Abstract
Background
Ectodermal dysplasia is a congenital genetic disorder with a prevalence of 1:10,000 to 1:100,000. The clinical features of ectodermal dysplasia include sparse hair, missing teeth, and abnormal development of the skin, sweat glands, and other tissues and organs. Since 1985, dentists have used implants to correct tooth defects in patients with ectodermal dysplasia with reasonable success rates. However, there is still no widely accepted treatment for the oral defects caused by this disease.
Conclusion
The patient’s mouth was restored through the use of bone augmentation and soft tissue grafting techniques, enabling appropriate function and appearance.
Keywords
Core tips
The anterior alveolar ridge of patients with ectodermal hypoplasia is rudimentary and underdeveloped, resulting in a severely reduced volume. To successfully carry out implant restoration in patients with ectodermal hypoplasia, it is necessary to restore the morphology and function of the edentulous area as well as ensure precise use of soft tissue grafting to improve the patient’s appearance. In this case, a bone grafting technique was combined with custom-made guard-free gingival flap grafting and a rootward repositioning flap, effectively solving the problems of insufficient bone volume, a shallow vestibular sulcus, and insufficient thickness of the keratinized gingiva in the implant area.
Introduction
Ectodermal dysplasias (EDs) are a varied group of malformations that are associated with genetic syndromes characterized by hypoplasia, aplasia, and dystrophy of ectodermal structures. 1 With respect to oral manifestations, patients with ED display abnormalities of the quantity, shape, and structure of the primary and secondary dentition.1,2 Much of the published literature on implant restoration associated with ED syndromes consists of case reports; few systematic statistical analyses of large samples have been performed. 3 Among the cases in which implantation was used to repair ED syndromes, most of the controversy focused on the choice of implant timing. Some scholars believe that implant repair can be initiated at the age of 3 to 5 years.4,5 Other scholars have indicated that implant placement should not be performed while the patient is still undergoing growth and development. 6 In one study, the implant retention rate of immature patients with ED syndrome (patients aged ≤16 years) was compared with that of adult patients, revealing no statistically significant difference between the two groups. 7 A general consensus recently indicated that the combination of several implant techniques is ideal for restoring oral defects in patients with ED syndrome.8,9
This case provides clinical guidance for clinicians who treat patients requiring extensive bone augmentation. In clinical practice, extensive bone augmentation is prone to tissue dehiscence and membrane exposure, which can lead to bone augmentation failure. This case demonstrates the need to treat such patients with strict adherence to the principles of bone augmentation while taking into account soft tissue morphology and function in order to achieve a relatively successful repair.
Case presentation
Chief complaint
A 19-year-old female patient was referred to the Dental Department of Zhejiang Chinese Medical University.
History of presenting illness
The patient described herself as having congenital ED syndrome with congenital absence of the majority of her teeth.
Physical examination
Examination of the oral cavity showed survival of teeth 65, 26, 36, 37, 85, and 46, whereas the remaining teeth were congenitally missing. Thin gingiva and severe alveolar ridge atrophy were noted (Figure 1).

Preoperative intraoral photographs of the patient. (a) Preoperative maxillary intraoral photograph. (b) Preoperative mandibular intraoral photograph. (c) Preoperative intraoral frontal photograph.
Imaging examination
Cone-beam computed tomography revealed severe atrophy of the alveolar bone throughout the entire mouth and severely reduced bone width and height in the maxillary and mandibular edentulous areas (Figure 2).

Preoperative imaging of the patient. (a) Preoperative stomatographic tomography. (b) Preoperative cone-beam computed tomography image.
Final diagnosis
The patient was diagnosed with dentition and bone defects due to ED syndrome.
Treatment
Initial surgery: maxillary augmentation and right maxillary sinus external lift
Blood was drawn from the patient to produce a concentrated growth factor (CGF) membrane (Figure 3(a)). Preoperative medications included routine use of an oral anti-inflammatory drug (cefradine capsule), anti-swelling drug (dexamethasone acetate tablet), and analgesic drug (loxoprofen sodium tablet). The patient’s oral cavity was rinsed twice with a chlorhexidine solution for 3 to 5 minutes per rinse. Routine disinfection of the surgical area was then completed, sterile towels were placed, and anesthesia with epinephrine and lorazepam was provided. A skin incision was made to expose the mandible at the 33 to 43 tooth position, specifically exposing the chin. A bone block drill was used to obtain two annular bone blocks approximately 7 to 8 mm in diameter and 3 mm in thickness (Figure 3(b)). Next, 4/0 absorbable suture material was used to complete a double layer of tight sutures to close the wound. The two bone blocks were used to prepare bone chips using a bone chip drill. The bone chips from the chin were mixed with bone powder (Bio-Oss; Geistlich Pharma AG, Wolhusen, Switzerland) using two units of 0.5 g each at a ratio of 1:1.
