilani 11 suggested that maintaining plasma CoQ10 concentrations may reduce anaerobic glycolytic metabolic activity, with decreased glycolysis enzyme activity resulting from reduced pH levels. In contrast, Broome et al. 19 suggested that MitoQ may enhance anaerobic metabolism during high-intensity exercise or preserve exercise endurance by increasing skeletal muscle tolerance to low pH conditions. It is necessary to recognize that the effects of CoQ10 on anaerobic metabolism may be bidirectional. VO2max is considered to be the most commonly used method to assess aerobic capacity and is closely related to the role of CoQ10. Accumulation of free radicals can lead to exercise-related exhaustion and fatigue. It has been suggested that CoQ10 functions not only as an antioxidant but may also enhance skeletal muscle aerobic metabolism through pro-oxidant mechanisms. 31 CoQ10 localizes to the mitochondria, where it increases electron transfer efficiency within the respiratory chain, potentially accelerating aerobic metabolism and reducing lactate accumulation.32,33 Shill et al. 22 demonstrated that MitoQ supplementation did not affect changes in aerobic capacity of skeletal muscle or whole-body during exercise. Overall, VO2max is a key determinant of the time from exercise to exhaustion, helping the body to tolerate high-intensity training; however, variability exists. Different individuals exhibit varying mitochondrial densities, leading to different abilities to adapt to the aerobic environment, and it has also been suggested that ensuring homogeneity in mitochondrial densities is beneficial for research. 3
This meta-analysis not only assessed the effects of CoQ10 supplementation on postexercise oxidative stress but also evaluated muscle damage and metabolic outcomes. In addition, we included studies related to mitochondria-targeted coenzyme Q (MitoQ) in the analysis, representing a novel analytical approach. This study also has significant practical implications. It demonstrates that CoQ10 supplementation aimed at optimizing athletes’ redox status may directly translate into faster post-training recovery and reduced muscle damage. Furthermore, it provides crucial theoretical foundations and a checklist of factors for future assessments of athletes’ baseline physiological states, enabling the development of individualized supplementation protocols based on dose and duration. Combined with other authors’ meta-analysis34–36 on CoQ10 and exercise, this study further validates the potential of CoQ10 to reduce oxidative stress and alleviate muscle damage. Furthermore, despite the limited number of included studies, this study aimed to examine the effects of CoQ10 on human metabolism using VO2max and lactate levels, with the aim of further exploring the impact on athletic performance. However, several limitations to this study must be acknowledged. First, significant heterogeneity was observed in the results of the studies, depending on the subjects recruited; the type, dose, and duration of antioxidants, the exercise protocols; and the method used to assess the body’s response to oxidative stress, making it difficult to homogenize the results. Second, as mentioned in many studies, the measurement samples were primarily based on blood samples, with limited analyses on the muscle tissues, reducing the strength of the conclusion. In addition, insufficient data were available to evaluate the contribution of CoQ10 to physical performance during exercise. Although CoQ10 was hypothesized to improve core performance indicators, such as VO2 max and lactate threshold, this study demonstrated no such improvements in these parameters as anticipated. This key finding suggests that the actual physiological role of CoQ10 may be more complex than traditionally understood. Its benefits may not directly enhance athletic performance but may instead involve alternative mechanisms, such as promoting recovery or reducing muscle damage. The study protocol was not registered (e.g. in Prospective Register of Systematic Reviews (PROSPERO)), which may have potentially introduced selection bias through modifications to the study design or replacement of outcome measures during the research process to obtain positive results. To comprehensively evaluate the physiological effects of CoQ10 in athletes, future research should consider longer-term, larger-scale, standardized clinical trials. Additional biochemical markers, such as prostaglandins and superoxide dismutase, can be measured to measure oxidative stress. Imaging data can be used to evaluate muscle damage. Furthermore, performance scales can be employed to assess physical athletic performance in different athletes, such as swimmers and weightlifters. In addition, the quality of evidence (rated as low to very low) suggests that future studies may likely alter the current conclusions. Such evidence should be interpreted with extreme caution when informing clinical guidelines or clinical decision-making. Finally, given the limited number of studies included, our analysis suggests the potential for publication bias, which may result from the nonpublication of studies with negative or neutral outcomes. Such bias may lead to an overestimation of the true intervention effect, potentially affecting the reliability of the results. Caution should be taken when interpreting these findings in clinical practice.
Conclusion
This meta-analysis concludes that supplementation with CoQ10-related analogs can increase CoQ10 concentrations, reduce oxidative stress on blood MDA levels, and protect muscle tissues; however, there was a high degree of heterogeneity in the included studies. Several prospects for future research in this area emerge from these findings. Future studies should place direct emphasis on muscle tissue assessments. Also, this analysis included fewer studies related to MitoQ, a novel type of CoQ10. There are also a few studies comparing MitoQ with traditional CoQ10, and further studies are required to specify its advantages.
Supplemental Material
sj-zip-1-imr-10.1177_03000605251411151 - Supplemental material for Effects of coenzyme Q10 analogs on oxidative stress, muscle, and metabolism after exercise: A meta-analysis and systematic review
Supplemental material, sj-zip-1-imr-10.1177_03000605251411151 for Effects of coenzyme Q10 analogs on oxidative stress, muscle, and metabolism after exercise: A meta-analysis and systematic review by Yangqi Zhang, Yilang He, Mingling Ma, Hui Dong and Zijian Ma in Journal of International Medical Research
Footnotes
Acknowledgments
The authors sincerely thank Professor Yongxiang Wang from Northern Jiangsu People’s Hospital for his valuable advice on topic selection and statistical analysis.
Author contributions
Yangqi Zhang: Research design, Search literature, Data Extraction, Quality Evaluation, Analysis and interpretation of data, Writing-original draft. Yilang He: Search literature, Data Extraction, Methodology, Quality Evaluation, Analysis and interpretation of data. Mingling Ma: Search literature, Acquisition of data, Quality Evaluation. Hui Dong: Research design, Revision, Methodology. Zijian Ma: Research design, Revision, Methodology.
Data availability statement
The data that support the findings of this study were derived from the following resources available in the public domain.
Declaration of conflicting interests
The authors have no relevant financial or non-financial interests to disclose.
Funding
This study did not receive any dedicated funding from public, commercial, or nonprofit funding agencies.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
