Abstract
Background
We aimed to analyze the epidemiological and drug-resistance trends among bacterial cultures from perioperative infections in patients with primary ovarian cancer.
Methods
Medical and bacteriological records for patients with ovarian cancer patients who developed perioperative infections after primary cytoreductive surgery from 1999 to 2018 were reviewed retrospectively.
Results
The incidence of perioperative infections and the culture-positive percentage among patients in the first 10 years were 20.2% and 29.3%, respectively, and the equivalent rates in the second 10 years were 18.0% and 33.5%. The most commonly isolated pathogens in both year-groups were Escherichia coli and Enterococcus spp., but the respective percentages differed between the groups. Some strains of Staphylococcus aureus and Enterococcus spp. in the second 10-year group were resistant to linezolid and vancomycin, and ciprofloxacin resistance among Gram-negative bacteria isolates also increased in this group. However, resistance of Gram-negative bacteria to imipenem and meropenem was low among in both groups.
Conclusion
The pathogen distribution in perioperative infections in patients with primary ovarian cancer undergoing cytoreductive changed slightly from 1999 to 2018, and the antibiotic resistance of the main isolated pathogens increased. These results indicate the importance of periodic bacterial surveillance of surgical infections in these patients.
Keywords
Background
Ovarian cancer remains a common gynecologic malignancy, with the third-highest incidence among malignant female gynecologic tumors, after cervical and endometrial cancer.1,2 Ovarian cancer is also the fifth-leading cause of cancer-related deaths among women, 3 with an increasing incidence year by year. 4 However, the symptoms of early-stage ovarian cancer are less obvious, and most patients are therefore initially diagnosed at an advanced-stage.3,5 Although primary or secondary cytoreductive surgery remains an important treatment for ovarian cancer, 6 residual cancer foci after cytoreductive surgery may affect the survival outcome,7,8 and relatively radical and extensive excision are usually carried out to obtain maximal cytoreduction and improve survival outcomes.9,10 However, the use of radical and extensive surgery is directly related to postoperative complications, including perioperative infection.9,11
Perioperative infection is one of the most common complications in patients with ovarian cancer, and is associated with not only surgical morbidity and mortality, but also with high health care-related costs.12,13 Perioperative infection was the third-leading cause of death within 30 days after primary cytoreductive surgery among patients with advanced-stage ovarian cancer. 14 Perioperative infections can also delay the onset of postoperative chemotherapy. 15 Perioperative infections after cytoreductive surgery therefore represent a crucial problem that requires further study. The urinary tract and surgical wound are common sites of infection after primary or secondary cytoreductive surgery in patients with ovarian cancer. 16 Regarding the causative organisms, Escherichia coli and Enterococcus spp. have been reported to be the most common and important isolated pathogens, 3 while the pathogen distribution and antibiotic resistance profiles of the main pathogens have gradually changed. 3 However, the morbidity and mortality of perioperative infections in patients with ovarian cancer remain unclear. 17 There have been no large, long-term studies of the species distribution and antibiotic resistance of perioperative bacterial infections in patients with primary ovarian cancer in mainland China. The present study therefore aimed to analyze the trends in bacterial epidemiology and antibiotic resistance in bacterial cultures isolated from perioperative infections in patients with primary ovarian cancer at an affiliated hospital in South China over a 20-year period.
Materials and methods
We retrospectively extracted and reviewed the records of patients with primary ovarian cancer admitted to a university teaching hospital (The Second Xiangya Hospital of Central South University, Changsha, Hunan) with perioperative infection from 1 January 1999 to 31 December 2018. Perioperative infection after surgery for primary ovarian cancer, advanced stage ovarian cancer, tumor grade, and culture-positive percentage were defined as reported previously.3,18,19 The inclusion criterion was patients with primary ovarian cancer who underwent cytoreductive surgery. The exclusion criteria were: (i) patients with metastatic ovarian carcinoma; (ii) patients with synchronous cancer types; (iii) patients with low-grade malignant ovarian tumor; and (iv) patients with incomplete information. Information from the patient’s medical records was extracted and reviewed, including clinical demographics, results of pathological examinations, clinical and surgical details, and details of inpatient treatment. We also retrospectively extracted the results of microbiology tests and antimicrobial sensitivity assays to evaluate perioperative infections. All samples were analyzed using standard microbiological methods and all antimicrobial susceptibility tests were carried out in our hospital using a unified protocol.20,21 To avoid duplicate counts, we extracted and recorded similar bacteria isolated from different specimens from the same patient or similar bacteria isolated at different times as one isolate.20,22 This study was approved by the ethics committee of The Second Xiangya Hospital of Central South University, Changsha, Hunan, and informed consent was obtained from the participating patients and/or their parents/guardians.
