Abstract
Objective
This study was performed to evaluate a new type of autologous muscle tamponade to repair dura mater that has undergone dural defects to prevent cerebrospinal fluid leakage or subcutaneous fluid accumulation.
Methods
Three hundred thirty-two patients who underwent retrosigmoid craniotomy were selected and divided into two groups: bone window craniotomy and bone flap craniotomy. Each group was further divided into two groups: artificial dura repair and autologous muscle repair. We then analysed the incidence of postoperative cerebrospinal fluid leakage or subcutaneous fluid accumulation and compared the effects of the two repair methods.
Results
For all patients, autologous muscle repair of the dura mater had a lower incidence of cerebrospinal fluid leakage than artificial dura mater repair, especially in patients with craniotomy.
Conclusions
Subdural craniotomy of the bone window is more effective than conventional methods in preventing cerebrospinal fluid leakage.
Keywords
Introduction
Cerebrospinal fluid leakage is one of the most common complications that occurs after neurosurgery. The entry of bacteria into the subarachnoid space from the leak can have serious consequences,1–3 especially in patients treated with the posterior sigmoid approach. Certain special anatomical features determine the incidence of cerebrospinal fluid leakage which in foreign countries is reportedly as high as 2% to 30% in patients with acoustic neuroma.1,3 The cerebrospinal fluid leaks from the dural incision, enters the tympanic cavity through the mastoid air chamber, and either moves into the nasopharynx through the Eustachian tube to form a cerebrospinal fluid rhinorrhoea or forms an ear leak through the ruptured tympanic membrane. It can also form a notch leak through the knife edge and, in some patients, form a subcutaneous effusion.
The root cause of cerebrospinal fluid leakage is destruction of the integrity of the dura mater. Therefore, restoring the integrity of the dura mater is a key factor in preventing cerebrospinal fluid leakage. 3 Gelatine sponges and autologous tendon fascia films have been used to repair dural defects. Various artificial materials are now widely used to repair the dura mater,2,4–6 with different techniques yielding different therapeutic effects. One approach involves acquisition of the fascia in the posterior sigmoid. However, this method is difficult, and muscle is easier to obtain. The opening of the mastoid airway during the sigmoid sinus approach and the use of artificial repair materials increase the chance of infection, especially if treatment is delayed. If left unhealed, a second surgery is sometimes required to remove foreign bodies. Given the possibility of such complications, we focused our research on the use of autologous tissue to repair the dural defect. Advantages of this technique is that autologous tissue is easier to obtain, the repair operation is simpler to perform, no additional surgical incision is required, and the muscle flap approach ensures minimal surgical trauma.
Materials and methods
Patients
In total, 360 patients with dural defects treated by the posterior sigmoid sinus approach from 2012 to 2018 were reviewed. The patients were divided into a bone window craniotomy group (n = 130) and a bone flap craniotomy group (n = 230). Each of these two groups was then further divided into an artificial dura mater repair group and muscle flap repair group (Table 1).
Data of patients who underwent muscle flap repair and artificial dura mater repair after bone window craniotomy or bone flap craniotomy.

Data are presented as n, n (%), or mean ± standard deviation.
M, male; F, female.
Surgical procedure
The non-rigid sutured dura mater was repaired by two methods. The first involved the use of sutureless artificial dura mater for the repair, whereas the second involved the use of a muscle strip larger than the dural defect to fill the defect under the dura mater. The edges of the muscle and defective dura mater were then continuously and tightly sutured. The intraoperative mastoid air chamber was closed with bone wax, and a subcutaneous drainage tube was inserted and retained for 1 day postoperatively. A computed tomography scan was performed 1 week after the surgery, and its results, along with the clinical manifestations, were used to determine the occurrence of cerebrospinal fluid leakage or subcutaneous effusion (Figure 1).

