Abstract
Objective
Patients receiving carbon-ion radiation therapy and astronauts exploring outer space are inevitably exposed to heavy ion radiation. The aim of this study was to develop radioprotectors to minimize the injuries induced by carbon ion radiation.
Methods
Heat-killed Salmonella Typhimurium (HKST) was administered to mice by gavage prior to irradiation with a 12C6+ heavy ion accelerator. Hematoxylin and eosin staining and immunofluorescence TdT-mediated dUTP Nick-End Labeling staining were used to assess the radioprotective effect of HKST on organ damage and levels of apoptosis, respectively, in mice. To investigate the mechanism underlying the radioprotective effect of HKST, levels of the pro-apoptotic proteins BAX and caspase 3 as well as interferon-regulatory factor (IRF) 3/7 in the femur, testis and intestine were assessed using immunofluorescence.
Results
Injuries induced by carbon ion radiation were significantly eased by pretreatment with HKST. Both apoptosis and high expression levels of pro-apoptotic proteins induced by heavy ion radiation were inhibited by HKST pretreatment. The radioprotective effect of HKST was associated with stimulation of Toll-like receptor signaling mediated by enhanced IRF3 and IRF7 signaling.
Conclusion
HKST was an effective radioprotector alleviating damage to multiple organs caused by heavy ion radiation.
Introduction
Carbon-ion radiation therapy (CIRT) represents a promising method to overcome tumor radioresistance and extend patient survival because of its high ionization density and unique ballistic properties (Bragg peak).1,2 Although CIRT can more precisely deliver high-dose radiation to tumors compared with photon-based intensity-modulated radiation therapy, 3 toxicity caused by heavy charged particles while traversing healthy tissues is also of significant concern. 4 In addition, human exploration of outer space inevitably results in exposure of astronauts to energetic particle radiation. 5 Previous studies have shown that about 30% of astronauts’ cells could be hit by heavy ions during a round trip to Mars, but current shielding measures are insufficient to block space radiation. 6 Compared with photon-based radiation including X-rays and γ-rays, the higher relative biological effectiveness and linear energy transfer (LET) of heavy ions results in clustered and irreparable damage. 1 Countermeasures to mitigate toxicity caused by ionizing radiation are urgently needed, and development of radioprotective agents is one of the most effective strategies. 7 Amifostine (ethyol or WR-2721) is the only compound that has been approved by the FDA to protect healthy tissues against the hazardous impacts of ionizing radiation. 8 However, its clinical applications are limited by its toxicity, short half-life and high cost.9,10 The development of novel radioprotectants against heavy ion radiation is urgently required for patients receiving CIRT and astronauts exploring space. Toll-like receptors (TLRs) are major pattern recognition receptors facilitating the elimination of pathogens. Since the first report that activation of TLR5 protected mice and monkeys against radiation injury, 11 stimulation of other TLRs (such as TLR2/6, TLR4 and TLR9) has been demonstrated to minimize radiation-induced damage.12–14 Salmonella Typhimurium was reported to be recognized by multiple TLRs, including TLR2 and TLR4, thereby stimulating the immune system.15,16 Previous studies have shown that pretreatment with attenuated Salmonella Typhimurium may induce protective cell-mediated immunity and facilitate the conversion of immunosuppressive myeloid-derived suppressor cells into tumor necrosis factor-α-secreting neutrophil-like myeloid cells, thereby protecting mice against malaria or tumor growth.17,18 In our preliminary work, we confirmed a radioprotective effect of heat-killed Salmonella Typhimurium (HKST) against damage induced by γ-irradiation through a TLR4-biased mechanism. 19 In this study, we investigated the protective effects of HKST against injury induced by carbon ion radiation. We found that intragastric infusion of HKST prior to carbon ion irradiation could significantly alleviate toxicity in vivo. The radioprotective effects of HKST against heavy ion radiation resulted from inhibition of apoptosis pathways and stimulation of TLR signaling mediated by upregulation of interferon-regulatory factor (IRF)3/7 signaling.
