Abstract
Objective
To investigate the prevalence of fragmented QRS (fQRS) on electrocardiograms (ECG) in patients with different stages of chronic kidney disease (CKD) and to examine the association between fQRS and left ventricular systolic function.
Methods
This retrospective study analysed clinical and laboratory data from consecutive patients with CKD. The relationship between fQRS and left ventricular systolic function was evaluated using univariate and multivariate logistic regression analyses.
Results
A total of 310 patients (186 males; mean ± SD age, 52.24 ± 15.72 years) with CKD participated in this study. The prevalence of fQRS was 30.32% (94 of 310 patients). The prevalence of fQRS was more common in the inferior leads (53.19%; 50 of 94) compared with the anterior leads (21.28%; 20 of 94) and the lateral leads (25.53%; 24 of 94). Multivariate logistic regression analysis showed that left ventricular ejection fraction (odds ratio [OR] 1.356; 95% confidence interval [CI] 1.022, 2.036) and coronary artery disease (OR 2.355; 95% CI 1.056, 5.251) were independent risk factors of fQRS in patients with CKD.
Conclusion
The fQRS was prevalent in patients with CKD regardless of the CKD stage.
Introduction
Cardiovascular mortality is the leading cause of morbidity and mortality in patients with chronic kidney disease (CKD). 1 It is well known that heart failure (HF) is prevalent and increases with the severity of CKD. 2 It is estimated that HF is 12–36 times more common in patients undergoing dialysis compared with the general population; and among dialysis patients, the incidence of HF is 7% per year. 2 Myocardial fibrosis caused by uraemic toxins with cardiovascular toxicity is associated with cardiac systolic dysfunction in patients with CKD.3–5
Fragmented QRS (fQRS) is defined as an additional R wave (R’), or notching in the nadir of the S wave, or the presence of >1 R’ in two contiguous leads on a 12-lead resting electrocardiogram (ECG). 6 The fQRS is highly prevalent in patients with ischaemic and non-ischaemic cardiomyopathy; and is a marker of myocardial scarring and myocardial fibrosis.7–9 Furthermore, the fQRS has been found to be associated with cardiac systolic dysfunction and increased adverse cardiac events in patients.9–11 The prevalence and function of fQRS in CKD remains unclear. This current study investigated the prevalence of fQRS during different stages of CKD and examined the association between fQRS and left ventricular function.
Patients and methods
Study population
This retrospective study analysed data from consecutive Chinese hospitalized patients with CKD that were admitted to the Department of Nephrology, First Affiliated Hospital of Xi'an Jiao Tong University, Xi’an, Shaanxi Province, China between January 2017 and June 2018. All patients had been diagnosed with CKD according to the Kidney Disease Improving Global Outcomes 2012 Clinical Practice Guideline for the Evaluation and Management of CKD. 12 The exclusion criteria were as follows: (i) patients with a history of malignancy, cardiac surgery, significant structural heart disease, findings of active infection, acute stroke, acute kidney injury, bundle-branch block (left bundle-branch block, incomplete or complete right bundle-branch block) or intraventricular conduction delay (duration of QRS >120 ms) on ECG; (ii) patients with permanent pacemakers.
The Ethics Committee of the First Affiliated Hospital of Xi’an Jiao Tong University approved this study (no. XJTU1AF2019LSK-021). All patients participating in the study provided verbal informed consent.
