Abstract
Objective
To evaluate the radiosensitivity effect of CpG oligodeoxyribonucleotide (ODN) 7909 on human epidermoid cancer strain-2 (Hep-2) cells
Methods
Toll-like receptor (
Results
Hep-2 cells were found to express
Conclusion
CpG ODN7909 enhanced the radiosensitivity of Hep-2 cells
Introduction
Laryngeal squamous cell carcinoma (LSCC), a highly aggressive malignancy with 5-year overall survival of approximately 61%, is one of the most common malignant otolaryngology tumours and ranks second among cancers derived from epithelial cells in the head and neck.1–3 Radiotherapy is a clinically important treatment method for patients with LSCC, however, the mode of LSCC treatment is changing and increasing numbers of clinicians focus on how to preserve the function and original structure of the larynx. Recent years have seen a trend toward improved post-treatment voice quality in patients with early-stage LSCC treated with external radiation.4–7
Radiotherapy is not only an adjuvant treatment following surgery, but also an important treatment for patients with locally advanced LSCC who can’t undergo surgical resection or who have a strong desire to protect the larynx.8,9 Various studies have shown that irradiation (IR) combined with conventional chemotherapeutic drugs, such as cisplatin and 5-fluorouracil (5-Fu), may achieve better results compared with IR alone.10–13 Nevertheless, there remains a risk of recurrence and metastasis in some patients with LSCC, for whom the efficacy of re-irradiation is poor and radiation resistance is prone to occur, resulting in reduced quality of life and shorter life span. 14 Hence, there is an urgent need to explore an effective radiosensitizer in the treatment of the patients with LSCC.
Recent studies have indicated that synthetic oligodeoxyribonucleotides containing unmethylated cytosine-phosphate-guanine motifs (CpG ODNs) may induce anti-tumour responses as immunoadjuvants in combination with other therapies, via interacting with toll-like receptors (TLRs) that play a fundamental role in the innate immune system.15–17 Moreover, a previous study by the present authors suggested that
In the present study,
Materials and methods
Cell culture and CpG ODN7909
Hep-2 cells purchased from the Chinese Type Culture Collection (CTCC; Wuhan, China) were maintained in the following culture medium: Minimum essential medium (MEM; BioWest, Loire Valley, France) supplemented with 10% foetal bovine serum (FBS; Gibco, ThermoFisher Scientific, Waltham, MA, USA), 100 U/ml penicillin G, and 100 μg/ml streptomycin at 37°C in a humidified atmosphere of 95% air and 5% CO2. CpG ODN7909 (5′-TCGTCGTTTTGTCGTTTTGTCGTT-3′) was obtained from Shanghai Sangon Biological Engineering Technology and Services Limited Company (Sangon, Shanghai, China), dissolved in phosphate buffer saline (PBS; 0.01 M, pH 7.4) and maintained at –20°C until use.
Western blotting
Whole cells were lysed in protein lysis buffer with 1 mM phenylmethylsulphonyl fluoride. Total proteins were harvested by centrifugation (14 000
Reverse transcription (RT) polymerase chain reaction (PCR)
Total RNA was extracted from 5 × 106 Hep-2 cells using TRIzol® Reagent (Invitrogen, Carlsbad, CA, USA), then reverse transcribed to cDNA using a PrimeScript™ RT Master Mix (TaKaRa, Dalian, China) according to the manufacturers' instructions. The cDNA was then amplified using the following TLR9 primer sequences: 5′-GCAAAGTGGGCG AGATGAGGAT-3′ (forward) and 5′-GA GTGAGCGGAAG AAGATGC-3′ (reverse), with AccuPower® 2X Greenstar™ qPCR Master Mix (Bioneer Corporation, Daejeon, South Korea). PCR was preformed using the LightCycler® 480 system (Roche Diagnostics, Mannheim, Germany) with the following thermal-cycling conditions: 5 min at 95°C for pre-denaturation, followed by 32 cycles of 30 s at 95°C for denaturation, 30 s at 56°C for annealing, 45 s at 72°C for elongation, and a final extension at 72°C for 10 min. The 578 bp reaction product was resolved by electrophoresis using a 1.5% agarose gel, stained with ethidium bromide, and photographed using an ultraviolet transilluminator.
