Abstract
Objective:
To assess the effect of cardioselective b-blockers on pulmonary function in patients with chronic obstructive pulmonary disease (COPD).
Methods:
The Embase and MEDLINE™ databases and the Cochrane Controlled Trials Register were searched comprehensively to identify all relevant clinical trials in humans published between January 1966 and May 2011. Randomized, blinded, placebo-controlled trials that studied the effects of cardioselective ß-blockers on the forced expiratory volume in 1 s (FEV1) and the responsiveness of the FEV1 to ß-agonist administration in patients with COPD were included in the analysis.
Results:
Five studies were identified: four of these assessed both cardioselective and nonselective ß-blockers; one assessed only cardioselective ß-blockers. The FEV1 was shown to decrease by 0.14 l with nonselective ß-blockers and by 0.03 l with cardioselective ß-blockers. The responsive -ness of the FEV1 to ß-agonist administration decreased significantly by 13.42% after nonselective ß-blockers, but did not change significantly after cardioselective -blocker administration.
Conclusion:
This meta-analysis suggests that the use of β-blockers, especially cardioselective β-blockers, should not be contraindicated in patients with COPD.
Introduction
The use of β-blockers is indicated in the management of angina pectoris, myocardial infarction, hypertension and congestive heart failure.1 – 5 Despite the proven benefits of β-blockers in treating hypertension, ischaemic heart disease and heart failure, many clinicians are often hesitant to prescribe them in the presence of a variety of common conditions for fear of adverse reactions.6,7 Cardiovascular disease and chronic obstructive pulmonary disease (COPD) often coexist because of the heightened risk of smoking-induced atherosclerosis in patients with COPD. 8 However, many review articles and practice guidelines list asthma and COPD as contraindications to β-blocker administration, citing cases of acute bronchospasm occurring during nonselective β-blocker use.5,9– 11 Cardioselective β-blockers have over 20 times more affinity for β1-receptors than for β2-receptors and theoretically should be associated with a considerably lower risk of bronchoconstriction. 12
Meta-analyses have shown that cardioselective β-blockers do not produce clinically significant adverse respiratory effects in patients with mild to moderate reversible airway disease, 13 or produce a significant reduction in airway function, or increase the risk of exacerbation in COPD. 14 The objective of the present meta-analysis was to evaluate the effect of cardioselective β-blockers on pulmonary function in patients with COPD, especially airway responsiveness to β-agonist administration.
Methods
Search Strategy
A literature search was performed to identify clinical trials that addressed the effects of cardioselective β-blockers on pulmonary function in patients with COPD. The Embase and MEDLINE™ databases and the Cochrane Controlled Trials Register were searched comprehensively, to identify all relevant clinical trials in humans published between January 1966 and May 2011. Terms used in the search were ‘wheeze', ‘dyspnoea', ‘COPD', ‘chronic airway obstruction', ‘chronic bronchitis', ‘obstructive lung disease', ‘obstructive airway disease', ‘β-blocker', ‘adrenergic antagonist', ‘sympatholytic' and ‘adrenergic receptor block'. Trials were limited to those published in English. The database search was further augmented by scanning the references of identified articles or reviews and clinical symposia abstracts.
Study Selection
Trials were included if: (i) they were randomized, placebo-controlled and single-or double-blinded; (ii) they reported forced expiratory volume in 1 s (FEV1) values at rest and the responsiveness of FEV1 values to β-agonist administration by inhalation for both the study drug and placebo; (iii) they included only subjects with COPD. Crossover trials were considered to be randomized if the different interventions were administered in random order.
Assessment of Validity
The methodological quality of each trial was assessed using the Jadad scale. 15 The main intervention studied was the use of intravenous or oral cardioselective β-blockers versus placebo, given either as a single dose or for an extended period. A second intervention studied was the administration of a β-agonist by inhalation. Funnel plots of effect size versus standard error were performed for each trial to detect reporting bias; in the absence of bias the plot should approximately resemble an inverted funnel.
Data Extraction and Synthesis
Outcomes measured were the mean FEV1 value measured after the study drug or placebo and the change in FEV1 in response to administration of a β-agonist. To estimate the net treatment effect, the mean ± SD FEV1 value after administration of the study drug was compared with the response after placebo. For those trials that did not provide standard deviation information for the study results, the mean standard deviation was calculated using methodology described in trials that did provide such data.
The mean treatment effects were pooled to get a weighted average of the study means with a fixed-effects model for continuous outcomes using Review Manager (RevMan) software version 4.2 (The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen, Denmark). The weighted averages of treatment effects were compared using the two-tailed Mann–Whitney U-test; differences were considered to be significant when the P-value was < 0.05. In addition, 95% confidence intervals (CI) were obtained for the pooled study means. In order to test for heterogeneity between studies, the χ2-value was calculated for the assumption of homogeneity.
In order to evaluate the response to treatment with a β-agonist, the baseline used was the FEV1 after administration of the study drug or placebo but before administration of the β-agonist. The net treatment effect was estimated by calculating the percentage change in FEV1 measured after β-agonist administration compared with the baseline, and was compared in the placebo and study drug groups.
A subgroup analysis was performed to evaluate the treatment response of participants known to have comorbid cardiovascular conditions such as hypertension or chronic heart failure.
Results
Search Results
The database search identified 310 potentially relevant articles. After reviewing the articles and their bibliographies, 27 trials of β-blockers in patients with COPD were found. Of these, five met the inclusion criteria: four studies assessed both cardio -selective and nonselective β-blockers;16 – 19 one study assessed only cardioselective β-blockers. 20 The cardioselective β-blockers included in the studies were atenolol, metoprolol, bisoprolol and celiprolol. Characteristics of the five studies identified are given in Table 1.
Characteristics of trials studying forced expiratory volume in 1 s (FEV1) values at rest and the responsiveness of FEV1 values to β-agonist administration after β-blocker or placebo treatment in chronic obstructive pulmonary disease (COPD)

