Abstract
Objective
To establish and evaluate a swine model of traumatic cardiac arrest (TCA) induced by haemorrhage and ventricular fibrillation.
Methods
Thirteen male pigs were divided into a sham group (
Results
Restoration of spontaneous circulation was achieved in seven of eight animals in the TCA group. After resuscitation, the heart rate was significantly increased while the mean arterial pressure and ejection fraction were significantly decreased in the TCA group. The TCA group had significant cardiac and neurological injuries post-resuscitation and had higher serum creatinine and blood lactic acid levels and lower PaO2 than the sham group. Animals in the TCA group also exhibited significantly higher apoptotic indices and caspase-3 protein levels in the heart, brain and kidney than the sham group.
Conclusion
Animals in this swine model of TCA exhibited high rates of successful resuscitation, significant vital organ injury and prolonged survival. The model is suitable for use in further TCA research.
Introduction
Trauma ranks among the top five causes of mortality in China. 1 Annually, several million trauma patients are treated and more than 600 000 deaths occur. 1 Globally, trauma is the leading cause of death among people under the age of 40 years and is recognized as a major contemporary public health issue. 2 Cardiac arrest is the most serious complication arising from trauma and current treatment approaches are ineffective. 3 One study showed that even when the initial recovery success rate following traumatic cardiac arrest (TCA) reached 34%, the discharge survival rate was only 2.2%; and only 0.75% of patients had a good neurological prognosis. 4 Reducing overall mortality from trauma will necessitate improved clinical efficacy in the treatment of TCA. Currently available animal models of post-trauma cardiac arrest exhibit insufficient tissue injury and shortened survival times, thus reducing their usefulness. 5 This study describes a swine model of TCA established using controlled blood loss and electrical stimulation to induce ventricular fibrillation. The suitability of this model for the study of TCA in human patients was evaluated.
Materials and methods
Animals
This study was conducted in the Department of Emergency Medicine, Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, Zhejiang Province, China. The study protocol was approved by the Institutional Animal Care and Use Committee at the Zhejiang University School of Medicine (approval number: IACUC-P201702). The procedures of this study adhered to the US National Institutes of Health 2011 Guide for the Care and Use of Laboratory Animals.
Thirteen male white domestic pigs (mean body weight of 33 ± 4 kg; age range 4–6 months), which were housed with a 12-h light/12-h dark cycle and fasted overnight with no limit on their water intake (animal license number SYXK Shanghai 2013-0087), were purchased from Shanghai Jiagan Biotechnology Co., Ltd. (Shanghai, China).
Instruments
The instruments used in this study were as follows: SynoVent E5 ventilator (Shenzhen Mindray Biomedical Electronics Co., Ltd., Shenzhen, China); BeneView T6 multiparameter monitor (Shenzhen Mindray Biomedical Electronics Co., Ltd.); M9 ultrasound system (Shenzhen Mindray Biomedical Electronics Co., Ltd.); US ZOLL defibrillator monitor (ZOLL Medical Corporation, Chelmsford, MA, USA); JHBP-2000B blood pump (Guangzhou Jihua Medical Instrument Co., Ltd., Guangzhou, China); Yongsheng 85L1-A voltmeter (debounce) (Guangzhou Yongsheng Electric Instrument Co., Ltd., Guangzhou, China); Sunlife EtCO2 handheld monitor (Shanghai San Eve Electronics Co., Ltd., Shanghai, China).
Experimental methods
Animal preparation
The animals were randomly assigned to the sham treatment group (
Blood loss and cardiopulmonary resuscitation
Baseline data were recorded 10 min after preparation and arteriovenous blood samples were collected. Animals in the sham group were intubated and monitored but did not undergo haemorrhage and resuscitation. After the baseline data had been recorded, animals in the TCA group underwent haemorrhage (40% of blood volume, with total blood volume estimated as 70 ml/kg body weight 6 ) through the right femoral artery over a 20-min period using a single pump (Figure 1). The blood was stored in a blood storage bag containing tannic acid. Immediately after haemorrhage, 1 mA of alternating current was applied via the fibrillation induction electrode, inducing ventricular fibrillation. Successful induction was indicated by a ventricular fibrillation waveform on the electrocardiogram, a significant decrease in the arterial blood pressure and disappearance of the pulsation waveform. Following induction of ventricular fibrillation, the ventilator was disconnected, mechanical ventilation was stopped and the animals were observed for 5 min. Manual chest compressions (30:2) and manual ventilation with a self-inflating bag (cardiopulmonary resuscitation [CPR]) were then begun using a compression depth of >5 cm and a frequency of approximately 100 compressions per min for 2.5 min.