All the data were analyzed using SPSS Statistics for Windows, version 21.0 (SPSS Inc., Chicago, IL, USA). Continuous variable were described as the mean ± standard deviation and categorical variables as number and percentage. Differences between patients treated in the first 10 years (1 January 1999 to 31 December 2008) and those treated in the second 10 years (1 January 2009 to 31 December 2018) were analyzed using χ2 and t-tests. A two-tailed P-value < 0.05 was considered significant for all tests.
Ethical approval
This study was approved by the ethics committee of The Second Xiangya Hospital Committee for clinical research (No. 2010-S233), and informed consent was obtained from the participating patients and/or their parents/guardians for publication of their individual clinical details.
Results
Demographics of perioperative infection
The demographic data of the enrolled patients with primary ovarian cancer are reported in Table 1 and Table 2. During the 20-year period, a total of 854 patients with ovarian cancer were treated with primary cytoreductive surgery in our hospital, including 322 patients from 1999 to 2008 and 532 from 2009 to 2018. Among these patients, 161 (161/854) patients developed a perioperative infection within 6 weeks after surgery, including 65 (65/322) patients in the first 10-year group and 96 (96/532) in the second 10-year group. There were no significant differences in any characteristics between the two groups.
Clinical demographics of patients with ovarian cancer.

aValues given as mean ± standard deviation (range); bvalues given as mean ± standard deviation. BMI, body mass index.
Characteristics of perioperative infections in women with ovarian cancer.

Causative organisms and bacterial resistance
Totals of 522 and 674 samples of bacterial cultures were analyzed for the first and second 10-year groups, respectively. The positivity rate of the bacterial cultures was higher in the second group (226/674) compared with the first group (153/522), but the difference was not significant. The top three most common sites of perioperative infections in both groups were the urinary tract, surgical wound, and blood (Table 2). Regarding the distribution of all causative organisms, the percentage of Gram-negative bacteria was higher than that of Gram-positive bacteria and fungi at all sites and in both groups. Among top five most common species, Enterococcus spp. were responsible for 22.9% of infections in the first 10-year group, followed by E. coli (20.9%), Klebsiella pneumoniae (13.1%), Acinetobacter baumannii (12.4%), and Staphylococcus aureus (11.8%), while E. coli was responsible for 27.4%, followed by S. aureus (15.0%), K. pneumoniae (14.2%), A. baumannii (13.3%), and Enterococcus spp. (10.2%) in the second 10-year group (Figure 1). Among urinary tract infections, the most commonly isolated pathogens were Enterococcus spp. (21.7%) and E. coli (20.7%) in the first group, compared with E. coli (23.8%) and S. aureus (15.6%) in the second group. E. coli was the most common organism isolated from surgical wound infections in both groups (Table 2).

Pathogen distributions of perioperative infections in women with ovarian cancer from 1999 to 2008 and 2009 to 2018.
