Abstract
Budd-Chiari syndrome is a disorder that is characterized by obstruction of hepatic venous outflow, with thrombosis being the primary cause of the obstruction. This case report describes a 47-year-old Indian male presenting with distension of the abdomen and umbilical swelling for 1 year. Investigations revealed hepatic venous outflow obstruction and a partial web with focal calcification in the Inferior Vena Cava. Despite using standard medical therapeutic options such as diuretics, beta-blockers, and anticoagulation, his ascites remained uncontrolled. Interventional radiology with inferior vena cava venoplasty using 10 and 14 mm angioplasty balloons was performed, leading to the resolution of inferior vena cava stenosis and improved condition. Post-procedure, the patient was put on anticoagulation therapy and was discharged in good condition. This case highlights the successful management of Budd-Chiari syndrome with inferior vena cava occlusion using a multidisciplinary approach combining interventional radiology and medical therapy.
Introduction
Budd-Chiari syndrome (BCS) is an uncommon condition that results from the blockage of hepatic venous outflow. This obstruction can be either thrombotic or non-thrombotic, occurring at various points along the venous pathway, ranging from the hepatic venules to the junction of the inferior vena cava (IVC) and right atrium. Thrombosis is the primary cause of hepatic vein obstruction, leading to increased pressure within the liver veins and subsequent blood congestion in the liver tissue. The majority of individuals with BCS have an underlying condition that increases the risk of blood clot formation. In fact, primary intravascular thrombosis is the most common cause of obstruction. 1
Approximately 75% of patients have at least one inherited or acquired hypercoagulable state, while 25% have multiple etiologic factors. 2 Hepatic vein thrombosis occurs in up to 12% of patients with paroxysmal nocturnal hemoglobinuria and is the leading cause of mortality in these individuals. Notably, hemoglobinuria may be absent at the time of thrombosis onset or diagnosis.3,4 BCS, in conjunction with underlying thrombophilias, may also be associated with conditions such as ulcerative colitis, celiac disease, or abdominal trauma. 1 In Asian countries, BCS is more prevalent in men, with the most common cause being obstruction of the IVC or a combination of IVC and hepatic vein occlusion. 5 The primary surgical treatments for BCS include surgical shunts, transjugular intrahepatic portosystemic shunt (TIPS), catheter-directed thrombolysis, and liver transplantation (LT).
In this case report, we describe a 47-year-old male with BCS due to IVC occlusion who did not have typical risk factors and was successfully treated using an IVC venoplasty procedure and standard medical management.
Case presentation
A 47-year-old Indian man was admitted to the hospital with complaints of swelling in the umbilical region and abdominal distention for 1 year. The distention had an insidious onset and gradually progressed. It was not associated with melena, jaundice, or changes in bowel habits. The patient sought medical attention due to difficulty in daily activities such as walking and sleeping for the past two weeks. He had no significant medical or family history.
On examination, the patient had significant ascites with dilated, tortuous veins. Per rectal (PR) examination was normal. Routine laboratory investigations were conducted, revealing largely normal liver function test results, except for an elevated alkaline phosphatase (ALP) level of 143 IU/L (reference range: 40–129 IU/L) and a total protein level of 9.17 g/dL (reference range: 6.7–8.3 g/dL). The patient’s prothrombin time was recorded at 14 s (control: 11.8 s), with an international normalized ratio (INR) of 1.19 (reference range: 0.8–1.1). Serum electrolyte levels and renal function parameters were within normal limits. The elevated ALP (143 IU/L) likely indicates hepatic congestion due to venous outflow obstruction. The high total protein (9.17 g/dL) could be attributed to hemoconcentration or inflammatory response secondary to venous stasis.
Ascitic fluid analysis revealed a slightly turbid deep yellow fluid, with mesothelial cells present. Microscopic analysis showed a total cell count of 1600 cells/cumm, with 85% lymphocytes and 15% neutrophils. The glucose, protein, LDH, adenosine deaminase, and albumin levels were 143 mg/dL, 6.2 g/dL, 144 IU/L, 4.4 IU/L, and 3.59 g/dL, respectively. Upper GI endoscopy revealed small esophageal varices, portal hypertensive gastropathy, portal duodenopathy, and gastric erosions. Ultrasound-guided diagnostic and therapeutic ascitic tapping confirmed a SAAG ratio above 1.1, indicating a transudate nature.
CT imaging revealed a suspicious thin wall in the suprahepatic portion of the IVC, along with focal wall calcification. Surface nodularity with volume redistribution suggested cirrhosis secondary to hepatic venous outflow obstruction. Multiple large pericardial, paraesophageal, splenorenal, retroperitoneal, omental, and abdominal wall collaterals were observed, along with mild ascites and diffuse omental haziness. Normal serum CA-19-9, CEA, and alpha-fetoprotein levels ruled out malignancy. Ascitic fluid adenosine deaminase (ADA) was tested to rule out tuberculosis (TB) peritonitis, given the lymphocyte-predominant ascitic fluid and the regional prevalence of TB. The ADA level of 4.4 IU/L is significantly below the typical TB threshold (>40 IU/L), making TB unlikely. Additional imaging such as Doppler ultrasound and MRI were considered but not performed as CT findings confirmed IVC obstruction with collateral circulation.