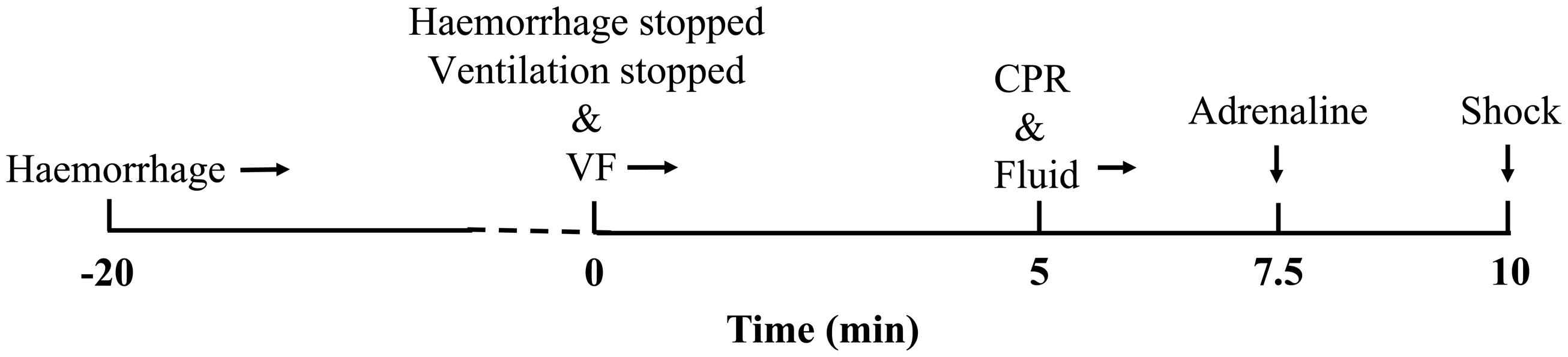
Timeline of resuscitation procedures in the traumatic cardiac arrest (TCA) group. VF, ventricular fibrillation; CPR, cardiopulmonary resuscitation.
Following CPR, 20 µg/kg of adrenaline was administered intravenously. At 5 min after cessation of CPR, biphasic defibrillation with 150 J of power was administered and return of spontaneous circulation (ROSC) was evaluated. The standard for ROSC was a supraventricular rhythm with a mean arterial pressure (MAP) of >50 mmHg for longer than 5 min. Upon failure to achieve ROSC, CPR was performed for 2 min followed by one-time defibrillation. Inability to achieve ROSC after five repetitions of this procedure was considered failure of recovery. Adrenaline was freely administered every 3 min after the first injection. Fluid resuscitation was started simultaneously with initiation of CPR: a volume of saline solution equivalent to 1.5 times the volume of removed blood was infused over the 1-h period after the initiation of CPR and 50% of the volume of removed blood was infused as an autologous transfusion over the succeeding 1-h period. Resuscitated animals were mechanically ventilated for 6 h and the ventilator tube and all invasive catheters were then removed. Incisions were disinfected and sutured, 1.2 g of amoxicillin/clavulanate potassium was injected intramuscularly and the animals were returned to the animal facility. At 24 h after resuscitation, the animals were anaesthetized with 20 mg/kg of ketamine injected intramuscularly. Next, 2 mg/kg of propofol was injected intravenously for general anaesthesia and the animals were euthanized by intravenous injection of 10% potassium chloride. The animals underwent necropsy and tissue specimens were collected.
Physiological indicators
Heart rate, blood pressure, body temperature and other vital parameters were continuously monitored. Recovery time, adrenaline use and defibrillation times were recorded before haemorrhage and 1, 3 and 6 h after successful resuscitation. Cardiac ultrasound was used to assess cardiac function (ejection fraction [EF], cardiac output [CO]). Arterial blood gas analysis was performed before haemorrhage and at 5 min and 1, 3 and 6 h after resuscitation. Phosphokinase isoenzyme (creatine kinase-MB [CK-MB]), neuron-specific enolase (NSE) and renal function (serum creatinine) were examined before haemorrhage and 1, 3, 6 and 24 h after resuscitation. Neurological function was evaluated according to the neurological deficit score (NDS) 24 h post-resuscitation. 7 The NDS was scored from 0 (no observed neurological deficits) to 400 (death or brain death).