Materials and methods
Irradiation
The 12C6+ heavy ion accelerator was located at the Chinese Academy of Sciences (Institute of Modern Physics, Lanzhou, Gansu, China). Radiation was administered in a 3.5 × 3.5 cm2 radiation field at an energy of 350 Mev/U (LET 7.28 keV/μm) with dose rate of 0.5 Gy/min.
Mouse procedures
Animal experiments were approved by the Ethics Committee of The Second Military Medical University and followed the instructions for the Care and Use of Laboratory Animals published by the NIH (Publication No. 96-01). Mice (C57BL/6J, male, 6–8 weeks old; Chinese Academy of Sciences, Lanzhou, China) were housed with a 12-hour light/dark cycle. Water and food were provided ad libitum. HKST (Invivogen, San Diego, CA, USA, Lot: HST-39-01) resuspended in phosphate-buffered saline (PBS) (107 cells/mouse) was administered by gavage 12 hours prior to irradiation.
Hematoxylin and eosin (HE) staining and immunofluorescence TdT-mediated dUTP Nick-End Labeling (IFTUNEL) staining
Mice were randomly divided into a control group (Con), an ionizing radiation group (IR), and an ionizing radiation plus HKST group (IR+HKST). Each group contained at least three mice. Mice were anesthetized by intraperitoneal injection with 7% chloral hydrate (100 μL/mouse) and pretreated with HKST (107 cells/mouse) or placebo 12 hours prior to administration of 4 Gy heavy ion radiation. Twenty-four hours post-irradiation, mice were euthanized with carbon dioxide. A femur, testis and intestine from each mouse were then isolated, fixed in paraformaldehyde and subjected to HE and IFTUNEL staining. Tissue damage was assessed following HE staining as previously described. 20 The TUNEL kit (Roche, Basel, Switzerland; Lot: 11684817910) was used for IFTUNEL staining following the manufacturer’s instructions. A fluorescent microscope (Olympus BX60, Center Valley, PA, USA) equipped with a digital camera (Retiga 2000R, Surrey, Canada) was used to take images of sections.
Immunofluorescence analysis
Expression of BAX, caspase 3, IRF3 and IRF7 was detected by immunofluorescence analysis. Briefly, femurs, testis and intestine were fixed in 4% paraformaldehyde followed by dehydration and paraffin embedding. The tissues were cut into 3-μm-thick sections. Following blocking with bovine serum albumin, cells were stained with anti-BAX (Abcam, Cambridge, UK; ab32503, 1:250), anti-caspase 3 (Abcam, ab179517, 1:150), anti-IRF3 (Abcam, ab68481, 1:100), and anti-IRF7 (Abcam, ab115352, 1:200) antibodies followed by a secondary antibody (Proteintech, Rosemont, IL, USA; 1:1000). Images were obtained using a fluorescent microscope (Olympus BX60) equipped with a digital camera (Retiga 2000R).
Statistical analysis
Data from each experiment were presented as means ± standard errors of mean. Differences between groups were assessed using the Student’s t test. Values of P < 0.05 were considered statistically significant. All experiments were repeated at least three times independently and quantification was conducted in a blinded fashion.
Results
HKST alleviated tissue damage induced by heavy ion radiation
The hematopoietic, gastrointestinal and genital systems are susceptible to radiation injury. To investigate the radioprotective effects of HKST against carbon ion radiation, HE staining and histological examinations were used to assess damage to bone marrow, intestine and testis. As shown in Figure 1, structures of the bone marrow, small intestine and testis were destroyed by carbon ion radiation. Levels of nucleated cells in bone marrow were dramatically decreased and villi became discontinuous, with villus cell loss (Figure 1, a and b). HKST pretreatment significantly mitigated radiation toxicity and preserved the structural organization of the bone marrow, intestine and testis. Decreased levels of nucleated cells in the bone marrow and villus cell loss were alleviated. In the testis, HKST administration maintained a relatively complete structure, with fewer necrotic cells and cavities in convoluted seminiferous tubules observed (Figure 1c).