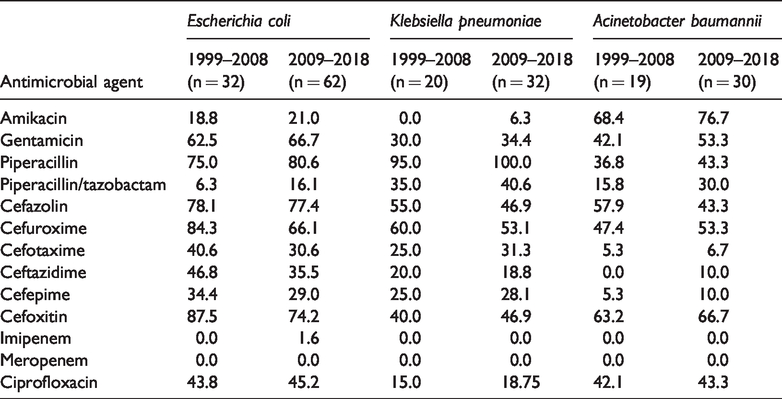
Gentamicin resistance remained high among the common causative organisms. One strain of S. aureus and one strain of Enterococcus spp. were resistant to vancomycin, and two strains of Enterococcus spp. were resistant to linezolid in the second 10-year group. Ciprofloxacin and erythromycin resistance were decreased in S. aureus in the second 10-year group (Table 3), and penicillin had no effect on most S. aureus. However, levofloxacin and ciprofloxacin resistance increased in Enterococcus spp. in the second 10-year group. Regarding E. coli, resistance to cefazolin, cefuroxime, and cefoxitin were high in both groups, and one strain of E. coli in the second group was resistant to imipenem. The resistance of K. pneumonia to piperacillin was nearly 100% in the second 10-year group. Resistance of Gram-negative bacteria to imipenem and meropenem was low in both groups, but resistance of Gram-negative bacterial isolates to ciprofloxacin resistance increased in the second 10-year group (Table 4).
Resistance rates (%) of Gram-positive bacteria to antimicrobial agents.

Resistance rates (%) of Gram-negative bacteria to antimicrobial agents.
Discussion
Ovarian cancer is the third-most prevalent gynecologic malignant tumor,1,2 and the fifth-leading cause of cancer-related deaths among women, 3 and thus presents a serious threat to women’s health and quality of life. Although treatments for ovarian cancer have become more effective in recent years, the best results occur in patients treated in the early stages of the disease; however, most patients with ovarian cancer are initially diagnosed at an advanced stage.3,5 Current therapeutic modalities for ovarian cancer comprise surgery and chemotherapy. 23 Primary cytoreductive surgery aims to eradicate cancer cells as thoroughly as possible, but aggressive cytoreductive surgery has been associated with perioperative infections, including surgical site, urinary tract, and other site infections. 24 Many factors contribute to the development of perioperative infections in patients with ovarian cancer after cytoreductive surgery, including the microorganism, the environment, and host defense mechanisms, while ovarian cancer patients frequently undergo extensive, prolonged, and highly invasive surgery, experience significant blood loss, have received preoperative chemoradiotherapy, and have primary or secondary hypoimmunity. 17 In the present study, the rate of perioperative infections in patients with ovarian cancer after primary cytoreductive surgery was higher (18.9%) than previously reported,3,17,18 suggesting the need to take preventive preoperative and perioperative actions to reduce the rate of perioperative infections in these patients in our hospital.
Perioperative infections were common in women with ovarian cancer after primary cytoreductive surgery in the present study. The urinary tract and surgical wound were the most common infection sites in both groups, 18 but the percentage of urinary tract infections was higher in the second 10-year group (65.0%) compared with the first 10-year group (60.1%). Numerous factors increase the risk of perioperative urinary tract infections during ovarian cancer surgery, including anatomical features, ascending infection from the lower genital tract, invasive instrumentation such as cystoscopy and catheterization, surgical intervention, chemoradiotherapy, and dysbiosis of vaginal microbiota.25–27
The distribution of the causative organisms changed slightly between the two decades in the current study. Some previous studies found that the most common isolated pathogens were Pseudomonas aeruginosa followed by Enterococcus and E. coli, 18 while other studies reported that E. coli was the main causative organism, followed by Enterococcus.3,17,25 Furthermore, a shift in the vaginal flora may have affected the distribution of causative organisms in perioperative infections.28–30 Many factors may alter the host microbial flora leading to pathogenic bacterial colonization, including surgery, antimicrobial therapy, chemoradiotherapy, hospital equipment, and patient to patient transmission. 17 Similarly, use of foreign bodies such as contraceptive devices, frequent use of vaginal tampons, previous ectopic pregnancies, prenatal infections, and a history of sepsis history are also possible factors. In the present study, although E. coli and Enterococcus spp. were the most commonly isolated pathogens in both 10-year groups, their respective percentages changed between the first and second group. The use of aseptic techniques, surgical skill, and periodic surveillance of the causative microbial pathogens are all required to help prevent perioperative infections in patients with ovarian cancer.