Heart, brain and kidney tissues were fixed in 4% paraformaldehyde for 24 h, embedded in paraffin and cut into 5-µm sections. The sections were stained using a terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick end labelling (TUNEL) assay kit (Roche Diagnostics GmbH, Mannheim, Germany). In each section, six views were randomly chosen to count the number of TUNEL-positive cells and the total cells at × 200 magnification under an optical microscope (CX31 biological microscope; Olympus, Tokyo, Japan). The rate of apoptosis was calculated as the percentage of TUNEL-positive cells/total cells.
For the immunohistochemical staining of cleaved caspase-3, tissue sections on slides were heated in citrate buffer solution (0.01 mol/l; pH 6.0; Solarbio, Beijing, China) in a microwave oven (800W; P70D20P-N9(W0); Galanz, Guangdong, China) to keep the liquid temperature at approximately 98°C for 10–15 min to facilitate antigen retrieval. The slides were then cooled at room temperature for 20–30 min. The slides were incubated with primary anti-cleaved caspase-3 antibody (1:200; no. 9664; Cell Signaling Technology Inc., Danvers, MA, USA) at 4°C overnight followed by washing five times with 0.01 M phosphate-buffered solution (PBS; pH 7.4). The slides were then incubated with horseradish peroxidase-conjugated mouse anti-rabbit IgG secondary antibody (1:200; BBI, Shanghai, China) for 30 min at room temperature followed by washing five times with 0.01 M PBS (pH 7.4). The immunostaining was visualized using a diaminobenzidine immunohistochemistry colour development kit (Boster Biological Technology Co. Ltd., Wuhan, China). Six immunohistochemical staining images were randomly captured under × 200 magnification. A semi-quantitative analysis of the intensity of cleaved caspase-3-positive staining was performed through optical density using Image-Pro Plus 6.0 software (Media Cybernetics, Silver Spring, MD, USA).
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 20.0 (IBM Corp., Armonk, NY, USA). The data are expressed as mean ±SD. Inter- and intra-group data were analysed with repeated-measures analysis of variance and the independent-samples
Results
With regard to the establishment of a TCA model, the baseline characteristics of the sham and TCA groups are shown in Table 1. After haemorrhage, the mean ± SD heart rate of the animals in the TCA group significantly increased to 160 ± 27 beats/min and the mean ± SD MAP decreased to 56 ± 11 mmHg. Fibrillation was successfully induced in all animals. Seven animals in the TCA group were successfully resuscitated (87.5%); the mean ± SD recovery time was 7.5 ± 3.5 min and the mean ± SD adrenaline use was 1.50 ± 1.74 mg. The mean ± SD number of defibrillations was 2.5 ± 1.8. The success rate for single defibrillation was 50% (four of eight). Resuscitated animals survived for 24 h (24-h survival rate was seven of eight [87.5%]). The mean ± SD NDS of animals in the TCA group was 176 ± 37; none of the animals in the sham group had neurological deficits. None of the animals required special care overnight in the animal facility.
Baseline characteristics of the animals in the sham (SHAM) and traumatic cardiac arrest (TCA) groups.

Data presented as mean ± SD.
HR, heart rate; bpm, beats per min; MAP, mean arterial pressure; PaO2, partial pressure of oxygen; PaCO2, partial pressure of carbon dioxide; BE, base excess; LAC, lactic acid; Cr, creatinine.
No significant between-group differences (
In the TCA group, the post-resuscitation heart rate was significantly increased and remained at a high level compared with baseline (
Haemodynamic characteristics of the animals in the sham (SHAM) and traumatic cardiac arrest (TCA) groups.

Data presented as mean ± SD.
aInter- and intra-group data were analysed with repeated-measures analysis of variance and the independent-samples
HR, heart rate; bpm, beats per min; MAP, mean arterial pressure; EF, ejection fraction; CO, cardiac output; NS, no significant difference (
The CK-MB level was significantly increased post-resuscitation in the TCA group compared with baseline (
Organ injury markers of the animals in the sham (SHAM) and traumatic cardiac arrest (TCA) groups.

