Abstract
Objective
This study aimed to investigate the effects of neuromuscular blocking drugs on the viability of human umbilical vein endothelial cells (HUVECs) and to investigate whether they cause vascular complications due to cell proliferation.
Methods
HUVECs were cultivated with 5% CO2 at 37°C in a predefined supplemented medium over 7 days until confluence of cell monolayers. Assays were conducted during the exponential growth phase. Suxamethonium chloride, vecuronium bromide, atracurium besylate, and rocuronium bromide were used at concentrations of 10–5, 10–6, and 10–7 M in proliferation assays in which cells were incubated with these drugs for 24, 48, and 72 hours. All experiments were performed in four replicates.
Results
The neuromuscular blocking drugs used had comparable effects on the survivability of HUVECs. Overall, no significant difference was observed in the survivability of HUVECs in a dose-dependent manner after exposure to the study drugs. However, some significant differences in the viability of HUVECs were found among the different measurement times.
Conclusions
The findings of the current study support the safety of the studied neuromuscular blocking drugs in clinically relevant concentrations regarding their effects on endothelial cell proliferation.
Keywords
Introduction
Neuromuscular blocking drugs (NMBDs) interfere with the transmission of impulses from motor nerves to skeletal muscle fibers at the neuromuscular junction, resulting in blockade of motor activities. NMBDs are categorized as depolarizing and non-depolarizing agents. These agents are structurally associated with acetylcholine (ACh) containing at least one positively charged quaternary ammonium radical (–N+[CH3]3) binding to the nicotinic receptor. 1 Depolarizing agents show their effects by acting as an ACh agonist on nicotinic receptors, but they cause prolonged depolarization at the motor end-plate, resulting in insensitivity of ion channels for further action potentials. Suxamethonium chloride (succinylcholine) is the only depolarizing NMBD used in anesthetic practices for short-term paralysis to facilitate tracheal intubation or electroconvulsive therapy. 2 However, non-depolarizing agents show their effects by directly binding to nicotinic receptors in a competitive manner against ACh, and this prevents neurotransmitter receptor binding. 1
Non-depolarizing agents are used in the balanced anesthesia technique to control undesired movements of the patient and to facilitate surgical and anesthetic procedures. 3 Non-depolarizing agents are associated with similar aminosteroidal 3-dimensional structures. However, experimental studies on different species have shown that these agents are effective in cardiovascular systems regarding various aspects, including positive inotropic/chronotropic effects and hemodynamic alterations.4,5 Currently, three non-depolarizing NMBDs, atracurium, vecuronium, and rocuronium, are widely used in anesthetic practice. 6
Cardiovascular effects related to non-depolarizing NMBDs have significant implications in anesthetic management of patients requiring prolonged neuromuscular blockade. Cardiovascular effects include increased heart rate and positive inotropic effects for pancuronium, vecuronium, rocuronium, and mivacurium. The positive inotropic effect related to pancuronium occurs at higher concentrations. These effects may be attributed to direct stimulation of β-adrenoceptors or pathological conditions, such as hypertension, where β-blocking agents, are generally used.5,7,8 Cardiovascular effects of NMBDs on the isolated atrium have been studied on rats. 8 Because endothelial cells play a significant role in regulating hemostasis, 9 cardiovascular effects could be mediated by the endothelium. Endothelial cells can be obtained from the human umbilical vein. Human umbilical vein endothelial cells (HUVECs) are used for hemostasis, angiogenesis, and immunity as an in vitro model. 10
This study aimed to investigate the effects of NMBDs on endothelial cell proliferation. Specifically, this study aimed to examine the effects of suxamethonium chloride, vecuronium bromide, atracurium besylate, and rocuronium bromide on the viability of HUVECs and to investigate whether vascular proliferation is associated with complications.
Materials and methods
Drugs
The effects of suxamethonium chloride (Lysthenon forte 2% 100 mg; Actavis Drugs Company, Istanbul, Turkey), vecuronium bromure (Norcuron 4 mg/ampoule; Schering Plough Company, MSD, Istanbul, Turkey), atracurium besylate (CURAMED Pharma GmbH, Karlsruhe Germany), and rocuronium bromide (Esmeron 50 mg/5 mL/ampoule, Esmeron® 50 mg/5 mL; Organon, Oss, Holland) were studied.