After the introduction of antibiotics into clinical practice, they were widely used as preoperative prophylaxis to prevent perioperative infections. The choice of prophylactic antibiotic should take into consideration numerous factors, including the common pathogenic bacteria and their drug resistance, the pharmacokinetics and pharmacodynamics of the drug, and its cost effectiveness and toxicity. 17 Rapid and accurate identification of causative organisms is thus an cornerstone of antimicrobial therapy. E. coli and Enterococcus were reported to be sensitive to nitrofurantoin, norfloxacin, gentamicin, amikacin, and cotrimoxazole in 1995; 25 by 2012, pathogens responsible for urinary tract infections showed high levels of resistance to ciprofloxacin, ampicillin/sulbactam, cefazolin, and levofloxacin, and some strains of E. coli were resistant to impenem and piperacillin/tazabactam. 3 In the present study, we detected vancomycin-resistant S. aureus and Enterococcus spp., linezolid-resistant Enterococcus spp., and imipenem-resistant E. coli in the second 10-year group. Treatment options for infections caused by multidrug-resistant pathogenic bacteria are thus limited, and inappropriate empirical antibiotic therapy may have poor outcomes. Periodic surveillance of drug resistance can guide the use of empirical clinical medications and the appropriate employment of antibiotics.
Penicillin has not been considered as an appropriate empiric antibiotic therapy for S. aureus in most countries since 1980, 31 and the overuse of penicillin meant that penicillin resistance among S. aureus isolates in the current study was nearly 100%. Furthermore, the wide use of macrolides in patients allergic to penicillin and cephalosporins, especially oral azithromycin and roxithromycin, has increased the incidence of erythromycin-resistant S. aureus and Enterococcus spp., according to a Spanish study. 32 Meanwhile, gentamicin is no longer considered as an appropriate empiric antibiotic therapy for Gram-negative bacteria. 33 During the 20-year period of this study, the incidence of resistance to second generation cephalosporins among Gram-negative bacterial isolates was high level, but the causative isolates showed high sensitivity to third or fourth generation cephalosporins such as cefotaxime, ceftazidime, and cefepime, and Gram-negative bacteria showed low resistance to imipenem and meropenem. We therefore recommend the use of third or fourth generation cephalosporins as the first-line treatment for perioperative infections in women with ovarian cancer, while carbapenems should be reserved for severe infections caused by Gram-negative bacteria.
Perioperative infections in women with ovarian cancer are associated with increasing health-care-related costs, reduced quality of life, and significant strains on inpatient care. 34 We should therefore adopt suitable prevention strategies to decrease the incidence of perioperative infections.34,35 Drug resistance is an increasing problem, especially in patients with malignant gynecologic tumors. Previous studies have suggested that numerous factors may contribute to the emergence of resistance, including inappropriate antibiotic selection, long-term incorrect antibiotic usage, and the duration of prophylactic antibiotic use.17,36 To reduce the rates of infection and bacterial resistance, it is therefore necessary to first take preventive measures against perioperative infections, followed by the use of narrow-spectrum antibiotics whenever possible, discontinue or change the antibiotics based on the results of bacterial cultures, and finally take heed of dynamic antibiotic resistance surveillance research. 37
We acknowledge some limitations of this study. First, the study had a retrospective design, which is associated with potential bias; however, the sample size was relatively large. Second, we did not record or analyze operation duration, the interval between the operation and infection, or the change in prophylactic antibiotics during the course of the ovarian cancer. Finally, this was a single-center study, and there is thus a clear need for larger, multicenter studies in the future.
Conclusions
Based on the results of this study, we concluded that the incidence of perioperative infections after cytoreductive surgery in women with primary ovarian cancer remains high. During the 20-year study period, the pathogen distribution changed slightly and antibiotic resistance of the major pathogens increased gradually. Periodic bacterial surveillance and various preventive actions during the preoperative and perioperative periods play important roles in the prevention and treatment of perioperative infections in women with ovarian cancer.
Footnotes
Availability of data and materials
All the data used and/or analysis during this study are included in this published article and its supplementary information files.
Acknowledgements
We would like to thank The Second Xiangya Hospital for official permission to carry out this study and the microbiology staff for helping with data collection.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The study was supported by National Natural Science Foundation of China (81801425) and Natural Science Foundation of Hunan (2018JJ3739).
