Abstract
Objectives
This study aimed to investigate the involvement of MCM3AP antisense RNA 1 (MCM3AP-AS1) in chronic obstructive pulmonary disease (COPD).
Methods
The expression levels of plasma MCM3AP-AS1 in COPD patients and healthy controls were measured by quantitative PCR before treatment and at 3 months after the initiation of treatment (post-treatment) from COPD patients. The role of MCM3AP-AS1 in regulating the proliferation of human bronchial smooth muscle cells (HBSMCs) was explored by a cell proliferation assay.
Results
We found that MCM3AP-AS1 expression was downregulated in the plasma of COPD patients compared with controls. Among controls, MCM3AP-AS1 expression was lower in smokers than never-smokers. A 3-year follow-up study showed that, among smokers, patients with low MCM3AP-AS1 expression showed a higher incidence of COPD. After treatment for COPD, MCM3AP-AS1 expression significantly increased. The cell proliferation assay showed that MCM3AP-AS1 overexpression decreased the proliferation rate of HBSMCs. MCM3AP-AS1 silencing had the opposite effect.
Conclusions
MCM3AP-AS1 appears to be downregulated in COPD and to predict its occurrence. MCM3AP-AS1 regulates the proliferation of HBSMCs to participate in airway remodeling.
Keywords
Introduction
As a type of obstructive lung disease, chronic obstructive pulmonary disease (COPD) is characterized by poor airflow and consistent breathing problems. 1 COPD is a progressive disease that causes a cough with sputum and shortness of breath, and it is also the main cause of chronic respiratory failure leading to hospital admission. 2 At present, there is no cure for COPD and current treatment approaches mainly focus on the relief of symptoms. 3 Therefore, early diagnosis is crucially important. 4 Although COPD also affects never-smokers, 5 smoking is still the major risk factor for this disease. 6 However, stopping smoking is a major intervention and not practical in many cases. Therefore, the development of novel predictive markers is warranted.
Smoking itself is not sufficient for the development of COPD and molecular players also have critical roles. 7 In addition to protein-coding genes, the development of COPD is accompanied by changes in the expression patterns of a large number of long (>200 nt) non-coding RNAs (lncRNAs). 8 Some differentially expressed lncRNAs have important functions in COPD and potential clinical values.9, 10 However, the roles of most lncRNAs in COPD are still unknown. MCM3AP antisense RNA 1 (MCM3AP-AS1) is a recently characterized cancer-related lncRNA involved in several types of cancer, such as liver cancer, glioma, and lung cancer.11–13 In lung cancer, MCM3AP‐AS1 overexpression mediated by YY1 targets the microRNA (miR)‐340‐5p/KPNA4 axis to promote cell proliferation and angiogenesis. 13 Our RNA-seq analysis revealed the downregulation of MCM3AP-AS1 in COPD tissues (data not shown), indicating the involvement of this lncRNA in COPD. The present study was therefore carried out to investigate the potential roles of MCM3AP-AS1 in COPD.
Materials and methods
Research subjects
This study included 60 COPD patients (all smokers; 49 men and 11 women; age range, 36–73 years; mean age, 55.4 ± 5.9 years old), 60 healthy controls (non-smokers; 49 men and 11 women; age range, 36–73 years; mean age, 55.2 ± 5.6 years old), and 120 smokers without COPD (100 men and 20 women; age range, 37–70 years; mean age, 54.1 ± 5.0 years old) who were admitted to our hospital between July 2014 and July 2016. All COPD patients were diagnosed for the first time and no other severe clinical disorders were observed. All 60 healthy controls received systemic physiological examinations and all parameters were within normal ranges. The 120 smokers had a normal lung function at the time of admission and a history of smoking for 10.6–17.7 years. This study was approved by the Ethics Committee of Jiading Central Hospital, Shanghai University of Medicine & Health Sciences. All participants provided their written informed consent.
Plasma, treatment, and follow-up
All participants were subjected to blood extraction (5 ml) from the elbow vein under fasting conditions. The 60 COPD patients were treated with bronchodilators and glucocorticosteroids through an inhaler to widen the airway and reduce inflammation. These patients also provided blood (5 ml) at 3 months after the initiation of treatment. All blood samples were centrifuged at 1,200 × g at room temperature for 10 minutes for plasma preparation. All 120 smokers were followed-up for 3 years after admission and the occurrence of COPD was recorded.