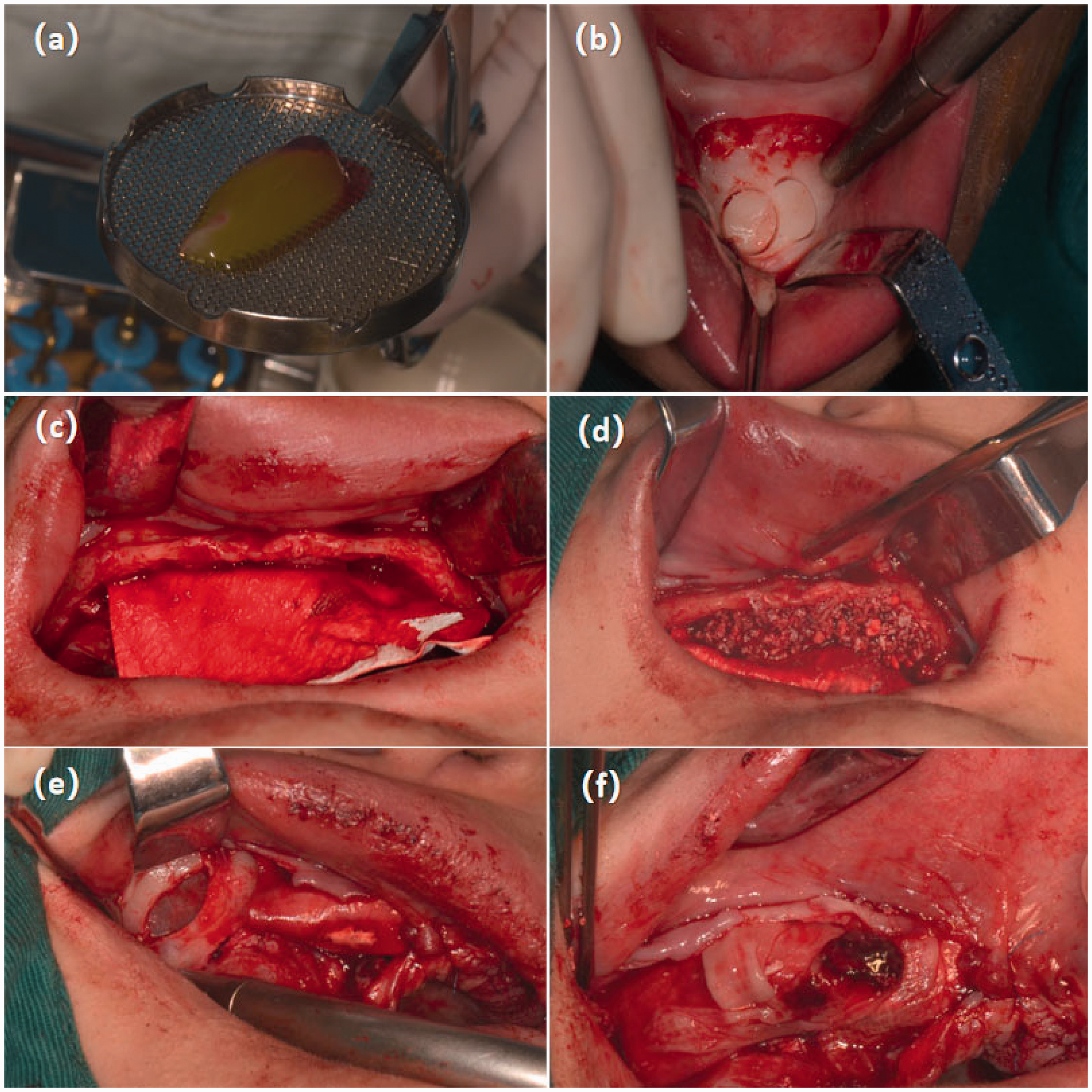
Intraoperative photographs of the first surgery. (a) Preparation of the concentrated growth factor membrane. (b) Acquisition of the annular bone block at the chin. (c) Placement of the Bio-Gide membrane on the palatal side. (d) Filling of the palatal side with mixed bone powder. (e) Opening of the lateral wall of the maxillary sinus. (f) Completion of the concentrated growth factor membrane fixation.