Radiation exposure
Hep-2 cells were exposed to 6 MV X-rays using a linear accelerator (Varian Medical Systems, Palo Alto, CA, USA) under the source-to-skin distance of 100 cm, with a dose rate of 2.0 Gy/min. Graded irradiated doses, ranging from 0 to 10 Gy, were used in Hep-2 clonogenic survival assays. For all other experiments, 10 Gy radiation was employed.
Detection of cell viability via cell counting kit-8 (CCK-8)
Each well of 96-well plates were seeded with 6 × 103 Hep-2 cells in 100 μl of culture medium. Various concentrations of CpG ODN7909 (0, 5, 10, 20, 40 and 60 μg/ml) were added, and the cells incubated for 24 or 48 h at 37°C. Following CpG ODN7909 treatment, 10 μl of CCK-8 reagent (Dojindo Laboratories, Kami Mashiki-gun, Japan) was added to each well, and the cells incubated for a further 3 h at 37°C in the dark. Optical densities were then measured at 450 nm, and cell viability of CpG-treated cells was calculated as a proportion of the untreated cells, as follows: absorbance of CpG-treated cells/absorbance of untreated cells (0 μg/ml CpG ODN7909) × 100. Hep-2 cells were then seeded as before, and equally randomized into four groups, comprising: control group, CpG ODN7909-treated group (CpG group), irradiation group (IR group), and CpG ODN7909 + irradiation group (CpG + IR group). Based on the initial cell viability results, Hep-2 cells in the CpG and CpG + IR groups were treated with CpG ODN7909 at a final concentration of 10 μg/ml, and cells in all groups were cultured for 24 h. Following 24 h culture at 37°C, cells in the IR and CpG + IR groups were then exposed to 10 Gy radiation. A further 24 or 48 h following irradiation, cell viability was determined in all cells using the CCK-8 assay. All experiments were performed three times for each condition.
Clonogenic survival assay
Hep-2 cells were divided into two treatment groups and incubated for 24 h with or without CpG ODN7909 at a final concentration of 10 μg/ml. Cells were then irradiated with varying IR doses of 0, 2, 4, 6, 8, and 10 Gy, and harvested using 0.05% trypsin-EDTA solution for 1–2 min at 37°C. Cells were then re-seeded into 60 mm dishes at 500–3 000 cells per dish, as previously described, 19 in triplicate. Following incubation for 14 days, colonies were stained with crystal violet and fixed in methanol. The number of stained colonies (not less than 50 cells) was imaged and counted manually under a microscope. The clonogenic survival fraction (%) was calculated as follows: (irradiated cell colony numbers/unirradiated cell colony numbers) × 100. The radiobiological parameters (D0, N, Dq) were analysed with a single-hit multi-target model. D0 is the radiation dose that can reduce survival by a factor of 1/e in the exponential region of the curve, N is the extrapolation number or zero-dose extrapolate, Dq is the quasithreshold dose, Dq = D0 × ln N. 20
Cell cycle and apoptosis analysed by flow cytometry
Hep-2 cells, growing in logarithmic phase in six-well plates, were divided into four treatment groups (control group, CpG group, IR group, and CpG + IR group) as above. After 24 and 48 h exposure to 10 Gy radiation (IR groups only), cells of each group were harvested. The cell cycle distribution (proportion of cells in Gap 2/Mitotic phase [G2/M]) was detected by measuring the DNA content stained with propidium iodide (PI; BD Biosciences, San Jose, USA) in the presence of RNase, according to the manufacturer's instructions. Briefly, at 24 or 48 h post irradiation, Hep-2 cells were harvested using 0.05% trypsin-EDTA and centrifuged at 233
Measurement of interleukin (IL)-12 and tumour necrosis factor (TNF)-α release
Hep-2 cells divided into four treatment groups were cultured in six-well plates and treated as above. At 24 or 48 h post-radiation, the cell culture supernatants were collected and stored at –80°C. Human IL-12 Valukine™ enzyme-linked immunosorbent assay (ELISA) kit and Human TNF-alpha Valukine™ ELISA Kit (R&D Systems, Minneapolis, MN, USA) were used to measure secreted IL-12 and TNF-α levels, respectively.
