Abstract
Background
Clear cell renal cell carcinoma (ccRCC) is a common urological disease. Expression of the protein tyrosine phosphatase 12 gene (PTPN12) is decreased in many cancers; however, the relationship between PTPN12 gene function and renal cancer remains unclear.
Methods
We detected PTPN12 protein expression in ccRCC and corresponding normal tissues from 64 patients with ccRCC by immunohistochemistry, and relative PTPN12 mRNA levels by real-time quantitative polymerase chain reaction. The relationships between the relative expression levels of PTPN12 mRNA and the patients’ clinical data were analyzed.
Results
PTPN12 protein and mRNA expression levels were significantly lower in ccRCC compared with the corresponding normal tissues. The mRNA expression levels in the ccRCC and corresponding normal tissues from the 64 patients with ccRCC were 0.459±0.445 and 1.001±0.128, respectively, compared with the control (glyceraldehyde 3-phosphate dehydrogenase). There was a significant correlation between relative expression of PTPN12 mRNA in ccRCC tissues and tumor diameter and clinical stage.
Conclusion
The expression levels of PTPN12 protein and mRNA were significantly lower in ccRCC tissues compared with normal tissues. The role of PTPN12 may provide new insights and evidence to aid the diagnosis and targeted therapy of ccRCC.
Keywords
Introduction
Clear cell renal cell carcinoma (ccRCC) is a common form of kidney cancer. According to World Health Organization statistics in 2018, renal cancer had the ninth highest incidence among male malignant tumors, second only to prostate cancer and bladder cancer, with the incidence continuing to increase year by year. 1 It is categorized into various subtypes, with clear cell renal cell carcinoma (ccRCC) representing about 85% of all renal cell carcinoma (RCC) tumors. 2 Some patients with metastatic disease face high morbidity and mortality, and a third of patients present with locally advanced or metastatic disease at initial diagnosis. 3 The overall prognosis of patients with advanced renal cancer has improved significantly in recent years, 4 in line with improvements in kidney cancer therapy, including the emergence of anti-angiogenic drugs such as tyrosine kinase inhibitors and mammalian target of rapamycin (mTOR) inhibitors, and immunotherapy. 5 However, targeted drugs are expensive, and the pathogenesis of renal cancer and the associated drug research has become a hot topic.
Numerous studies found that an absence of tumor suppressor gene expression was closely related to the occurrence of malignant tumors.6–8 Several recent studies have confirmed that protein tyrosine phosphatase 12 (PTPN12) gene expression is reduced in lung cancer, liver cancer, and breast cancer;6–8 however, the relationship between PTPN12 gene function and ccRCC remains unclear.
In this study, we detected PTPN12 protein and mRNA expression levels in tumor tissue and corresponding normal tissue from patients with ccRCC. We also examined the relative expression levels of PTPN12 mRNA in ccRCC tissues in relation to clinical data. This study lays the foundation for further research into the mechanism of the PTPN12 gene in ccRCC.
Materials and methods
Subjects and sample collection
Surgical specimens were collected from patients aged 18 to 80 years diagnosed with ccRCC who underwent surgery for RCC at Hebei Medical University from 2014 to 2020. The exclusion criteria were age < 18 or > 80 years, patients with poor cardiac, pulmonary, or liver and kidney function who could not tolerate surgery, patients requiring emergency surgery, and patients with preoperative surgical site infection. All the tissue specimens were confirmed as ccRCC or normal tissues by hematoxylin and eosin (HE) staining. Tissues classified as normal showed no tumor infiltration. One part of each specimen was fixed with formalin, dehydrated, and embedded in paraffin, and the other part was stored in RNA protection solution at −80°C for RNA extraction. The ccRCC stage was evaluated based on computerized tomography reports.
This study was approved by the ethics committee of Hebei Medical University. Written informed consent was obtained from the patients.