With the patient under local anesthesia, the flap was incised and the alveolar ridge was observed to be quite narrow. A 30- × 40-mm piece of periosteum (Bio-Gide; Geistlich Pharma AG) was then placed in the alveolar ridge (Figure 3(c)). Three membrane nails were fixed on the palatal side, and the mixed bone powder was placed on the labial-palatal side of the 15 to 25 tooth position (Figure 3(d)). Three additional membrane nails were fixed on the labial side, and the top of the alveolar ridge at the 21 to 25 tooth position was covered with a piece of CGF membrane. The lateral bone wall of the maxillary sinus was opened in the direction of the roots where teeth 16 and 17 are usually located, and the mucosa of the superior frontal floor was separated to elevate the maxillary sinus floor (Figure 3(e)). The mucosa of the maxillary sinus floor was lined with Bio-Gide collagen membrane (13 × 25 mm), and 0.5 g of Bio-Oss bone powder was implanted. Next, Bio-Gide periosteum (13 × 25 mm) was placed at the 16 and 17 sites, and three CGF membranes were placed at the top of the alveolar crest at the 15 to 17 tooth positions (Figure 3(f)). Finally, 6/0 absorbable sutures were used to tightly close the wound. Immediate postoperative computed tomography images revealed that the width of the lateral labial-palatal bone was increased. Adequate elevation of the maxillary floor was achieved, and the dense bone powder showed no spaces. No shadows of abnormal density were observed in the maxilla (Figure 4(a), (c), and (e)).

Intraoperative photograph of the second surgery. (a) Flap and soft tissue reduction in the mandibular region. (b) Completion of the nonresorbable membrane fixation in the lower anterior region.
Second surgery (16 weeks after first surgery): mandibular augmentation
Routine preoperative medications included an oral anti-inflammatory drug (cefradine capsule), an anti-swelling drug (dexamethasone acetate tablet), and an analgesic drug (loxoprofen sodium tablet). The patient’s oral cavity was rinsed twice with a chlorhexidine solution for 3 to 5 minutes per rinse. Routine disinfection of the surgical area was completed, and a sterile towel was placed in the surgical area. After local infiltration of anesthesia with epinephrine at tooth positions 37 to 47, the gingiva was incised and the gingival flap was peeled off with a stripper to expose the bone surface (Figure 5(a)). Autologous bone powder was obtained from the mandible and left mandibular external oblique line and mixed with three units of 0.5 g of Bio-Oss bone powder in a ratio of 1:1. Holes were drilled through the cortical bone to reach the bone marrow cavity in the mandibular chin and bilateral posterior regions, providing a blood supply source for the bone graft area, and the mixed bone powder was implanted.