Data presented as mean ± SD.
aInter- and intra-group data were analysed with repeated-measures analysis of variance and the independent-samples
CK-MB, creatine kinase-MB; NSE, neuron-specific enolase; Cr, creatinine; NS, no significant difference (
Blood pH was lower in the TCA than the sham group after resuscitation, but returned to baseline levels after 3 h; the differences between the two groups were not significant (Table 4). After resuscitation, the base excess (BE) significantly decreased and the lactic acid (LAC) level significantly increased in the TCA group compared with baseline (
Blood gas and internal environmental indicators of the animals in the sham (SHAM) and traumatic cardiac arrest (TCA) groups.
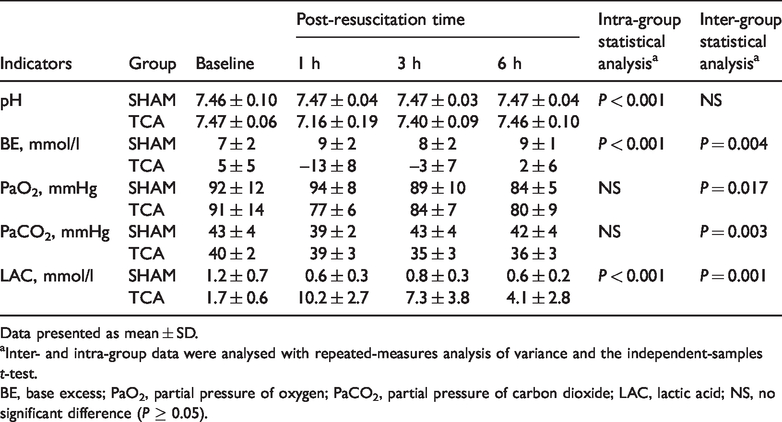
Data presented as mean ± SD.
aInter- and intra-group data were analysed with repeated-measures analysis of variance and the independent-samples
BE, base excess; PaO2, partial pressure of oxygen; PaCO2, partial pressure of carbon dioxide; LAC, lactic acid; NS, no significant difference (
In terms of major organ pathology, apoptosis was evident in the heart, brain and kidney tissues of the animals in the TCA group 24 h after resuscitation, as shown by TUNEL and increased levels of the apoptosis marker protein caspase-3 (Figure 2). Differences in the TUNEL apoptotic index and caspase-3 immunohistochemical staining in the heart, brain, and kidney were significant between the TCA and sham groups (

Representative photomicrographs demonstrating major organ pathology of the animals in the sham (SHAM) and traumatic cardiac arrest (TCA) groups as determined by measuring the levels of apoptosis using terminal deoxynucleotidyl transferase-mediated dUTP nick end labelling (TUNEL) and the levels of the apoptosis maker protein caspase-3. Apoptosis was evident in heart, brain and kidney tissues of animals in the TCA group 24 h after resuscitation. Scale bar 150 µm. The colour version of this figure is available at: http://imr.sagepub.com.
Major organ pathology in the animals in the sham (SHAM) and traumatic cardiac arrest (TCA) groups as determined by measuring levels of apoptosis in tissue samples at 24 h after resuscitation.

Data presented as mean ± SD.
aInter-group data were analysed with repeated-measures analysis of variance and the independent-samples
TUNEL, terminal deoxynucleotidyl transferase-mediated dUTP nick end labelling.