The patient underwent IVC venoplasty under fluoroscopic guidance in the interventional radiology department under aseptic precautions. A 14 French transvenous femoral access was obtained, and a venogram demonstrated a partial web in the IVC with slow contrast filling into the right atrium (Figure 1). Venoplasty was performed using 10 and 14 mm angioplasty balloons. The post-procedure venogram demonstrated smooth and rapid filling of the IVC and right atrium, with no residual stenosis or obstruction (Figure 2). His symptoms improved over time, and he was initiated on anticoagulants (acenocoumarol). He remained stable and was later discharged with stable vitals. Monthly ultrasound and INR monitoring were conducted, with dose adjustments to maintain INR within the therapeutic range (2–3). Beta-blockers (propranolol) were prescribed for portal hypertension. Regular follow-ups every 3 months with Doppler ultrasound and INR monitoring were planned to assess IVC patency and anticoagulation efficacy. Portal hypertension and cirrhosis progression will be assessed through liver function tests and endoscopic screening for varices. Long-term anticoagulation will be maintained via dose adjustments based on INR values. Potential complications, such as venoplasty restenosis or anticoagulant-induced bleeding, will be monitored closely. At 3 months post-venoplasty, the patient demonstrated significant clinical improvement, with resolution of abdominal pain, ascites, and lower limb edema. Doppler ultrasound revealed improved IVC flow velocities, reduced collateral circulation, and a patent IVC venoplasty site. Laboratory tests showed normalized liver function tests (LFTs) and improved coagulation profile. At 6 months, CT venography confirmed a patent IVC with no evidence of re-stenosis or occlusion, reduced hepatic vein thrombosis, and improved hepatic perfusion. Clinical assessment revealed sustained symptom improvement and enhanced quality of life, with stable or improved LFTs and normal coagulation profile.
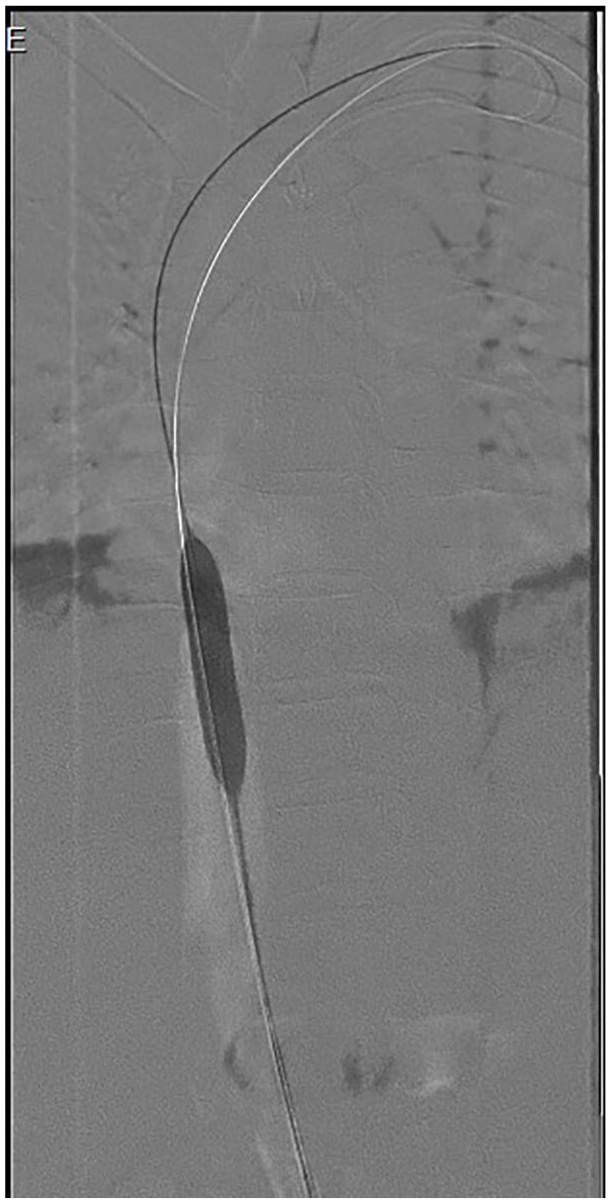
Pre-procedure venogram showing partial web in the IVC with slow contrast filling into the right atrium.

Post-procedure venogram demonstrating successful venoplasty with smooth and fast filling of the IVC and resolution of the partial web.