Clinical and laboratory data collection
Clinical data including age, sex, medicines used, history of coronary heart disease, diabetes mellitus and hypertension were recorded. The laboratory data, which included haemoglobin, blood urea nitrogen, creatinine, potassium, calcium, phosphorus, magnesium and parathyroid hormone (PTH), were retrieved from the hospital database. The staging of CKD was based on the estimated glomerular filtration rate (eGFR) and used the Chronic Kidney Disease Epidemiology Collaboration equation. 12 The stages of CKD were CKD3 (30 ≤ eGFR < 60 ml/min), CKD4 (15 ≤ eGFR < 30 ml/min) and CKD5 (eGFR< 15 ml/min). 12
ECG and echocardiographic examination
Twelve-lead surface ECGs were recorded at a paper speed of 25 mm/s and a voltage of 10 mm/1 mV in the supine position. The ECG criteria of QRS fragmentation was acquired from a previous study. 6 The fQRS pattern includes various morphologies of the QRS interval (Figure 1). fQRS was defined by the presence of an additional R wave (R′), or notching in the nadir of the S wave, or the presence of >1 R′ (fragmentation) in two contiguous leads. The location of the fQRS in the ECG leads was categorized as lateral (I, aVL, V4–V6), anterior (V1–V3) and inferior (II, III, aVF). Left ventricular hypertrophy was assessed according to the Sokolow–Lyon criteria. 13 All tracings were studied by two independent investigators (P. L. & D.H.) and a consensus was reached in cases where there was a disagreement. The conventional M-mode, B-mode and Doppler parameters were measured in accordance with the American Society of Echocardiography guidelines. 14 Left ventricular ejection fraction (LVEF), left ventricular end-diastolic and end-systolic diameters, and posterior and septal wall thicknesses were measured. Mitral inflow E- and A-wave velocities, E-wave deceleration time and pulmonary venous reverse A-wave duration were used to categorize left ventricular diastolic function using well-established criteria.

Different morphologies of a fragmented QRS (fQRS) on a 12-lead resting electrocardiogram, including an additional R wave (R’), or notching in the nadir of the S wave, or the presence of >1 R’ in two contiguous leads. rSr’, the QRS wave begins with an r wave followed by an S wave and ends with a second r wave; rSR’, the QRS wave begins with an r wave followed by an S wave and ends with a second R wave; RSR’, the QRS wave begins with an R wave followed by an S wave and ends with a second R wave.
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp., Armonk, NY, USA). Continuous variables are presented as mean ± SD and categorical variables are presented as n of patients (%). Continuous data were analysed using Student’s t-test or Mann–Whitney U- test. Categorical data were compared using χ2-test or Fisher’s exact test. Comparisons of multiple mean values were carried out by Kruskal–Wallis test or analysis of variance as appropriate. Multivariate logistic regression analysis was used to assess the association between fQRS and various independent variables. The odds ratio (OR) and 95% confidence interval (CI) are presented. A P-value < 0.05 was considered statistically significant. In multivariate regression models, the effect size was adjusted for variables with a significance level of P < 0.05 in the univariate analysis.
Results
A total of 310 patients (186 males; mean ± SD age, 52.24 ± 15.72 years) with CKD participated in this retrospective study. The prevalence of fQRS was 30.32% (94 of 310 patients). Among patients with CKD stages 3, 4 and 5, the proportion of patients with fQRS was 30.77% (12 of 39 patients), 20.00% (8 of 40 patients) and 32.03% (74 of 231 patients), respectively (Figure 2). The prevalence of fQRS was more common in the inferior leads (53.19%; 50 of 94) compared with the anterior leads (21.28%; 20 of 94) and the lateral leads (25.53%; 24 of 94) (Figure 3).

The prevalence of fragmented QRS (fQRS) during different stages of chronic kidney disease (CKD). ECG, electrocardiogram.