Cell culture
HUVECs were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). The cells were maintained in a gelatin-coated 75/cm2 flask in M199 (Sigma, St. Louis, MO, USA) supplemented with 20% fetal bovine serum (Sigma), 100 units/mL penicillin, 100 mg/mL streptomycin, and 3 ng/mL basic fibroblast growth factor (Thermo Fisher Scientific, Waltham, MA, USA). HUVECs were cultivated with 5% CO2 at 37°C in predefined supplemented medium over 7 days until confluence of cell monolayers. The assays were conducted during the exponential growth phase.
Cell proliferation assays
Culture media containing cleavage of tetrazolium salt with sodium 3´-[1-(phenylaminocarbonyl)-3,4-tetrazolium]-bis (4-methoxy-6-nitro) benzene sulfonicacid hydrate (XTT) labeling reagent (Roche, Manheim, Germany) was used to assess cell viability. This assay relies on cleavage of tetrazolium salt to formazan by cellular enzymes. Any expansion in viable cells leads to an increase in the total activity of mitochondrial dehydrogenase enzymes in the sample, resulting in an increased amount of formazan dye correlating with the number of metabolically active cells in the culture medium. The formazan dye was measured by a scanning multi-well spectrophotometer using absorbance quantification of dye solution at 450 nm. The cells were inoculated into 96-well microtiter plates coated with gelatin at a concentration of 1 × 104 cells/mL, with a final volume of 100 µL per well.
Suxamethonium chloride, vecuronium bromide, atracurium besylate, and rocuronium bromide were used at the concentrations of 10−5, 10−6, and 10−7 M in proliferations assays. The cells were then incubated with these drugs for 24, 48, and 72 hours in a humidified atmosphere with 5% CO2 at 37°C. Following incubation, 10 μL XTT labeling reagent was added to each well containing culture medium (10 μL). The absorbance of the samples was then measured at 450 nm against a control by using a micro-plate reader (Microplate Photometer Multiskan FC; Thermo Fisher Scientific). Equal volumes of culture medium and XTT labeling reagent (10 μL of XTT labeling reagent/100 μL of culture medium) were added to a blank well for use as a background control (absorbance of culture medium plus XTT without cells). The absorbance measurements were performed 2 hours after initiation of the tetrazolium reaction. All experiments were performed in four replicates.
The viability of the control was defined as 100%. The viability of HUVECs that were incubated with the drugs used in the study was calculated compared with their negative control. The optical density (OD) of samples was compared with a negative control to obtain a viability score using the following equation: viability score (%)=[OD450 (sample)/OD450 (negative control)]×100.
Statistical analysis
Statistical analysis was performed using Statistical Package for Social Sciences software (version 22.0; IBM Corp., Armonk, NY, USA). Data are presented as the mean ± standard deviation. Among multiple groups, mean viability scores were compared by using analysis of variance with Tukey’s post hoc test. A p value <0.05 was considered as statistically significant.
Ethics
Ethics committee approval was not required because the study was carried out on HUVECs. Because this was not a human study, consent did not need to be obtained from any patients or parents/carers of patients.
Results
Figure 1 shows the viability of HUVECs at 24, 48, and 72 hours after administration of suxamethonium chloride, vecuronium bromide, atracurium besylate, and rocuronium bromide at the concentrations of 10−5, 10−6, and 10−7 M. We found that the effects of the drugs used in this study on the viability of HUVECs were comparable. After administration of the drugs, there was no significant difference in the viability of HUVECs in relation to dose.