Hematoxylin and eosin staining
The sections were placed in an electric oven at 60°C for 1 hour to soften the wax (Tianjin Zhiyuan Chemical Reagent Co. Ltd., Tian, China). After cooling, the sections were successively soaked in xylene and ethanol, followed by hematoxylin stain for 2 to 4 minutes and three washes in water. The sections were then soaked for 10 to 30 s in hydrochloric acid alcohol (until pink), washed three times, soaked in distilled water for 15 minutes, then washed three times in water. Finally, they were soaked in eosin solution for 1 to 3 minutes, 70% ethanol for 1 minute, 80% ethanol for 3 minutes, 95% ethanol for 5 minutes, anhydrous ethanol I for 5 to 10 minutes, and anhydrous ethanol II for 5 to 10 minutes. After drying, the sections were sealed with neutral resin and examined under a light microscope.
Immunohistochemistry
The sections were placed in xylene followed by 95% and 80% ethanol, rinsed with tap water three times, soaked in phosphate-buffered saline (PBS) twice for 5 minutes each, followed by soaking in methanol hydrogen peroxide for 20 minutes. The slides were then wiped and placed in a wet box, and the appropriate concentration of PTPN12 monoclonal antibody (1:3000 dilution; Sigma, St. Louis, MO, USA) was added to the tissue drip until it fully covered the tissue. The slides were then placed in the refrigerator at 4°C overnight. Immunochromogenic agent (50 µL; Beijing Zhongshan Jinqiao Co. Ltd., Beijing, China) was added to cover the tissue, followed by incubation at room temperature for 25 minutes, sealing with neutral resin, and observation under a light microscope.
Detection of relative PTPN12 mRNA expression levels by real-time quantitative PCR (qPCR)
Total RNA was extracted from ccRCC and normal renal tissues using an RNAiso Plus (TRIzol) kit (Thermo Fisher, MA, USA), and reverse transcribed to cDNA. RT-qPCR was performed using a Light Cycler® 4800 System (Beijing Zhongshan Jinqiao Co. Ltd.) with specific primers for PTPN12, according to the manufacturer’s instructions (Table 1). The mRNA expression levels in the tumor and corresponding normal tissues were detected by real-time fluorescence quantitative PCR and presented as levels relative to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as an endogenous control. Total RNA (1 µg) was used to generate the first-strand cDNA using random primers and SuperScript II reverse transcriptase (Invitrogen Corp., Carlsbad, CA, USA). PCR was performed in triplicate using a SYBR PrimeScript RT-PCR Kit (Takara, Dalian, China). The expression of GAPDH was measured as an internal control. Thermocycler conditions included an initial hold at 50°C for 2 minutes and then 95°C for 10 minutes, followed by a two-step PCR program of 95°C for 15 s and 60°C for 60 s repeated for 40 cycles. Data were collected and quantitatively analyzed using an Mx4800 system (Applied Biosystems, Foster City, CA, USA). The relative expression levels of the genes via PCR assay (F) were calculated as F = 2–[(average CT value of tested genes − average CT value of housekeeping gene) − (average CT value of control genes − average CT value of housekeeping gene)]. ΔΔCT > 1 indicated high expression of the target gene in ccRCC.
Primer sequences for polymerase chain reaction.

Statistical analysis
The data were analyzed using SPSS Statistics for Windows, version 21.0 (SPSS Inc., Chicago, IL, USA), and figures were drawn using GraphPad Prism 7 (GraphPad Software Inc., La Jolla, CA, USA). Relationships between PTPN12 expression levels in ccRCC and normal tissues were analyzed by χ2 or corrected χ2 tests. Differences between the relative expression levels of PTPN12 mRNA in ccRCC and corresponding normal tissues and the relationship with clinicopathological data were analyzed by paired samples t-tests. P<0.05 indicated a significant difference.