Discussion
This current study established a swine model of TCA. Pigs are useful experimental animals because of the high level of functional and metabolic similarities of the main organs of pigs and humans 8 , 9 and pigs have been considered optimal for modelling studies of human cardiac arrest. 10 , 11
Frequent causes of TCA include low blood volume, hypoxia, head or spinal injury and tension pneumothorax. Among these, low-volume factors caused by blood loss account for more than 40% of cases. 12 Accordingly, this current study used controlled blood loss combined with induction of ventricular fibrillation to establish a TCA model and evaluate the accompanying pathophysiological changes. The model was based on a simple haemorrhagic shock protocol for cardiac arrest and the 2015 American Heart Association Cardiopulmonary Resuscitation Guidelines. 13 Prior to haemorrhage, the TCA and sham groups were not significantly different in terms of their baseline characteristics and haemodynamics. After haemorrhage, the animals in the TCA group exhibited a significantly increased heart rate and decreased MAP as well as other features consistent with haemodynamic changes following general trauma. Following traumatic haemorrhage, cardiac arrest and CPR, the body may undergo severe damage from systemic ischaemia–reperfusion injury.14–16 In traumatic blood loss-induced cardiac arrest, the body’s circulatory function is gradually depleted. This leads to severe systemic ischaemia, hypoxia and acidosis, causing cell death and tissue damage. During CPR and reperfusion, many harmful substances, including oxygen free radicals and inflammatory factors, are released; this release can cause diffuse cell and tissue damage, secondary changes to cell structure, increased permeability, and cell death from necrosis, apoptosis, autophagy, and necrotic apoptosis. 17 At the organ level, CPR and reperfusion can result in heart and brain injuries, 17 , 18 which progressively lead to post-cardiac arrest syndrome (PCAS). 19 Prevention and treatment of PCAS is considered key to improving survival of patients with cardiac arrest.
Current animal cardiac arrest models mainly involve simple traumatic blood loss or cardiac arrest. 5–7 These models have a minimal focus on TCA and very few induce ventricular fibrillation after blood loss. 5 Such models exhibit less damage than clinical TCA, but nonetheless short survival times. 5 This is not consistent with clinical TCA, which frequently involves more severe tissue damage and serious ischaemia–reperfusion injury.18,20 A new animal model that better replicates the features of PCAS is thus needed.
In preliminary experiments conducted by this research group, which involved 50% haemorrhage and various durations of ventricular fibrillation, the animals failed to recover. These preliminary studies resulted in the establishment of a pig TCA model using 40% haemorrhage over a 20-min period and 5 min of ventricular fibrillation. After resuscitation, the blood lactate level peaked at approximately 10 mmol/l, suggesting that the tissue damage caused by blood loss and cardiac arrest was severe. This current study focused on the assessment of damage to the heart, brain, and kidneys after resuscitation. The CO and EF in the TCA group were decreased more than 20% after resuscitation. The myocardial injury index (CK-MB level) and brain tissue damage index (NSE level) were both significantly increased compared with baseline in the TCA group. These findings indicate obvious heart and brain damage and reduced heart function after resuscitation. These indicators had not returned to baseline levels at 6 or 24 h after resuscitation, suggesting serious injury beyond the limits of self-correction. This model provides an experimental platform to explore active post-resuscitation interventions for TCA to improve heart and brain function. This current study also showed alterations in renal function post-resuscitation, further suggesting that this PCAS model exhibits multi-organ damage.
The tissue pathology of animals in the TCA group indicated apoptosis of cells in three major organs, as confirmed by increased levels of caspase-3 compared with the sham (control) tissue samples. Thus, this model exhibits features of organ damage consistent with clinical TCA, suggesting that this model would be valuable in further studies designed to elucidate and test clinically relevant interventions.
This current study had several limitations. First, it lacked a group without arrest but with 7.5 min of chest compressions; therefore, elevation of CK-MB secondary to mechanical cardiac compression cannot be excluded. 21 Secondly, these animals were able to leave medical care within 6 h of cardiac arrest, which would be very unusual in a clinical setting. Thirdly, the relatively short period (24 h) of monitoring of heart, brain and kidney function prevented complete dynamic detection and analysis of maximal organ injury and the extent of recovery following TCA. Finally, although organ injury was documented after resuscitation, there were no in-depth insights into the injury mechanisms gained. These limitations may be addressed in future studies using the swine TCA model established herein.
In conclusion, this study demonstrated the successful establishment of a new swine model of TCA. The resuscitation rate, degree of major organ damage and prolonged survival time meet the requirements of an animal model suitable for the productive study of TCA and elucidation and testing of potential interventions to increase survival rates.
Footnotes
Acknowledgements
We thank Moli Wang and Chunxiao Wu from the Department of Emergency Medicine, Yuyao People’s Hospital, Medical School of Ningbo University, Ningbo, Zhejiang Province, China for their assistance with the animal preparation.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported by the Welfare Scientific Research Project of Zhejiang Province (No. LGF18H150003), the Key Joint Research Project of Chinese Ministry of Health & Zhejiang Province (No. 2018271879) and the Welfare Scientific Research Project of Zhejiang Province (No. LGD19H150003).