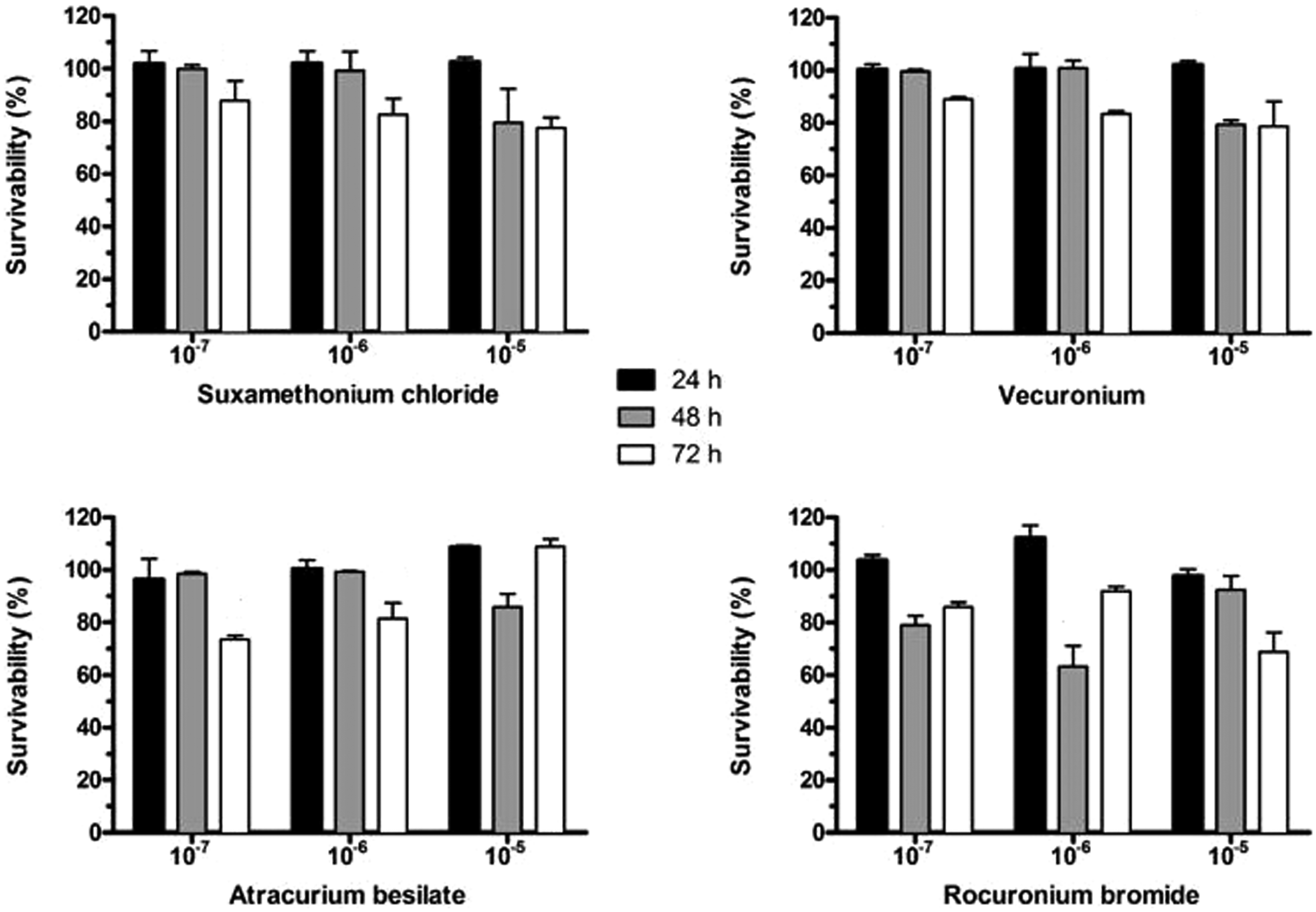
Effect of suxamethonium chloride, vecuronium, atracurium besylate, and rocuronium bromide on survivability of human umbilical vein endothelial cells in a dose- and time-dependent manner. Data are expressed as mean ± standard deviation.
After administration of suxamethonium chloride and vecuronium bromide, the viability of HUVECs at 72 hours was significantly lower compared with that at 24 and 48 hours at concentrations of 10−6 and 10−7 M (all p<0.05). However, at a concentration of 10−5 M, the viability of HUVECs was significantly higher at 24 hours compared with that at 48 and 72 hours (both p<0.05). After administration of atracurium besylate, the viability of HUVECs at 72 hours was significantly lower compared with that at 24 and 48 hours at concentrations of 10−6 and 10−7 M (all p<0.05). However, the dose of 10−5 M atracurium resulted in significantly lower viability of HUVECs at 48 hours compared with that at 24 and 72 hours (both p<0.05). After administration of rocuronium bromide, the viability of HUVECs was significantly higher at 24 hours compared with that at 48 and 72 hours at a concentration of 10−7 M (both p<0.05). The dose of 10−6 M rocuronium bromide resulted in significantly higher viability of HUVECs at 24 hours compared with that 48 and 72 hours (both p<0.05). Furthermore, the dose of 10−5 M resulted in significantly lower viability of HUVECs at 72 hours compared with that at 24 and 48 hours (both p<0.05).