Cells and transfection
Human bronchial smooth muscle cells (HBSMCs; PromoCell, Heidelberg, Germany) were cultured in smooth muscle cell medium (PromoCell) at 37°C with 95% humidity and 5% CO2 until 80% confluent. An MCM3AP-AS1 expression vector was constructed using the pcDNA3.1 vector (Invitrogen Corp., Carlsbad, CA, USA) as a backbone. MCM3AP-AS1 small interfering (si)RNA and negative control (NC) siRNA were synthesized by Invitrogen Corp. HBSMCs (106) were transiently transfected with 10 nM vector or 45 nM siRNA by Lipofectamine 2000 (Invitrogen Corp.). Untransfected cells were used as controls (C). NC cells were transfected with NC siRNA or empty vector. The following experiments were performed at 48 hours post-transfection.
RNA preparation
TRIzol reagent (Tiangen, Beijing, China) was used to extract total RNA from plasma and HBSMCs. All RNA samples were treated with gDNA eraser (Takara Biotechnology (Dalian) Co. Ltd., Dalian, China) at 37°C for 2 hours to remove genomic DNA. RNA concentrations were measured using a NanoDrop™ 2000 Spectrophotometer (Thermo Fisher Scientific Inc., Rockford, IL, USA).
RT-qPCR
Reverse transcriptions were performed to synthesize cDNA from total RNA using the QuantiTect Reverse Transcription Kit (Qiagen Inc., Valencia, CA, USA). Quantitative (q)PCR reactions were performed on the ABI Prism 5700 Sequence Detection System (Applied Biosystems, Foster City, CA, USA) using SYBR® Premix Ex Taq™ II (Takara Biotechnology (Dalian) Co. Ltd.). qPCR conditions were: 10 minutes at 95°C then 40 cycles of 10s at 95°C, 30s at 58°C, and 30s at 72°C. Primers were: forward, 5′-GCTGCTAATGGCAACACTGA-3′ and reverse, 5′-AGGTGCTGTCTGGTGGAGAT-3′. Three replicates were included in each experiment and fold-changes of gene expression levels were calculated using the 2−ΔΔCt method.
CCK-8 assay
HBSMC cells were harvested 48 hours post-transfection and cell proliferation rates were measured using the CCK-8 assay (Sigma-Aldrich, St Louis, MO, USA). HBSMCs were seeded into each well of a 96-well cell culture plate (5000 cells per 0.1 mL of medium), cultivated as described and collected every 24 hours until 96 hours. At 4 hours before cell collection, CCK-8 solution was added to each well to reach a final concentration of 10%. Cell proliferation rates were calculated using optical density values measured at 450 nm.
Enzyme-linked immunosorbent assay (ELISA) analysis
Cytokine concentrations from the supernatants of human pulmonary microvascular endothelial cells (HPMECs) after appropriate treatment were measured using commercial interleukin (IL)-6, IL-8, and tumor necrosis factor (TNF)-α ELISA kits (R&D Systems, Minneapolis, MN, USA) following the manufacturer’s instructions.
Statistical analysis
Mean values of three biological replicates were analyzed by SPSS 17.0 software (SPSS Inc., Chicago, IL, USA). The paired t test was used to compare two time points within the same group. Comparisons among multiple groups were analyzed by one-way analysis of variance combined with Tukey’s test. Using the median plasma expression level of MCM3AP-AS1 as a cutoff value (2.335), the 120 COPD patients were divided into high- and low-expressing MCM3AP-AS1 groups (n = 30 per groups). COPD-free curves were plotted and compared by the log-rank test. The chi-squared test was used to explore the relationship between MCM3AP-AS1 expression levels and patient clinical data. P < 0.05 was considered statistically significant.
Results
MCM3AP-AS1 expression was downregulated in COPD
MCM3AP-AS1 expression levels in COPD patients (n = 60), healthy controls (n = 60), and smokers (n = 120) were measured by qPCR. Compared with the control group, the expression levels of MCM3AP-AS1 in plasma were significantly reduced in the COPD and smoker groups (Figure 1, p < 0.05). MCM3AP-AS1 expression levels were also significantly lower in the COPD group than the smoker group (p < 0.05). Chi-squared test analysis showed that MCM3AP-AS1 expression was not significantly correlated with patient age, gender, complications (hypertension, diabetes, ischemic heart disease, and renal dysfunction), or alcohol consumption (Table 1).