Statistical analyses
GraphPad Prism software, version 5.0 (GraphPad Software, La Jolla, CA, USA) was used for data analyses and graph production. All data are presented as mean ± SD of at least three independent experiments. A single-hit multi-target model was used to fit colony counts to a clonogenic survival curve. Differences in cell viability, flow cytometric analyses or secreted cytokine levels between the treatment groups were determined by one-way analysis of variance (ANOVA) using Tukey’s honest significant difference (HSD) multiple comparisons test. The two treatment groups in the clonogenic survival assay were analysed using Student’s
Results
Expression of TLR9 in Hep-2 cells
Western blots and RT-PCR results showed that

Expression of
Cell viability
In Hep-2 cells treated with CpG ODN7909 at 0, 5, 10, 20, 40 and 60 μg/ml, cell viability was shown to be inhibited in a dose-and time-dependent manner (

Effects of CpG ODN7909 (CpG) and irradiation (IR) on cell viability of Hep-2 cells quantified using cell counting kit-8 assays. (a) Cells were treated with CpG ODN7909 at concentrations of 0, 5, 10, 20, 40 and 60µg/ml and CCK-8 was added at 24 and 48 h following treatment; and (b) cell viability in different treatment groups (control, 10 µg/ml CpG ODN7909 only, 10 Gy irradiation at only, or 10 µg/ml CpG ODN7909 with 10 Gy irradiation). Data presented as mean ± SD; ★
Clonogenic survival analysis
Hep-2 cell dose-survival curves were fitted using a single-hit multi-target model (Figure 3). In Hep-2 cells treated with CpG ODN7909 plus IR, there were markedly lower values (

Dose-survival curves fitted using a multi-target single-hitting model to demonstrate the impact of CpG ODN 7909 on irradiation (IR) of Hep-2 cells. Hep-2 cells were irradiated with 6 MV X-rays at 0, 2, 4, 6, 8, and 10 Gy with or without CpG ODN 7909 at 10 µg/ml (CpG + IR group). CpG + IR showed decreased clonogenic survival compared with IR alone (
Comparison of radiosensitivity in Hep-2 cells treated with irradiation (IR) alone (6 MV X-rays at 10 Gy; IR group), or with irradiation plus unmethylated cytosine-phosphate-guanine motif oligodeoxyribonucleotides (CpG ODN)7909 (CpG + IR group)

The