By 9 months, Doppler ultrasound continued to show patency of the IVC venoplasty site, with stable or improved IVC flow velocities. Clinical assessment demonstrated ongoing symptom improvement, with no evidence of disease progression, and stable LFTs and coagulation profile. At 12 months post-venoplasty, CT venography revealed long-term patency of the IVC, with sustained improvement in hepatic perfusion. Clinical assessment showed continued symptom improvement, enhanced quality of life, and stable or improved LFTs, with a normal coagulation profile. These findings suggest that IVC venoplasty can be an effective treatment for BCS, with significant clinical and radiological improvement at short-term and long-term follow-up.
Discussion
BCS is a rare disorder characterized by hepatic venous outflow obstruction, leading to liver congestion and dysfunction. The prevalence of BCS in the general population is approximately 1 in 100,000. 1 The most common causes of BCS include myeloproliferative disorders, malignancy, hepatic lesions, pregnancy, oral contraceptives, and idiopathic factors. In Asian populations, IVC obstruction is a common cause whereas in non-Asian countries hepatic vein blockage predominates.5,6 The differential diagnosis of BCS includes hepatic malignancy, portal vein thrombosis, TB-related liver disease, and myeloproliferative disorders. In this case, malignancy was ruled out based on normal serum CA-19-9, CEA, and alpha-fetoprotein levels, and TB peritonitis was excluded based on a low ADA level. JAK2 mutation testing was not performed due to financial constraints but remains a consideration in cases with unclear etiology. There are also a few studies that highlight thrombophilia as well as JAK-2 mutations as contributors to BCS. 7 Treatment goals for BCS include reducing obstruction, preventing clot progression, minimizing liver damage, and managing complications. 8
For patients experiencing acute thrombosis in its early phases, standard medical treatment—including diuretics and anticoagulation—has demonstrated favorable outcomes. 9 In this patient, diuretics, beta-blockers (propranolol), ursodeoxycholic acid, and anticoagulants were initially administered. Anticoagulation (acenocoumarol) was initiated to prevent clot propagation. Beta-blockers were prescribed to reduce portal pressure. Diuretics were started for ascites management but were ineffective, necessitating interventional therapy. The choice of anticoagulant was based on availability and patient compliance. However, due to persistent ascites, IVC venoplasty was considered. IVC venoplasty is a radiological intervention that uses a balloon-tipped catheter and X-ray image guidance to restore normal blood flow in a blocked IVC. Venoplasty was chosen over TIPS because the obstruction was localized to the IVC without significant hepatic vein involvement. TIPS is typically preferred in cases with widespread hepatic venous thrombosis. LT was not considered as the patient’s liver function was preserved. The procedure successfully restored venous outflow, reducing symptoms. The study performed by Tripathy et al. showed that IVC with or without hepatic vein venoplasty produced a progressive decline in hepatic congestion as well as an improvement in liver function. 10 Moreover, balloon venoplasty is considered a treatment option that holds a lot of promise in treating BCS in children. 11 Although effective, this procedure carries risks such as bleeding, infection, structural damage, and potential thrombosis. If a patient experiences worsening hepatic function or uncontrolled ascites, LT becomes necessary. Long-term anticoagulation and regular Doppler monitoring are essential for preventing restenosis. The patient will require lifelong INR monitoring and follow-up imaging every 3–6 months. The risk of portal hypertension progression will be managed with continued beta-blocker therapy and periodic endoscopy for variceal surveillance. To our knowledge, this is the first reported case of BCS due to IVC occlusion successfully treated with IVC venoplasty. Table 1 lists the reported cases of BCS and its treatment using different surgical interventions.
Reported cases of Budd-Chiari syndrome and its treatment using different surgical interventions.

BCS: Budd-Chiari syndrome; IVC: inferior vena cava.
Learning points
BCS is a rare condition with various causes, including hypercoagulable states and idiopathic factors.
Standard medical therapy such as diuretics, beta-blockers, and anticoagulation, may not be sufficient enough to manage complications like refractory ascites.
Successful management of BCS often requires a combination of medical, radiological, and surgical interventions tailored to individual patient needs.
Conclusion
This case report highlights the significance of a multidisciplinary approach in managing BCS, especially in patients with uncommon presentations such as IVC obstruction. IVC venoplasty proved to be a safe and effective procedure, leading to substantial symptom relief and disease stabilization. In complex cases of BCS, integrating medical therapy with interventional radiology presents a promising strategy for optimizing patient outcomes.
Footnotes
Acknowledgements
Joel Sabu and Kevin Jose Madapat have contributed equally to the work and should be considered as co-first authors.
Author contributions
J.S. conceptualization, methodology, data analysis, writing—original draft; N.K.B. methodology, literature review, data collection, writing—review & editing; K.J.M. statistical analysis, interpretation of results, writing—review & editing; S.S. project supervision, writing, interpretation of results—final.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for his anonymized information to be published in this article.