Illustration of muscle flap repair. (a) Diagram of muscle flap repair. The black bar shows cerebrospinal fluid flow. (b) Free muscle flap before subdural packing. (c) Subdural muscle after subdural packing. The black bar shows the fixed muscle flap.
Ethical standards
All procedures performed in this study were in accordance with the ethical standards of the recommendations of the Shandong University institutional review board, and informed consent was obtained from all individual participants included in the study.
Statistical analysis
The frequency of occurrence of cerebrospinal fluid leakage or subcutaneous effusion in each group of patients was assessed. The data were analysed using the parallel chi-square test. SPSS version 16.0 (SPSS Inc., Chicago, IL, USA) was used for the statistical analysis.
Results
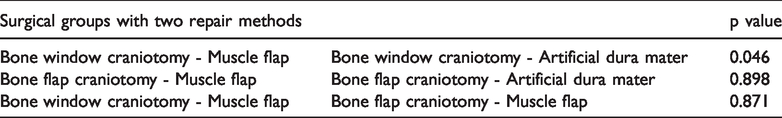
The statistical comparisons between patients with cerebrospinal fluid leakage or subcutaneous effusion who underwent either muscle flap repair or artificial dura mater repair are summarized in Table 2. Patients who underwent bone window craniotomy with muscle flap repair had a significantly lower incidence rate of cerebrospinal fluid leakage or subcutaneous fluid accumulation than patients who underwent manual dural repair (p = 0.046 ± 0.05). Artificial dura mater may hinder the adhesion of subcutaneous muscle to dura mater, and a muscle flap may increase the incidence of cerebrospinal fluid leakage or subcutaneous fluid accumulation after craniotomy. In patients with craniotomy, the two repair methods were similarly effective in repairing the dura mater. There was no significant difference in the rate of cerebrospinal fluid leakage between the craniotomy group who underwent artificial dura mater repair and the group who underwent muscle flap repair. This indicates that the dural defect was effectively repaired by the muscle flap subdural technique. The effect was stable and had little association with the method of closing the skull. From the perspective of postoperative cerebrospinal fluid leakage and subcutaneous effusion, bone window craniotomy showed the lowest levels (2.53%) of leakage and effusion, while bone window craniotomy showed the highest levels (11.76%) relative to other methods. Thus, we were able to demonstrate that the incidence of cerebrospinal fluid leakage and subcutaneous fluid accumulation is low when an autologous muscle repair technique is used.2,6 The patients’ demographic information is summarized in Table 1.
Comparisons between patients with cerebrospinal fluid leakage or subcutaneous effusion who underwent either muscle flap repair or artificial dura mater repair.
Discussion
Cerebrospinal fluid leakage is a common complication associated with the sigmoid sinus approach. Although most cases can be treated, a few may have serious consequences due to intracranial infections. Factors that cause cerebrospinal fluid leakage include destruction of the dura mater integrity and opening of the mastoid air chamber. Given that a compromised dura mater is an underlying cause of cerebrospinal fluid leakage, 7 restoring the integrity of the dura mater is a critical step in preventing cerebrospinal fluid leakage or subcutaneous fluid accumulation. The traditional repair method focuses more on closure of the mastoid air chamber and inner ear canal. To obtain satisfactory repair outcomes, a larger flap range is often necessary, and more autologous tissue is consequently needed to fill the mastoid air chamber or the internal auditory canal. In some cases, bioadhesive aids are also required.8–12
Artificial dura mater has been widely used in dural repair because of its high biocompatibility, and it has largely solved the problem of autologous dural defects. However, the water-tightness between artificial dura and autologous dura is poor, resulting in a high incidence of cerebrospinal fluid leakage. We focused on and restored the integrity of the dura mater by repairing it with subdural tamponade of an autologous muscle flap rather than by use of an epidural covering, and we achieved satisfactory results. Our method involved the use of a free muscle flap twice as large as the dural defect, and the flap was placed under the defective dura mater. We sutured the muscle ends at both ends of the dura mater, then used a continuous suture to tightly suture the muscle flap. These edges fixed the muscles under the dura mater and closed the region. The muscle window repair group showed a significantly lower frequency of cerebrospinal fluid leakage or subcutaneous fluid accumulation after muscle repair than the manual dural repair group (p = 0.046 ± 0.05).