CHF, congestive heart failure; FEV1, forced expiratory volume In 1 s; FVC, forced vital capacity; PC20, concentration of β-agonist required to decrease FEV1 by 20% from baseline; SRAW, specific airways resistance.
Of the 22 trials that were excluded, nine did not study the responsiveness of FEV1 to β-agonist administration, three were not randomized, three were not blinded, one did not provide placebo controls and six were reviews of other trials.
Methodological Quality of Included Studies
Four of the studies evaluated were small crossover trials that included a washout period.16 – 19 One was a standard randomized controlled trial. 20 All of the trials identified were double-blinded. One trial did not provide standard deviation information for the FEV1 treatment effect. 19 Funnel plots of effect size versus standard error for each trial showed no evidence of bias.
Quantitative Data Synthesis
Nonselective β-blockers
The four studies that included nonselective β-blockers16 – 19 had a total of 46 participants, giving a mean of 11.5 participants per study. The mean ± SD age of the participants was 63.1 ± 6.5 years and the baseline mean ± SD FEV1 value was 1.83 ± 0.36 l. Baseline characteristics were the same for the placebo and treatment groups because the trials were crossover by design.
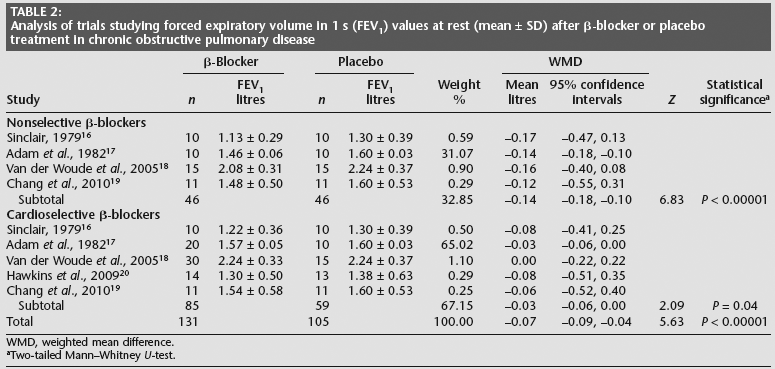
After treatment with nonselective β-blockers, the FEV1 showed a significant decrease of 0.14 l (P < 0.00001) (Table 2). The responsiveness of the FEV1 to β-agonist administration also decreased significantly, by 13.42% (P < 0.00001) (Table 3).
Analysis of trials studying forced expiratory volume in 1 s (FEV1) values at rest (mean ± SD) after β-blocker or placebo treatment in chronic obstructive pulmonary disease
WMD, weighted mean difference.
Two-tailed Mann-Whitney U-test.
Analysis of trials studying forced expiratory volume in 1 s (FEV1) values in chronic obstructive pulmonary disease, showing the percentage change in FEV1 from baseline after β-blocker or placebo treatment (mean ± SD) following β-agonist administration