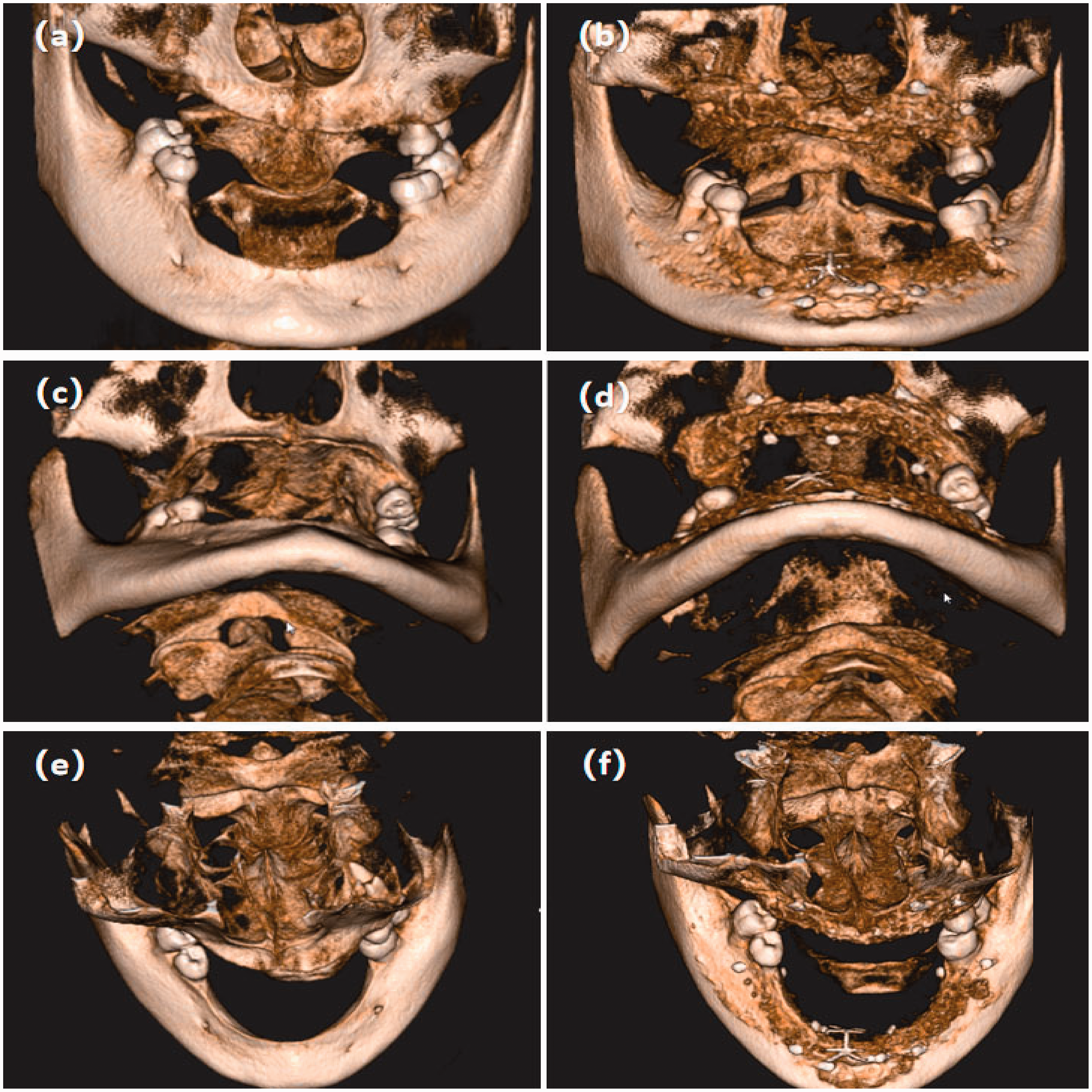
Comparison of three-dimensional imaging of the patient’s upper and lower jaws before and after augmentation surgery. (a, c, e) Three-dimensional images of the patient’s upper and lower jaws before surgery. (b, d, f) Three-dimensional images of the patient’s upper and lower jaws after surgery.
A 25- × 25-mm Bio-Gide membrane was placed in the bilateral posterior region, two membrane nails were placed on the buccolingual side for fixation, and a CGF membrane was placed on the surface of the right lower implant area. A titanium-reinforced Teflon membrane was placed on the mandibular anterior region, two membrane nails were placed on the labial lingual side, and three CGF membranes were placed on the surface (Figure 5(b)). Immediate postoperative computed tomography indicated that the bone powder at the mandibular augmentation site was dense with no cavities and exhibited sufficient volume (Figure 4(b), (d), and (f)).
Third surgery (40 weeks after first surgery): maxillary implant placement and apically repositioned flap
Preoperative medications consisted of an oral anti-inflammatory drug (cefradine capsule) and an analgesic drug (compound acetaminophen tablet). The patient’s oral cavity was rinsed twice with a chlorhexidine solution for 3 to 5 minutes per rinse. After routine disinfection of the surgical area and placement of a sterile towel, anesthesia with epinephrine and lorazepam was provided. The flap was then reflected, and sockets were prepared for the implants for teeth 11, 14, 16, 21, and 24. Superhydrophilic implants (ELEMENT MC INICELL; Thommen Medical AG, Grenchen, Switzerland) (types: 4.0 × 9.5 mm, 4.0 ×9.5 mm, 4.5 × 9.5 mm, 4.0 × 9.5 mm, 4.5 ×9.5 mm, and 4.5 × 9.5 mm) were used and torqued to 35 Ncm, and healing caps were then positioned. The flap was precisely separated using a scalpel blade from the top of the alveolar ridge to the buccal root side, forming a semi-thick flap that was then pushed to the root side. The flap edge was secured using a mattress suture on the periosteum located at the bottom of the vestibular sulcus. The wound was closed using 5/0 nonabsorbable sutures with reduced tension (Figure 6).