Tissue damage induced by carbon ion radiation was alleviated by HKST pretreatment. Mice receiving 12C6+ heavy ion radiation were pretreated with PBS or HKST. Twenty-four hours post irradiation, mice were sacrificed and the protective effects of HKST on femur (a), intestine (b) and testis (c) were assessed by HE staining. Abbreviations: HKST, heat-killed Salmonella Typhimurium; IR, ionizing radiation.
HKST reduced apoptosis following heavy ion radiation
Radiation-induced syndrome can be initiated by apoptosis. Thus, TUNEL staining was used to determine whether HKST could alleviate radiation-induced apoptosis in the femur marrow, intestine and testis. Levels of apoptosis in the bone marrow, intestine and testis were dramatically increased following exposure to carbon ion radiation (Figure 2). HKST pretreatment effectively reduced levels of apoptosis in the bone marrow and intestine following heavy ion radiation. The anti-apoptotic effect of HKST was more apparent in intestinal villus cells compared with crypt cells (Figure 2b).

Post-carbon ion irradiation apoptosis was inhibited by HKST pretreatment. Intestine (a), testis (b) and femur (c) were isolated 24 hours post-irradiation and levels of apoptosis was measured IFTUNEL staining. Abbreviations: Con, control; DAPI, 4′,6-diamidino-2-phenylindole; HKST, heat-killed Salmonella Typhimurium; IR, ionizing radiation; ns, not significant.
HKST attenuated overexpression of pro-apoptotic proteins induced by heavy ion radiation
Cleaved caspase 3 and Bax are two major pro-apoptotic proteins that initiate apoptosis. We assessed the influence of HKST treatment on levels of cleaved caspase 3 and Bax following carbon ion irradiation by immunofluorescence analysis. Levels of Bax in the small intestine were significantly decreased by HKST pretreatment (Figure 3). Moreover, levels of caspase 3 in the femur marrow, intestine and testis were decreased by HKST pretreatment (Figure 4), indicating inhibition of radiation-induced apoptosis pathways by HKST.

Influence of HKST treatment on the expression level of BAX protein post-carbon ion irradiation. Femur (a), intestine (b) and testis (c) were cut into 3-μm-thick sections, then blocked with bovine serum albumin. Tissues were stained with an anti-BAX antibody followed by a secondary antibody. Levels of BAX protein in tissues were detected by fluorescent microscopy and analyzed using ImageJ software. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; HKST, heat-killed Salmonella Typhimurium; IR, ionizing radiation; ns, not significant.

Upregulation of caspase 3 protein in different tissues post-carbon ion irradiation was inhibited by HKST pretreatment. Tissues (a: femur, b: intestine, and c: testis) were stained with anti-caspase 3 antibody followed by a secondary antibody. Levels of caspase 3 protein in tissues were assessed by fluorescent microscopy and analyzed using ImageJ software. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; HKST, heat-killed Salmonella Typhimurium; IR, ionizing radiation.
HSKT stimulated the IRF3/7 pathways following heavy ion radiation exposure
IRF3/7 signaling was reported to be involved in DNA damage repair which is vital to cellular recovery following radiation exposure. The effects of HKST pre-treatment on IRF3/7 signaling following carbon ion radiation exposure were assessed by immunofluorescence analysis. As shown in Figure 5, levels of IRF3 and IRF7 in the bone marrow, intestine and testis were slightly increased following heavy ion radiation exposure. However, HKST pre-treatment induced overexpression of IRF3 in the bone marrow and intestine. Moreover, levels of IRF7 in the femur marrow, intestine and testis were all upregulated by HKST treatment.