WMD, weighted mean difference; NS, no statistically significant between group differences (P ≥ 0.05).
Two-tailed Mann-Whitney U-test.
Cardioselective β-blockers
The five studies that included cardioselective β-blockers16– 20 had a total of 85 participants, giving a mean of 17 participants per study. The mean ± SD age of the participants was 65.4 ± 6.8 years in the treatment group and 64.3 ± 7.6 years in the placebo group. The baseline mean ± SD FEV1 value was 1.73 ± 0.60 l in the treatment group and 1.72 ± 0.60 l in the placebo group.
After treatment with cardioselective β-blockers, the FEV1 showed a significant decrease of 0.03 l (P = 0.04) (Table 2). However, cardioselective β-blockers did not significantly change the responsiveness of FEV1 to β-agonist administration (Table 3).
Subgroup analyses
To evaluate the effect of treatment in patients with comorbid cardiovascular conditions, a subgroup analysis was performed (Tables 4 and 5). Two of the trials included patients with COPD and hypertension or chronic heart failure.17,20 When only these trials were included in the analysis, there was a similar decline in FEV1 after treatment with cardioselective β-blockers (0.03 l) and no significant change in the responsiveness of the FEV1 to β-agonist administration.
Subgroup analysis of trials studying forced expiratory volume in 1 s (FEV1) values at rest (mean ± SD) after β-blocker or placebo treatment in patients with chronic obstructive pulmonary disease and comorbid conditions

WMD, weighted mean difference.
Two-tailed Mann-Whitney U-test.
Subgroup analysis of trials studying forced expiratory volume in 1 s (FEV1) values in patients with chronic obstructive pulmonary disease and comorbid conditions, showing the percentage change in FEV1 from baseline after β-blocker or placebo treatment (mean ± SD) following β-agonist administration

WMD, weighted mean difference; NS, no statistically significant between group differences (P ≥ 0.05).
Two-tailed Mann-Whitney U-test.
Sensitivity analyses
A sensitivity analysis was performed to evaluate the effect of excluding the one trial that did not provide standard deviation information 19 (Tables 6 and 7). Excluding this trial did not significantly affect the change in FEV1 values or the responsiveness of the FEV1 to β-agonist administration for nonselective or cardioselective β-blockers.
Sensitivity analysis of trials studying forced expiratory volume in 1 s (FEV1) values at rest (mean ± SD) after β-blocker or placebo treatment in chronic obstructive pulmonary disease patients, excluding trials without standard deviation information

WMD, weighted mean difference.
Two-tailed Mann-Whitney U-test.
Sensitivity analysis of trials studying forced expiratory volume in 1 s (FEV1) values in chronic obstructive pulmonary disease patients, excluding trials without standard deviation information, showing the percentage change in FEV1 from baseline after β-blocker or placebo treatment (mean ± SD) following β-agonist administration

WMD, weighted mean difference; NS, no statistically significant between group differences (P ≥ 0.05).
Two-tailed Mann-Whitney U-test.
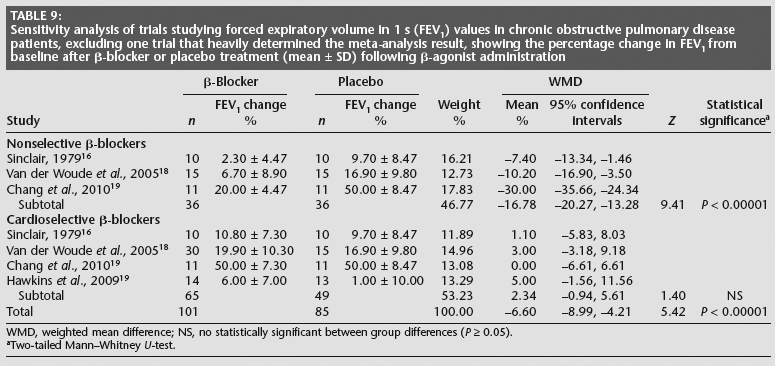
A sensitivity analysis was also performed to evaluate the effect of excluding one trial that heavily determined the meta-analysis result 17 (Tables 8 and 9). The decline in FEV1 after β-blocker administration became nonsignificant when excluding this trial. However, the difference in the FEV1 values when including or excluding this trial was small (–0.14 l and –0.16 l, respectively, for nonselective β-blockers; –0.03 l and –0.04 l, respectively, for cardioselective β-blockers). Interestingly, the change in the responsiveness of the FEV1 to β-agonist administration was in the opposite direction when excluding this trial, but this change was still not statistically significant.
Sensitivity analysis of trials studying forced expiratory volume in 1 s (FEV1) values at rest (mean ± SD) after β-blocker or placebo in chronic obstructive pulmonary disease patients excluding one trial that heavily determined the meta-analysis result