Results
Patients
Tumor and corresponding normal tissue samples were collected from 64 patients (mean age 54 years; 53 male, 11 female) who underwent surgery at Hebei Medical University from 2014 to 2020.
Hematoxylin and eosin staining results
HE staining showed normal glomerular structure in the normal renal tissues (Figure 1a, b) and altered morphology, characterized by hemorrhage, necrosis, cystic degeneration, and calcification in the ccRCC tissues (Figure 1c, d).

Hematoxylin–eosin staining of renal tissues (×200). (a, b) Normal renal tissues and (c, d) corresponding clear cell renal cell carcinoma tissues.
Immunohistochemistry
PTPN12 expression levels were significantly higher in normal renal tissues (Figure 2a, b) compared with ccRCC tissues (Figure 2c, d) (P<0.001). PTPN12 expression was significantly related to ccRCC, based on Pearson’s χ2 test (P<0.001) (Table 2).

Immunostaining of PTPN12 protein in normal renal tissues and corresponding clear cell renal cell carcinoma (ccRCC) tissues. (a, b) High expression of PTPN12 in normal tissues, and (c, d) low expression in corresponding ccRCC tissues. ×200.
PTPN12 protein expression levels in clear cell renal cell carcinoma and corresponding normal tissues.
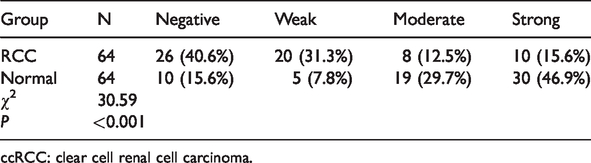
ccRCC: clear cell renal cell carcinoma.
PTPN12 mRNA expression level
The relative PTPN12 mRNA expression levels in the ccRCC and corresponding normal tissues were 0.459±0.445 and 1.001±0.128, respectively. The relative PTPN12 mRNA expression levels were significantly lower in the ccRCC compared with the corresponding normal tissues (P = 0.002) (Figure 3).

PTPN12 mRNA expression levels in ccRCC and corresponding normal tissues. RCC, renal cell carcinoma. *P<0.05.
Correlation between relative PTPN12 mRNA expression levels and clinicopathological data in patients with ccRCC
PTPN12 relative mRNA expression levels were higher in patients aged ≤60 years compared with those aged >60 years, and higher in men than in women, but the differences were not significant. Relative expression levels were significantly higher in patients with a tumor diameter ≤7 cm compared with >7 cm (P = 0.012), and expression was significantly higher in patients with clinical stage I or II compared with clinical stage III or IV (P = 0.047) (Table 3).
Relationships between PTPN12 relative mRNA expression levels in clear cell renal cell carcinoma tissues and clinical data.
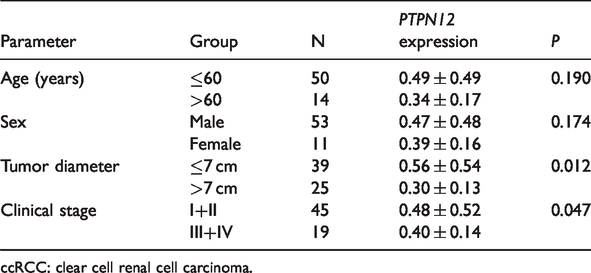
ccRCC: clear cell renal cell carcinoma.
Discussion
Advances in diagnostic techniques mean that patients with renal cancer receive earlier treatment, resulting in an overall 5-year survival rate of 74%. However, 12% of patients still develop advanced metastasis, 9 and the search for markers to aid the diagnosis and early treatment of renal cancer has become a hot research topic.