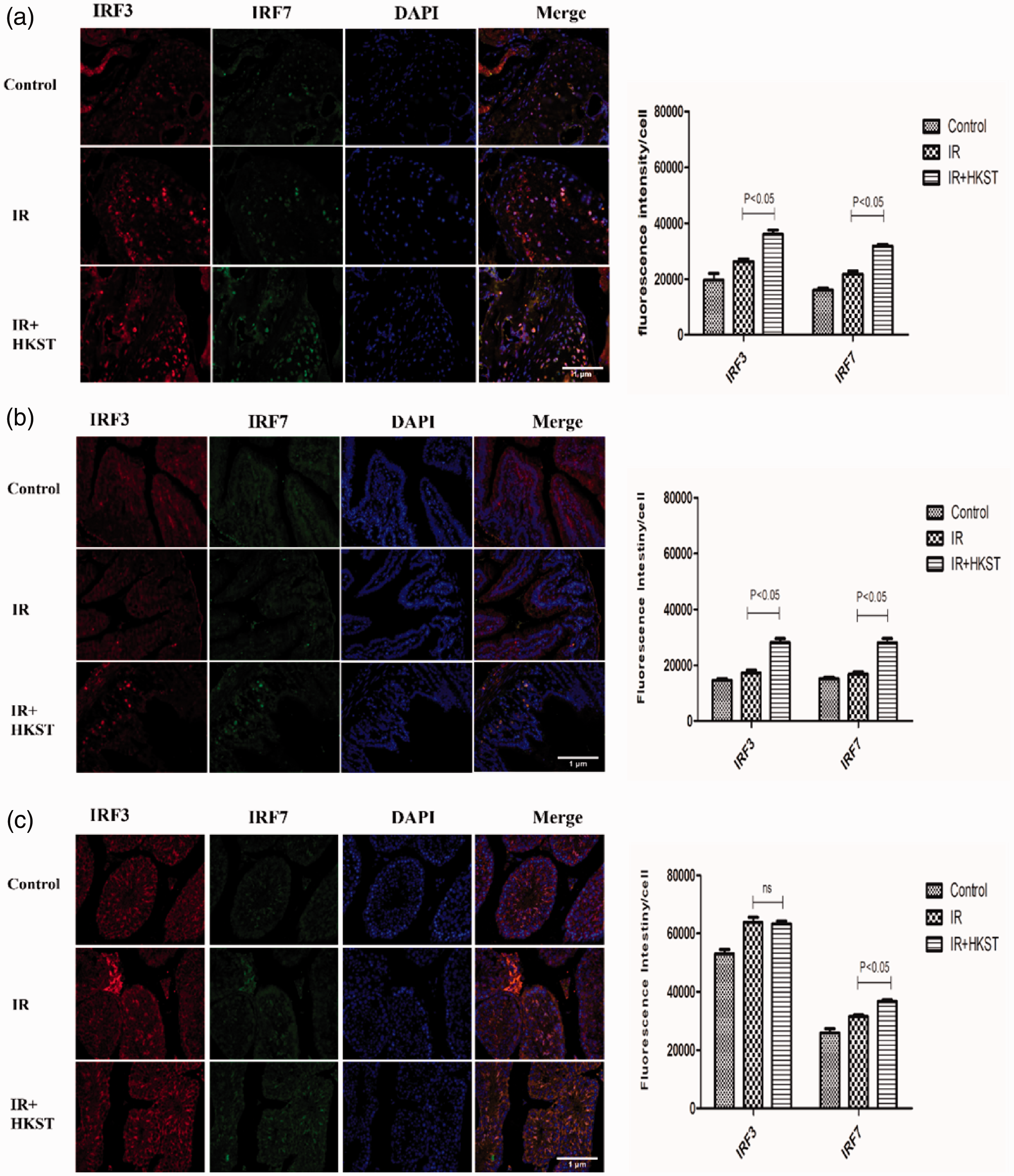
The IRF3/7 pathways were stimulated by HKST treatment. Tissues (a: femur, b: intestine, and c: testis) were cut into 3-μm-thick sections and stained with anti-IRF3 or -IRF7 antibodies followed by a secondary antibody. Levels of IRF3 or IRF7 protein in tissues were assessed by fluorescent microscopy and analyzed with ImageJ software. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; HKST, heat-killed Salmonella Typhimurium; IR, ionizing radiation; ns, not significant.