Intraoperative photograph of the third surgery. (a) Maxillary implant placement. (b) Root repositioning flap in the left upper posterior region. (c) Design of the soft tissue transfer flap (d) Postoperative stomatographic tomography. (e) Cone-beam computed tomography before maxillary implant placement surgery.
Fourth surgery (50 weeks after first surgery): mandibular implant placement
Preoperative medications consisted of an oral anti-inflammatory drug (cefradine capsule) and an analgesic drug (compound acetaminophen tablet). The oral cavity was rinsed with a chlorhexidine solution twice for 3 to 5 minutes per rinse. After routine disinfection of the surgical area and positioning of a sterile towel, anesthesia with epinephrine and lorazepam was administered. The flap was reflected, and the nonresorbable membrane in the anterior region was removed. The sockets were prepared for implants 45, 43, 42, 31, 33, and 35, and superhydrophilic implants (Thommen Medical AG) (models: 4.5 × 9.5 mm, 4.0 ×9.5 mm, 3.5 × 12.5 mm, 3.5 × 12.5 mm, 4.0 ×9.5 mm, and 4.5 × 9.5 mm) were placed and torqued to 35 Ncm. Healing caps were then positioned, and 5/0 nonabsorbable sutures were used to close the wound (Figure 7).

Intraoperative photograph of the fourth surgery. (a) Mandibular implant placement. (b) Postoperative stomatographic tomography. (c) Cone-beam computed tomography before maxillary implant placement surgery.
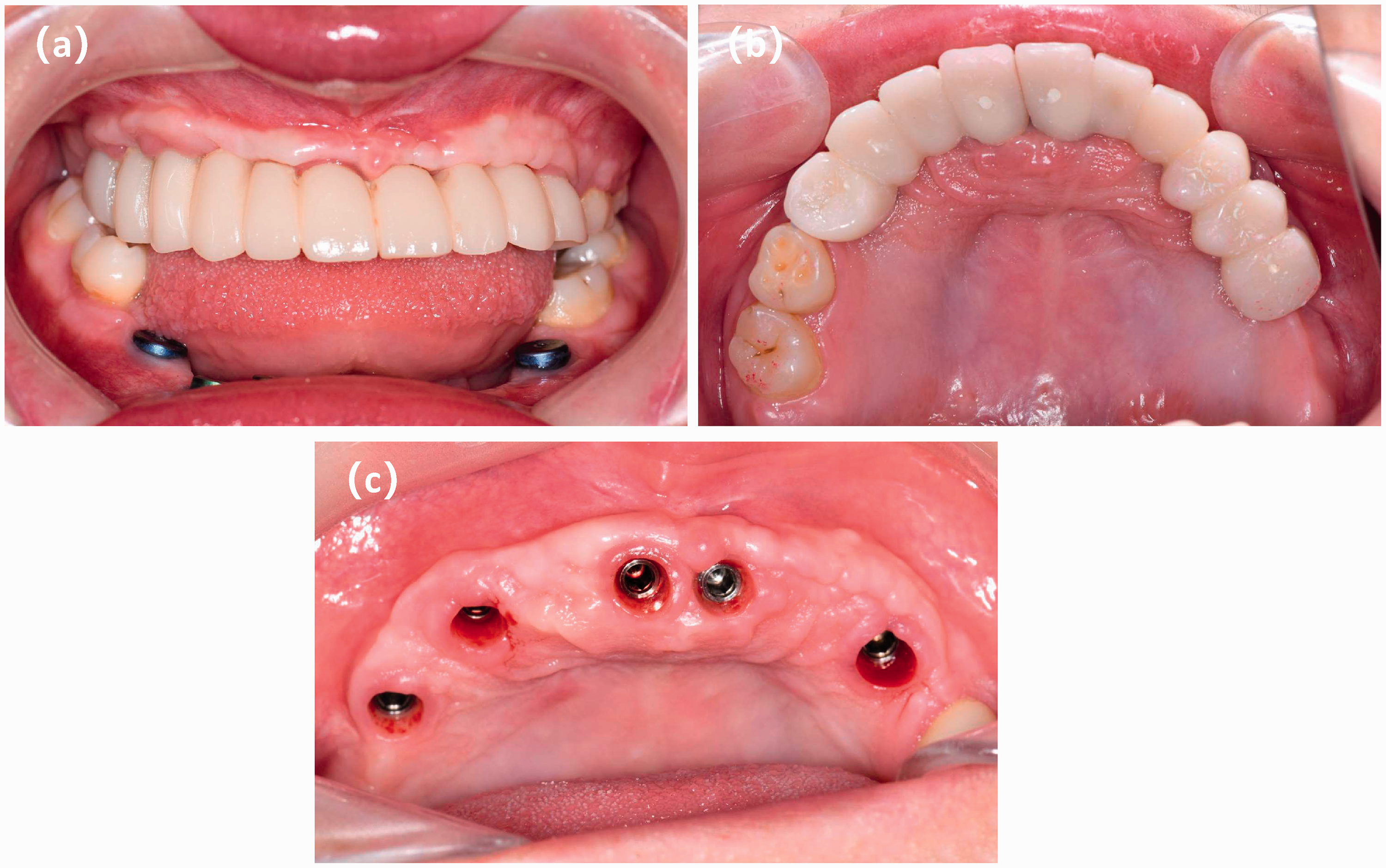
Trial wearing of maxillary provisional restoration. (a) Frontal view of the patient wearing the provisional restoration. (b) Joint view of the patient with the provisional restoration. (c) Gingival condition after removal of the temporary restoration.
Postoperative follow-up (54 weeks after first surgery): trial of maxillary temporary implant bridge and gingival contouring (Figure 8(a)–(c))
The patient was fitted with temporary maxillary implant bridges to shape the gums for a more aesthetic fit.
Fifth surgery (66 weeks after first surgery): mandibular free gingival flap graft (with custom-made shield)