N, extrapolation number; D0, mean death dose; Dq, quasi field dose; SER, sensitivity enhancement ratio.
Cell cycle arrest at G2/M phase
Proportions of Hep-2 cells in G2/M phase cell cycle arrest were significantly different between the four treatment groups (untreated control, CpG ODN 7909 only, IR only, or CpG ODN 7909 plus IR;
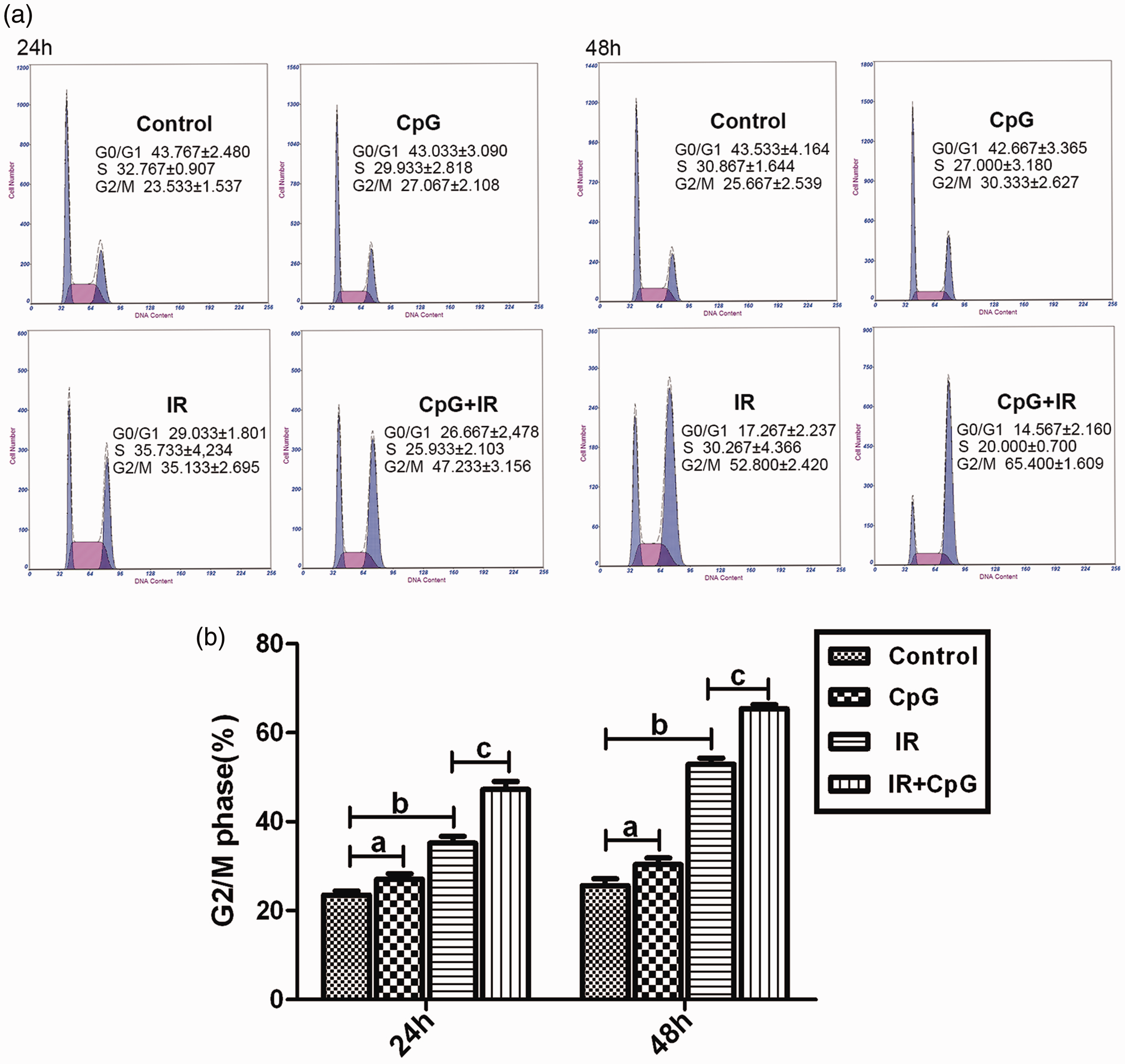
Hep-2 cell cycle assessment by flow cytometry, showing: (a) Representative flow cytometry results at 24 and 48 h following no treatment (control) or treatment with CpG ODN7909 alone (CpG group), irradiation alone (IR group) or CpG plus IR; (b) The proportion of cells in G2/M phase in the four treatment groups. Data represent the mean of three independent experiments, and are presented as mean ± SD; a
Increase of IR-induced cellular apoptosis
Significant differences were observed in Hep-2 cell apoptosis index between the four treatment groups (

Analysis of apoptotic Hep-2 cells using annexin V/PI and flow cytometry: (a) Representative flow cytometry results at 24 and 48 h following no treatment (control) or treatment with CpG ODN7909 alone (CpG group), irradiation alone (IR group) or CpG plus IR showing B1, dead cells; B2, late apoptosis; B3, viable cells; and B4, early apoptotic cells; (b) Apoptotic fraction of cells in the four different treatment groups; Data presented as mean ± SD; ano statistically significant difference (
Release of IL-12 and TNF-α
A significant difference was observed in IL-12 concentration between the four treatment groups (

Interleukin (IL)-12 and tumour necrosis factor (TNF)-α release by Hep-2 cells measured by enzyme-linked immunosorbent assay: (a) Cell culture supernatant IL-12 concentrations in four groups at 24 and 48 h following no treatment (control) or treatment with CpG ODN7909 alone (CpG group), irradiation alone (IR group) or CpG plus IR; (b) Cell culture supernatant TNF-α concentrations in the four treatment groups; Data presented as mean ± SD; ⋆
Discussion
The application of CpG ODNs, which recognize TLR9, has shown a remarkable degree of synergy with conventional remedies for malignant tumours.22–25 In the past,
The occurrence, infiltration and metastases of tumours is related to cell proliferation, and CCK-8 can be used to evaluate cell proliferation dynamics.