The expression of MCM3AP-AS1 was downregulated in COPD.
Chi-squared test analysis of the relationship between the expression levels of MCM3AP-AS1 and patients’ clinical data.

COPD treatment increased MCM3AP-AS1 expression levels
MCM3AP-AS1 expression levels in plasma collected from COPD patients 3 months after the initiation of treatment (post-treatment) were significantly increased compared with pre-treatment levels according to paired t test analysis (Figure 2, p < 0.05).

COPD treatment increased MCM3AP-AS1 expression levels.
Low MCM3AP-AS1 expression levels predicted a high incidence rate of COPD
COPD-free curves were plotted for both high- and low-expression MCM3AP-AS1 level groups. Patients in the low-expression MCM3AP-AS1 group experienced a significantly higher incidence rate of COPD than those in the high-expression group (Figure 3, p < 0.05).
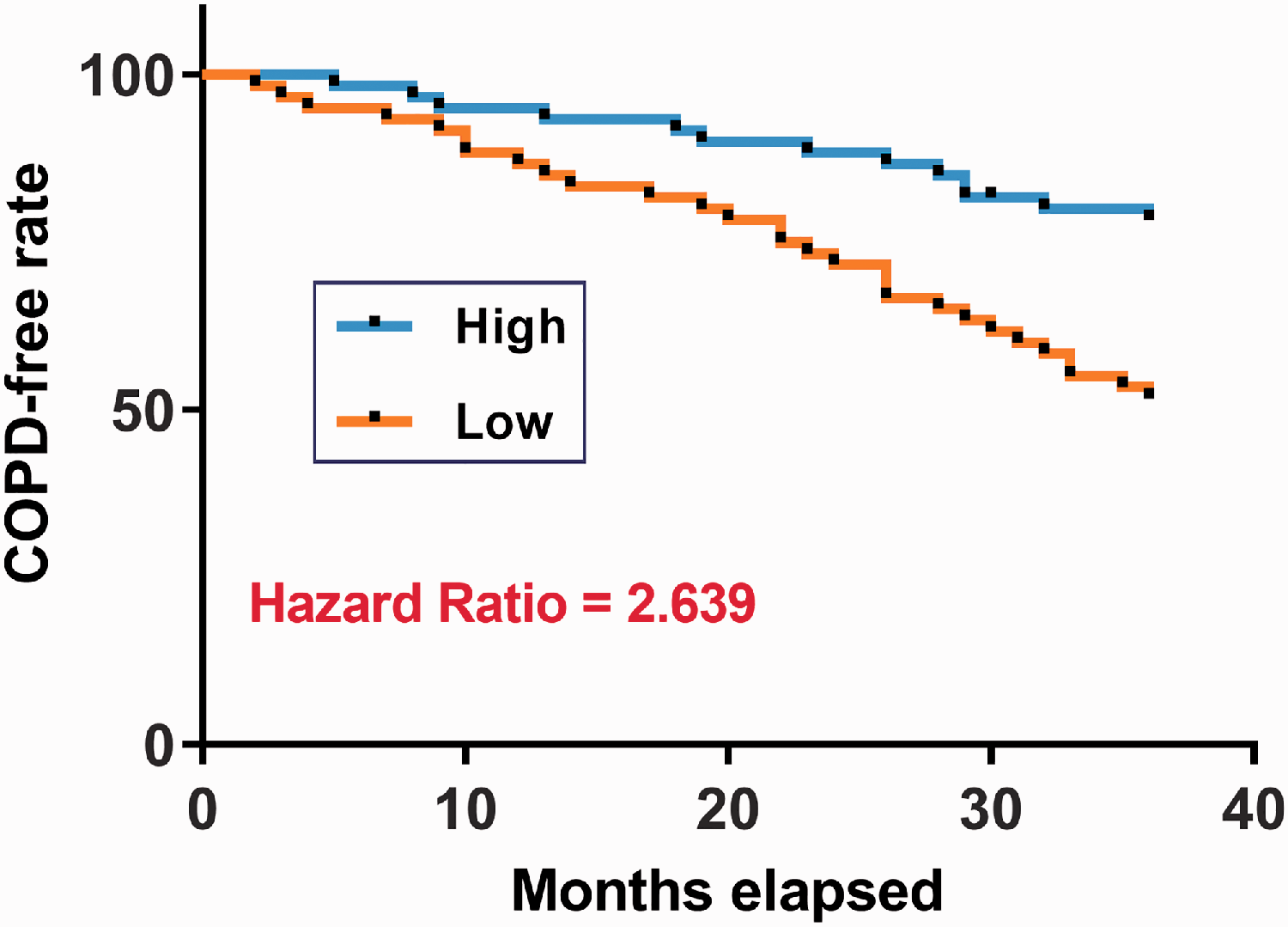
Low MCM3AP-AS1 expression levels predicted a high incidence rate of COPD.