Discussion
Suxamethonium, which is a short-acting agent with rapid onset of action, provides an excellent condition for intubation. 11 Non-depolarizing NMBDs, including vecuronium, atracurium, and rocuronium, block the ACh action at postsynaptic receptors that are localized on the end-plate of motor nerves in a competitive manner. 12 In experimental animal studies, the effects of NMBDs on the heart, coronary arteries, and vascular tone have been investigated.13–15 However, only one study evaluated the effects of NMBDs on proliferation of HUVECs. 16 The cytotoxic and direct effects of suxamethonium chloride, vecuronium bromide, atracurium besylate, and rocuronium bromide on the endothelium were investigated in HUVECs in this previous study. After administration of suxamethonium chloride, vecuronium bromide, atracurium besylate, and rocuronium bromide at concentrations of 10−5, 10−6, and 10−7 at 24, 48, and 72 hours, cytotoxicity was comparable among the drugs studied. The authors of this previous study concluded that there was a dose-dependent increase in cytotoxicity in all of the drugs examined. However, there was no consistent change in cytotoxicity of the drugs at the different measurement times.
Melnikov et al. 8 studied the cardiac effects of non-depolarizing NMBDs in isolated rat atrium. These authors found that pancuronium caused a positive chronotropic effect, while vecuronium and rocuronium caused a positive inotropic effect. These findings suggested that cardiac effects of these agents were induced at high concentrations. Similarly, in a study by Gursoy et al., 7 cardiac effects of non-depolarizing NMBDs were examined in isolated rat atrium and the results were consistent with the relevant literature. 8 This previous study suggested that such effects could be particularly important in hypertensive patients on β-blockers in clinical practice. All cardiovascular effects are caused by vascular tone, which is affected by vasoactive hormones, sympathetic nervous system activity, and local humoral factors. Generally, the endothelium has an important role in regulation of local vasoactive factors. Moreover, the effects of non-depolarizing NMBDs were investigated in isolated coronary segments of pigs in another study. 15 This previous study showed that these drugs (atracurium, rocuronium, and vecuronium) had no effect on the vasomotor tone of coronary arteries at clinically relevant concentrations. However, the authors concluded that high-dose pancuronium had an antimuscarinic effect in vascular smooth muscle. In our study, there was no significant difference in HUVEC viability at 24, 48, and 72 hours after dose-dependent administration of suxamethonium chloride, vecuronium bromide, atracurium besylate, and rocuronium bromide at concentrations of 10−5, 10−6, and 10−7 M. In another study, the effects of atracurium, cisatracurium, and mivacurium on HUVECs were investigated. 16 This previous study showed that atracurium and cisatracurium attenuated HUVEC proliferation, while mivacurium had no effect. Rieder et al. 17 examined the effects of mivacurium and cisatracurium on apoptosis in HUVECs. These authors found cisatracurium-induced apoptosis and observed that mivacurium had no effect on apoptosis. Apoptosis was likely induced in HUVECs by cisatracurium because of the reactive nature of acrylate metabolites generated during Hoffmann elimination. In our study, atracurium at doses of 10−7 and 10−6 M decreased proliferation at 72 hours compared with that at 24 and 48 hours, while atracurium at the dose of 10−5 increased proliferation at 24 hours. However, no significant difference was found in the effects of drugs on HUVEC viability in general.
Jeong et al. 13 reported that non-depolarizing NMBDs had a dose-dependent protective effect against free radical injury in isolated rabbit abdominal aorta endothelium. This finding suggests that vecuronium and rocuronium could act as superoxide anion scavengers.
Conclusion
In this in vitro study, we investigated the effects of NMBDs on proliferation of endothelial cells. Our study showed that there was no significant decrease in viability of endothelial cells with respect to drug- and dose-dependency. In conclusion, the results of the present study support the safety of studied NMBDs, including suxamethonium chloride, vecuronium bromide, atracurium besylate, and rocuronium bromide, in clinically relevant concentrations regarding their effects on endothelial cell proliferation.