Preoperative medications included an oral anti-inflammatory drug (cefradine capsule) and an analgesic drug (compound acetaminophen tablet). The oral cavity was rinsed with a chlorhexidine solution twice for 3 to 5 minutes per rinse. The surgical area was routinely disinfected, and a sterile towel was positioned. An incision was made under local anesthesia, 1 mm above the buccal lateral mucogingival junction at the 45 to 35 tooth location. The semi-thick flap was reflected with blunt separation in the root direction to expose the periosteum (Figure 9(a)). Next, two 12- × 3-mm free gingival flaps were made in the left and right upper palate (Figure 9(b)). The wound was tightly sutured with 4/0 absorbable sutures at the palatal incision site to provide compression using a custom-made rigid palatal shield. After placing the free gingival flap on the recipient location and tightly positioning it, the flap was fixed in place with 4/0 nonabsorbable sutures (Figure 9(c)). A prefabricated custom-made polyethylene membrane compression plate was applied to compress the wound (Figure 9(d)), and the wound was tightly closed with 4/0 nonabsorbable sutures.

Intraoperative photograph of the fifth surgery. (a) Turning of the semi-thick flap using blunt rootwise separation to expose the implant recipient area. (b) A free gingival flap was taken from the left palate. (c) The free gingival flap was sutured. (d) A custom-made shield was used to compress the implant recipient area.
Outcome and follow-up
Excellent horizontal and vertical increments of the alveolar ridge bone width were achieved in the edentulous area. During treatment, the tension was adequately reduced in all wounds, and closure was performed with tension-free sutures. The final restoration was completed 72 weeks after the initial surgery (Figure 10(a) and (b)). The gingival width of the peri-implant restoration in the anterior area was adequate, and the gingiva was pink, strong, and stable. The patient regained the ability to chew, obtained a satisfactory smile, and was satisfied with the overall final restorative results (Figure 10(c) and (d)).

Intraoral photograph of the permanent outcome. (a) Intraoral photograph of the zirconia prosthesis. (b) Stomatographic tomography of the completed repair. (c) The patient’s appearance before surgery. (d) The patient’s outward appearance and smile after the final procedure.