32
The present results showed a decrease in CCK-8 after Hep-2 cells were exposed to CpG ODN7909 for 24 and 48 h, indicating that cellular viability was inhibited, and this inhibition was shown to be in a dose-dependent with increasing CpG ODN7909 concentrations. In addition, the subtoxic dose of CpG ODN7909 (at 10 μg/ml) combined with 10 Gy X-ray radiation was shown to significantly enhance the decrease in cell viability compared with 10 Gy X-ray radiation alone. Consistent with the present results, the authors previously published study demonstrated that CpG ODN1826 alone could delay the growth of Lewis lung carcinoma in mice, and the combined inhibitory effect of CpG ODN1826 and X-ray on Lewis lung cancer was greater than X-ray radiation alone.
33
The present study also employed clonogenic survival analysis to investigate the role of CpG ODN7909 on radiation sensitivity in Hep-2 cells, and showed that CpG ODN7909 combined with radiation was able to significantly inhibit Hep-2 cell colony formation. Compared with the IR group, D0 of the combined treatment group was 1.545 Gy versus 1.892 Gy, the N value was 2.112 versus 4.394, and Dq value was 1.155 versus 2.801, indicating that CpG ODN7909 could enhance the
Cell cycle distribution is well documented to relate to radiosensitivity, with cells being the most sensitive to radiation during the G2/M phase and the least sensitive at the end of S phase. 34 Therefore, mediating cell cycle progression into G2/M phase would be an effective approach to increase the radiosensitivity of tumours. CpG ODN has been reported to directly act on the human lung carcinoma cell line 95D by promoting cancer cells to move into the G2/M phase, and the signalling pathway was shown to be mediated by TLR9. 35 Consistent with these results, the present study showed that CpG ODN7909 significantly potentiated X-ray induced cell-cycle arrest, and the number of cells arrested at G2/M phase was significantly increased in cells treated with CpG ODN7909 and X-ray combined, compared with X-ray alone.
Apoptosis induced by irradiation is vital in the use of X-rays to eliminate tumour cells, and it is widely recognized that irradiation-induced apoptosis may be used for assessing the sensitivity of tumour cells to irradiation, with an increased apoptotic rate indicating higher tumour cell radiosensitivity. 36 In the present study, CpG ODN7909 alone at 10 μg/ml could not directly induce cellular apoptosis, but the apoptotic index was significantly increased when CpG ODN7909 was combined with radiation. The mechanism may be that CpG ODN7909 combined with radiation altered the cell cycle distribution, with cell arrest at G2/M phase, in which the cells were more sensitive to radiation and were more likely to enter the apoptotic pathway.
Furthermore, the present study showed that at 24 and 48 h following irradiation, IL-12 and TNF-α secretion by Hep-2 cells was significantly increased in cells treated with CpG ODN7909 and X-rays combined compared with X-rays alone. IL-12 is known to play a significant role in inhibiting tumour development and metastasis, 37 and TNF-α is a cell factor with strong direct antitumour activity, that is reported to be closely involved with radiation sensitivity. 38 TNF-α has been shown to inhibit the proliferation of various tumour cells, and to induce apoptosis.39,40 One study showed that IL-12 and TNF-α could be up-regulated in the human glioma cell line (CHG-5) when these cells were treated with a combination of CpG ODN and ß-rays, which apparently inhibited cell clonogenic survival. 41 Similarly, the present results showed that secretion of IL-12 and TNF-α had an increasing trend following treatment with CpG ODN7909. The secretion of IL-12 and TNF-α was increased more significantly following treatment with CpG ODN7909 and X-rays combined. The present authors hypothesize that Hep-2 cells can secrete these factors through autocrine mechanisms, and these factors may participate in improving the roles of CpG ODN7909 on radiosensitivity of tumour cells.
In conclusion, the present results showed that CpG ODN7909 combined with X-ray radiation decreased cellular clonogenic survival and increased cellular apoptosis, percentage of cells at G2/M phase and secretion of specific cytokines, compared with X-ray radiation alone, suggesting that CpG ODN7909 could enhance the radiosensitivity of Hep-2 cells
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research was supported by the Jinshan District Health and Family Planning Commission Project (No. JSKJ-KTQN-2014-01).