MCM3AP-AS1 negatively regulated the proliferation of HBSMCs
HBSMCs were transfected with a MCM3AP-AS1 expression vector or MCM3AP-AS1 siRNA. Compared with C and NC groups, MCM3AP-AS1 expression levels were significantly altered after transfection (Figure 4a, p < 0.05), indicating their success. The CCK-8 assay showed that MCM3AP-AS1 overexpression significantly decreased the cell proliferation rate, while MCM3AP-AS1 silencing had the opposite effect compared with controls (Figure 4b, p < 0.05). Additionally, MCM3AP-AS1 knockdown significantly increased inflammation in HPMECs compared with controls (Figure 4c, p < 0.05).

MCM3AP-AS1 negatively regulated the proliferation of HBSMCs.
Discussion
In this study, we investigated the involvement of MCM3AP-AS1 in COPD. We found that MCM3AP-AS1 was downregulated in COPD and was predictive of disease. Additionally, it appeared to regulate the proliferation of HBSMCs.
The functionality of MCM3AP-AS1 has only been investigated in cancer biology.11, 12 MCM3AP-AS1 is upregulated in liver cancer and regulates the miR-194-5p/FOXA1 axis to promote tumor growth. 11 It is also upregulated in glioblastoma and its overexpression interacts with the miR-211/KLF5/AGGF1 axis to regulate angiogenesis. 12 However, the roles of MCM3AP-AS1 in other types of disease remain unclear. Our study is the first to report its downregulation of in COPD, and suggests that MCM3AP-AS1 has varying expression patterns in different types of human disease.
Smoking is a major risk factor for COPD,14, 15 and more than 20% of smokers older than 45 years have been reported to suffer from COPD. The higher incidence rate of COPD observed in this study likely reflects the long patient history of smoking (>10 years). Interestingly, we observed that MCM3AP-AS1 was downregulated in patients with COPD, and further downregulated in smokers with COPD. This indicates that smoking induces the development of COPD, which further downregulates the expression of MCM3AP-AS1. Our study also showed that low MCM3AP-AS1 expression levels predicted a high incidence rate of COPD among smokers. Therefore, measuring the expression of plasma MCM3AP-AS1 may help the screening of smokers with a high risk of COPD. However, the accuracy of this remains to be validated.
The development of COPD is accompanied by airway remodeling16, 17 in which the abnormal proliferation of HBSMCs plays critical roles. 18 We found that MCM3AP-AS1 negatively regulated the proliferation of HBSMCs, indicating that the control of MCM3AP-AS1 expression could be used to treat COPD. However, the mechanism of MCM3AP-AS1 remains to be further explored.
The annexin II (ANXA2) pathway was previously reported to play a critical role in the proliferation of HBSMCs, 19 while follistatin-related protein (FSTL)-1 is crucial to lung remodeling in COPD. 20 Our future studies will therefore aim to explore the interactions between MCM3AP-AS1 and the ANXA2 pathway or FSTL-1. This study is limited by the lack of in vivo experiments and its small sample size. We plan to confirm our findings in studies involving more patients and using animal models.
In conclusion, MCM3AP-AS1 is downregulated in COPD and is predictive of disease. It appears to regulate the proliferation of HBSMCs in airway remodeling in COPD.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by grants from the Shanghai Municipal Jiading District Natural Science Foundation (JDKW-2019-W03), and Shanghai Jiading District Health System Research Project (discipline):(2017ZD05).