Sun et al. found that PTPN12 protein was inactivated in triple-negative breast cancer, and that restoration of PTPN12 protein activity effectively inhibited tumorigenicity and metastasis of breast cancer cells, thereby identifying PTPN12 protein as a tumor suppressor for triple-negative breast cancer, and the PTPN12 gene as a candidate tumor suppressor gene for breast cancer. 10 Numerous studies have confirmed that PTPN12 is down-regulated in many cancers, including lung cancer, esophageal squamous cell carcinoma, breast cancer, and liver cancer,6–8,11–13 and the role of PTPN12 in the prognosis and treatment of related tumors have therefore been studied. Liang et al. 14 found that the microRNA miR-194 enhanced ovarian cancer development and tumor metastasis by negatively regulating PTPN12, and was thus a potential target of drug-development efforts to restore PTPN12 activity. Luo et al. 7 found that decreased expression of PTPN12 was not only closely related to the occurrence of liver cancer, but was also significantly related to the recurrence and recurrence-free survival rates in patients with liver cancer. Cao et al. 6 analyzed the expression of PTPN12 using immunohistochemistry and western blotting and found that high PTPN12 expression levels were associated with a favorable survival duration in patients with non-small cell lung cancer (NSCLC), suggesting that PTPN12 expression may be a valuable prognostic biomarker for NSCLC. Xu et al. 15 found that reactive oxygen species-induced oxidation inactivated PTPN12 in papillary renal cell carcinoma, decreasing dephosphorylation of ABL1 and resulting in increased tumor growth and survival. These studies represent the translation of PTPN12 gene research from the experimental level into clinical treatment. However, little is known about the role and expression of PTPN12 in ccRCC, and the effects of PTPN12 on the occurrence and prognosis of this cancer require further confirmation.
In this study, we examined the expression levels of PTPN12 protein in normal and ccRCC tissues qualitatively by immunohistochemistry, and found that the relative expression of PTPN12 protein was reduced in ccRCC compared with normal kidney tissues. These results suggest that a lack of PTPN12 protein may lead to the development of ccRCC. Piao and Jin 16 found that the phosphoinositide-3 kinase/mTOR pathway was negatively regulated by PTPN12, and played an important role in PTPN12-induced antiproliferation in kidney cells. This may be the molecular mechanism by which low PTPN12 expression could promote ccRCC. In addition, we also showed that PTPN12 mRNA expression levels were not significantly correlated with age and sex, but were correlated with the size of the primary tumor and with clinical stage.
Further studies should include expanding the sample size, improving the clinical follow-up data, and examining the effect of PTPN12 gene deletion on the clinical progress and prognosis of renal cancer. Cell experiments should also be carried out to examine the effects of drug-induced activation of the PTPN12 gene in renal cancer cell lines.
In conclusion, PTPN12 protein and mRNA expression levels were significantly reduced in ccRCC tissues compared with normal tissues. PTPN12 may thus provide insights and novel ideas for improving the diagnosis and targeted therapy of ccRCC.
Supplemental Material
sj-pdf-1-imr-10.1177_0300060520936041 - Supplemental material for Expression and clinical significance of PTPN12 in clear cell renal cell carcinoma
Supplemental material, sj-pdf-1-imr-10.1177_0300060520936041 for Expression and clinical significance of PTPN12 in clear cell renal cell carcinoma by Yi Jin, Tian-xi Wang, Hao Li, Peng Guo and Qing-qing Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_0300060520936041 - Supplemental material for Expression and clinical significance of PTPN12 in clear cell renal cell carcinoma
Supplemental material, sj-pdf-2-imr-10.1177_0300060520936041 for Expression and clinical significance of PTPN12 in clear cell renal cell carcinoma by Yi Jin, Tian-xi Wang, Hao Li, Peng Guo and Qing-qing Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_0300060520936041 - Supplemental material for Expression and clinical significance of PTPN12 in clear cell renal cell carcinoma
Supplemental material, sj-pdf-3-imr-10.1177_0300060520936041 for Expression and clinical significance of PTPN12 in clear cell renal cell carcinoma by Yi Jin, Tian-xi Wang, Hao Li, Peng Guo and Qing-qing Wang in Journal of International Medical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