Discussion
With a high local dose delivered along particle tracks leading to irreparable DNA damage and cell death,21,22 CIRT is increasingly used to treat radioresistant tumors. 2 Nonetheless, damage to normal tissue surrounding the tumor induced by heavy ion radiation remains a major concern. In addition, astronauts exploring space will inevitably be hit by heavy ions during long missions. 6 It is imperative to develop radioprotective agents to minimize injury caused by heavy ion radiation. In the current study, the radioprotective effects of HKST against carbon ion radiation were investigated. Carbon ion radiation-induced damage to multiple organs, including the bone marrow, intestine and testis of mice, was dramatically alleviated by pre-treatment with HKST. HKST is a potent co-agonist of TLR2, TLR4 and TLR5, and its recognition by multiple TLRs can induce protective cell-mediated immunity.16,18 TLR stimulation has been demonstrated to minimize injuries induced by photon-based ionizing radiation.12–14 However, the radioprotective effects of TLRs against heavy ion radiation have not been investigated. To the best of our knowledge, HKST is the first TLR co-agonist reported to protect organisms against heavy ion radiation.
Following carbon ion irradiation, more significant apoptosis will be induced compared with photon-based radiation. Apoptosis is one of the major forms of cell death and initiates the syndrome caused by radiation injury,23,24 and it was reported that activation of TLRs signaling could prevent apoptosis induced by radiation. 25 Thus, we hypothesized that the radioprotective effect of HKST against heavy ion radiation resulted from inhibition of apoptosis. We found that levels of apoptosis in the bone marrow and intestine following carbon ion irradiation were significantly inhibited by HKST. Consistently, decreased levels of pro-apoptotic proteins including cleaved caspase 3 26 and BAX 27 were observed in the bone marrow, intestine and testis in mice pretreated with HKST, reflecting inhibition of apoptotic signaling pathways. Given that DNA damage can lead to apoptosis following radiation exposure,28,29 inhibition of apoptotic signaling following carbon ion radiation with HKST pretreatment might involve stimulation of the DNA damage response pathway. This possibility remains to be further investigated.
TLR signaling involves several adaptor proteins, including MyD88, MAL, TRIF, TRAM and SARM. It was reported that MyD88 promoted stimulation of IRF7 independently from NF-κB activation, while TRAM recruited TRIF to TLR4 resulting in activation of IRF3. 30 Following DNA damage induced by chemical agents or radiation, signal transduction pathways were triggered by IRF3 or IRF7 to mediate either protection or apoptosis of damaged cells.31,32 IFN signaling was also reported to promote recovery of hematopoiesis following ionizing radiation exposure. 33 Because HKST is a co-agonist of the TLR family, the radioprotective effects of HKST against carbon ions might involve stimulation of IRF3/7 signaling. Our results showed that levels of IRF3 and IRF7 were significantly increased in the intestine and bone marrow following irradiation with HKST pretreatment, and IRF7 expression was stimulated in the testis by HKST upon radiation damage. A previous study demonstrated crosstalk between ataxia telangiectasia mutated (ATM) signaling and IRF7 in DNA damage responses induced by paramyxoviruses. 34 In addition, the ataxia telangiectasia and Rad3 related (ATR) pathway is essential for activation of IRF3 in lymphoma cells during DNA damage responses. 35 Because ATM and ATR are essential regulators of two major checkpoint pathways required for DNA damage repair, 36 it is worth investigating whether the upregulation of IRF3 and IRF7 by HKST pretreatment stimulates DNA damage responses mediated by ATM or ATR signaling.
Conclusions
The TLR co-agonist HKST significantly alleviated damage induced by carbon ion radiation. The protective effects of HKST against heavy ion radiation involved inhibition of apoptotic pathways and stimulation of IRF3 and IRF7 signaling in multiple organs.