When compared with the traditional repair method, our most notable improvement was to place the repair material under the dura mater, which is more consistent with the requirements for cerebrospinal fluid circulation. The muscle flap was placed under the dura mater and tightly sutured to its edge. Under the action of intracranial pressure, the muscle was tightly pressed against the edge of the dura mater and the seal was enhanced. In fact, higher intracranial pressure provides a better seal. During epidural repair, the repair material was separated from the dura mater by the action of intracranial pressure. Once the cerebrospinal fluid flows out, the intracranial pressure drops, further increasing the separation of the epidural repair material and the dura mater and thereby aggravating the cerebrospinal fluid leak to form a vicious circle. We also used the muscle flap instead of the fascia as the repair material. Compared with the fascia, the muscle tissue is softer and more deformable, and it is more suitable to fix the dural defect and enhance the sealing effect. Muscles slowly become mechanized in the surgical area, gradually adhering to the dura mater and arachnoid, thus facilitating complete closure of the dura mater. Our use of muscle as a repair material also made it easy to acquire and use surgical materials because no special tools are required to handle muscle.
The following items should be given attention when using muscle to repair dural defects.
The muscle flap must be placed under the dura because this is the only way to enable the sealing effect under the action of intracranial pressure. The muscle flap must be larger than the dural defect. Our experience is that the edge of the muscle flap should be at least 0.5 cm larger than the edge of the dural defect to increase the contact area between the repair material and the dura. The thickness of the muscle flap should be 0.5 to 1.0 cm to ensure that the muscle flap can herniate into the dura defect to function in a manner similar to a rubber bottle stopper, thus closing the gap.
Postoperative intracranial pressure control should also be performed irrespective of the material used to repair the dural gap. This is also important for temporary closure or self-repair. Increased intracranial pressure is a key contributor to repair failure; therefore, the following two points should also be noted.
During the operation, intracranial pressure can reduce the pulling on the brain tissue and injury to the reflux vein. By this action, it can actively eliminate cerebral oedema after the operation, improve the local cerebrospinal fluid circulation, and reduce the intracranial pressure. During the operation, the cerebral cisterna near the operative area should be opened to promote cerebrospinal fluid circulation. Patients should be encouraged to get out of bed and move around at an early stage after surgery to promote cerebrospinal fluid circulation.
Our conservative treatments for postoperative cerebrospinal fluid leakage or subcutaneous effusion have achieved good results with no requirements for reoperation. We used three methods and saw promising results. For nine patients with a small amount of effusion, we used a combination of dehydration drugs, local compression, and an elevated head position; among these nine patients, seven were cured within 1 week and two were treated by lumbar drainage for 5 days after the initial week. For six patients with a large amount of effusion, we used a combination of local drainage and compression where the cerebrospinal fluid had accumulated. For the bone window craniotomy, after 3 to 4 days of continuous drainage, the subcutaneous muscle, dura mater, and muscle flap had adhered tightly, allowing a valve adhesion to form and the cerebrospinal fluid leakage to be repaired. For the last patient, because repositioning of the skull cannot result in the formation of an adhesion with the dura mater in a short time, the drainage time was extended so that the subdural muscle flap and the dura mater could completely adhere.
Conclusion
For dural repair, we found that subdural craniotomy of the bone window is better than conventional methods. After craniotomy, repair of the dural ciliary muscle and conventional repair have similar effects. When combined with bone wax closure and short-term subcutaneous drainage in the mastoid air chamber, this technique can effectively prevent the occurrence of cerebrospinal fluid leakage.