WMD, weighted mean difference; NS, no statistically significant between group differences (P ≤ 0.05).
Two-tailed Mann-Whitney U-test.
Sensitivity analysis of trials studying forced expiratory volume in 1 s (FEV1) values in chronic obstructive pulmonary disease patients, excluding one trial that heavily determined the meta-analysis result, showing the percentage change in FEV1 from baseline after β-blocker or placebo treatment (mean ± SD) following β-agonist administration
WMD, weighted mean difference; NS, no statistically significant between group differences (P ≥ 0.05).
Two-tailed Mann-Whitney U-test.
Discussion
The results of the present meta-analysis indicated that both nonselective and cardioselective β-blockers produced a significant reduction in FEV1 in patients with COPD. However, nonselective β-blockers also produced a significant reduction in the responsiveness of the FEV1 to β-agonist administration, whereas cardioselective β-blockers did not. A subgroup analysis performed to evaluate the effect of cardioselective β-blockers on patients with comorbid cardiovascular conditions such as hypertension or chronic heart failure showed a similar reduction in FEV1 to that obtained in the analysis of all five studies; in addition, no significant change in the responsiveness of FEV1 to β-agonist administration was observed.
This meta-analysis has several limitations. It only reports on published literature and is therefore subject to publication bias. However, funnel plots of effect size versus standard error for the trials analysed showed no evidence of bias. A sensitivity analysis performed to evaluate the effect of excluding one trial that did not provide standard deviation information for the FEV1 values produced results that were consistent with those obtained in the analysis of all five studies. In addition, excluding one study that had a great weight within the meta-analysis also had little effect. The change in the responsiveness of the FEV1 to β-agonist administration when excluding this trial was in the opposite direction to that obtained in the analysis of all five studies; however, this change was still not significant.
The traditional view is that β-blockers are contraindicated in COPD because of their presumed bronchoconstrictive properties. 21 Therefore, many physicians avoid prescribing β-blockers in patients with COPD. 22 However, cardiovascular disease and COPD often coexist because of the risk of smoking-induced atherosclerosis in patients with COPD, 8 and β-blockers have been shown to have beneficial effects in hypertension, ischaemic heart disease and heart failure.23 – 25 The present meta-analysis indicated that the use of nonselective and cardioselective β-blockers in patients with COPD decreased the FEV1 slightly (by 0.14 l and 0.03 l, respectively). One study with a long duration of 4 months reported that all measures of health status exhibited a consistent nonsignificant improvement in patients treated with a cardioselective β-blocker compared with placebo, and that the mean number of COPD exacerbations was similar in the treatment and placebo groups. 20 These findings suggest that β-blockers, particularly cardioselective β-blockers, should not be contraindicated in patients who have COPD. These results are consistent with other studies that have shown that the use of β-blockers in patients with COPD and concomitant cardiovascular disease is well tolerated.26 – 30
In the present meta-analysis, three studies showed an increase in the responsiveness of FEV1 to β-agonist administration after cardioselective β-blockers,16,18, 20 which may be explained by upregulation of β-receptors in the lung after taking β-blockers. 31 Interestingly, previous studies have also shown that β-agonists can downregulate β2-receptors in asthma and COPD. 32
There are concerns that the use of β-agonists could increase all-cause mortality. 33 β-Blockers could theoretically exert beneficial effects in patients with COPD by tempering the sympathetic nervous system and reducing the ischaemic burden. 34 It has been suggested that heart rate reduction itself is an important mechanism of the benefit of β-blockers, and that resting heart rate is an independent predictor of all-cause mortality in individuals with or without cardiovascular disease. 35 Due to the lack of data on mortality in randomized controlled trials, it was impossible to carry out a meta-analysis to measure the effect of β-blockers on mortality in patients with COPD. However, observational clinical trials with a large sample size have already shown that β-blockers reduced all-cause mortality and risk of exacerbation in patients with COPD.36,37
Due to the proven benefit of β-blockers on mortality for a number of conditions, many of the other relative or absolute contraindications traditionally listed for β-blockers have been questioned and disproved: these include impaired left ventricular function, peripheral vascular disease, diabetes mellitus, depression and advanced age.4,6,38– 43 The present meta-analysis suggests that the use of cardioselective β-blockers should not be contraindicated in patients with COPD, given their demonstrated benefit in conditions such as heart failure, coronary artery disease and hypertension.
Footnotes
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article.