Discussion
Despite international efforts toward a standard dental treatment for patients with ED, no specific clinical treatment protocol has been established for dental management of the heterogeneous anomalies that occur in ED.10,11 In our patient with ectodermal hypoplasia, bone augmentation and CGF techniques were combined with free gingival grafting to simultaneously address the presence of insufficient bone volume and inadequate keratinized gingiva in the anterior region of the mouth. The treatment process was challenging, and the treatment time span was long. The procedure required a detailed understanding of the implant location and the bone volume and morphology, as well as the ability to precisely predict the expected effect of each stage of the restoration. Advanced surgical skills were needed to complete the intricate manipulations of the soft tissue in the aesthetic region of the anterior teeth and successfully carry out the tension-free suturing.
The soft tissue augmentation on the labial side of the mandible effectively covered the dark color of the implant abutment without affecting the aesthetics of the anterior region of the patient’s facial appearance. Research has shown that maintaining appropriate spacing might play a more important role in the osteogenesis process than, for example, cellular isolation. 12 Therefore, in this case, a titanium-reinforced polytetrafluoroethylene membrane was placed in the anterior mandibular region for adequate maintenance of the osteogenic space.
CGF is the latest-generation platelet concentrate. It is obtained by collecting the patient’s venous blood and applying differential centrifugation, which releases a range of growth factors associated with healing of damaged tissues and bone regeneration. 13 In response to this patient’s severe bone deficiency, insufficient soft tissue thickness, and thin gingiva, a CGF membrane was applied along with extensive bone grafting to prevent wound dehiscence, and satisfactory results were obtained. However, no consensus has been reached on the specific mechanism by which CGF promotes bone tissue regeneration and healing.14,15
In this case, the patient’s preoperative keratinized gingiva was severely underdeveloped, and the vestibular sulcus in the mandibular anterior region was nearly flush with the crest after bone augmentation. The presence of adequate keratinized gingiva surrounding the cuff is widely considered to act as a mucosal barrier around the implants. Thus, the presence of adequate keratinized gingiva ensures long-term stability of the peri-implant tissues from both biological and aesthetic standpoints. A free gingival flap graft with a custom-made polyethylene shield was therefore used in the mandibular area of our patient, and a root-repositioning flap was used in the maxillary area. A gingival width of 1.5 to 2.5 mm was obtained with excellent postoperative results. In a previous study, rootward repositioning flap surgery could predictably produce keratinized tissue and attached gingiva that exhibited long-term stability. 16 This case demonstrates that free gingival flap grafting accompanied by rootward repositioning flap surgery can provide sufficient soft tissue augmentation in the short term. However, determination of the final morphology of the soft tissue in this patient’s surgical area requires further observation through long-term follow-up visits.
During the recovery period after free gingival grafting, patients often experience significant discomfort between the graft extraction area and the recipient area due to daily masticatory behaviors and movements of the lip, cheek, and tongue muscles. In this case, the palatal shield protected the flap area, and the custom-made shield protected the implantation area. It also provided fixation for the grafted soft tissue, which isolated some of the mechanical stimulation of the wound and reduced the patient’s discomfort caused by oral physiological activities during the healing period. These effects helped to improve the patient’s physical and mental health. Thus, applying multiple techniques such as those used in this case should allow successful restoration of the aesthetic zone of the anterior teeth in patients with ED. However, the long-term health and stability of this restorative procedure require additional evaluation for longer periods of time.
Conclusion
Based on high-quality bone augmentation techniques, soft tissue grafting, and the combination of multiple techniques, implant prostheses can be used to treat defects in teeth or replace missing teeth in patients with ED syndromes. This procedure facilitated the morphological and functional restoration of our patient’s teeth and jaw and further promoted her physical and mental health.
Footnotes
Author contributions
Zhu Q wrote this manuscript and treated the patient. Wang R, Jiang Y, and Yu J provided advice regarding possible treatments and gave valuable comments regarding this manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics statements
Written informed consent was obtained from the patient to publish this report and any accompanying images. The authors have read the CARE Checklist (2016) statement, and the manuscript was prepared and revised accordingly. 17 This report was not approved by an ethics committee or institutional review board because of the nature of the study (case report).