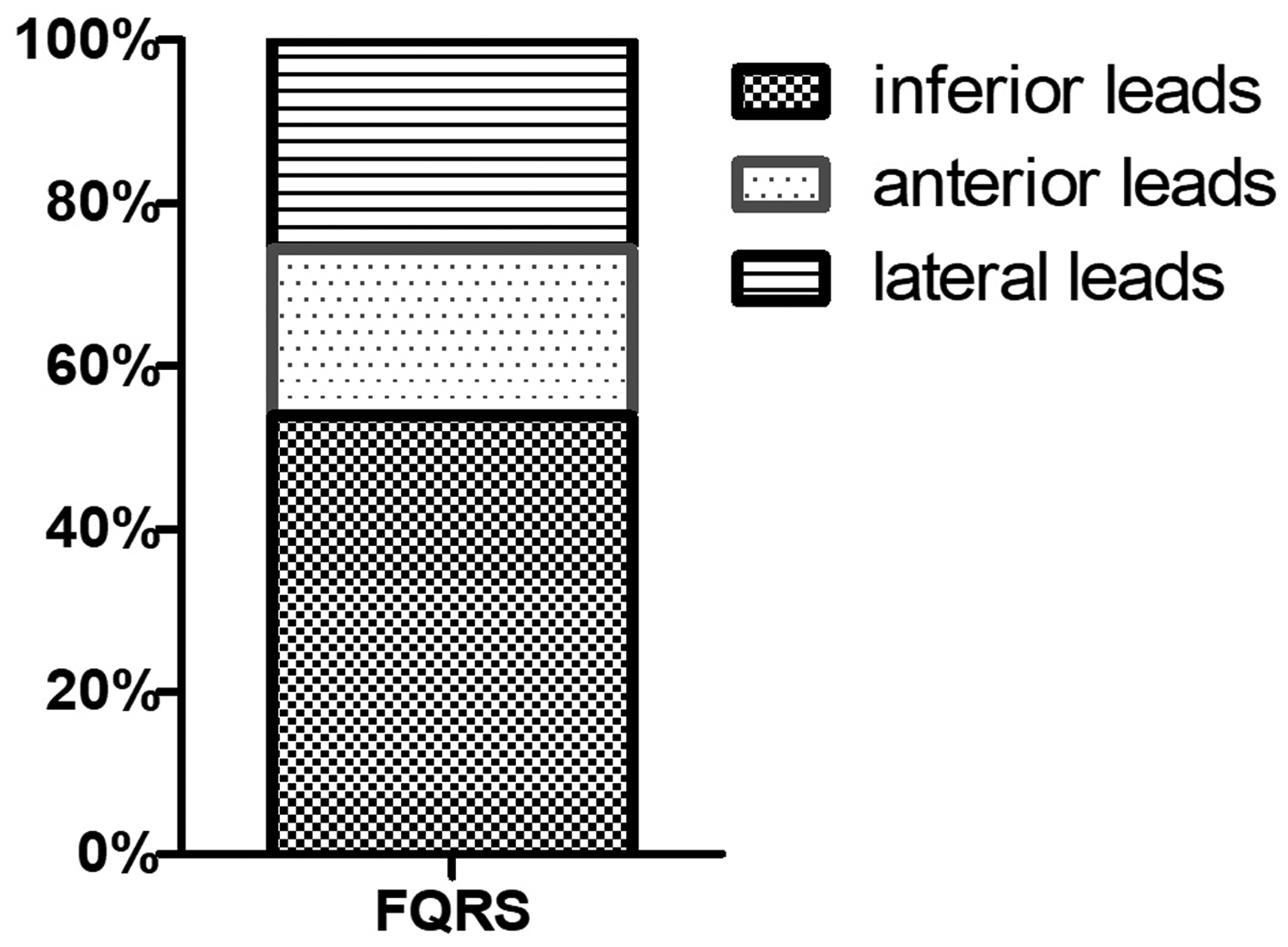
The distribution of fragmented QRS (fQRS) in different electrocardiogram leads.
Clinical, echocardiographic and laboratory data of the study population are presented in Table 1. According to univariate analysis, patients with fQRS had a significantly higher prevalence of coronary artery disease (CAD), significantly higher PTH levels and significantly lower LVEF (P < 0.05 for all comparisons). Patients with fQRS in the lateral leads of ECG had significantly lower mean ± SD LVEF (56.67 ± 6.24) compared with those in the other leads or those without fQRS (ECG without fQRS, 65.12 ± 6.45; fQRS in the inferior leads, 59.97 ± 5.12; fQRS in the anterior leads, 60.13 ± 3.24) (P < 0.05 for all comparisons) (Figure 4). Patients with fQRS in the inferior and anterior ECG leads did not have significantly lower LVEF than those without fQRS. The prevalence of left ventricular diastolic dysfunction was 72.58% (225 of 310 patients), which had no significant relationship with fQRS. Multivariate logistic regression analysis showed that LVEF (OR 1.356; 95% CI 1.022, 2.036; P = 0.042) and CAD (OR 2.355; 95% CI 1.056, 5.251; P = 0.036) were significantly associated with fQRS in patients with CKD after adjustment of all other variables (Table 2).
Clinical and echocardiographic characteristics of the study population (n = 310) with or without fragmented QRS (fQRS) on electrocardiograms (ECG).
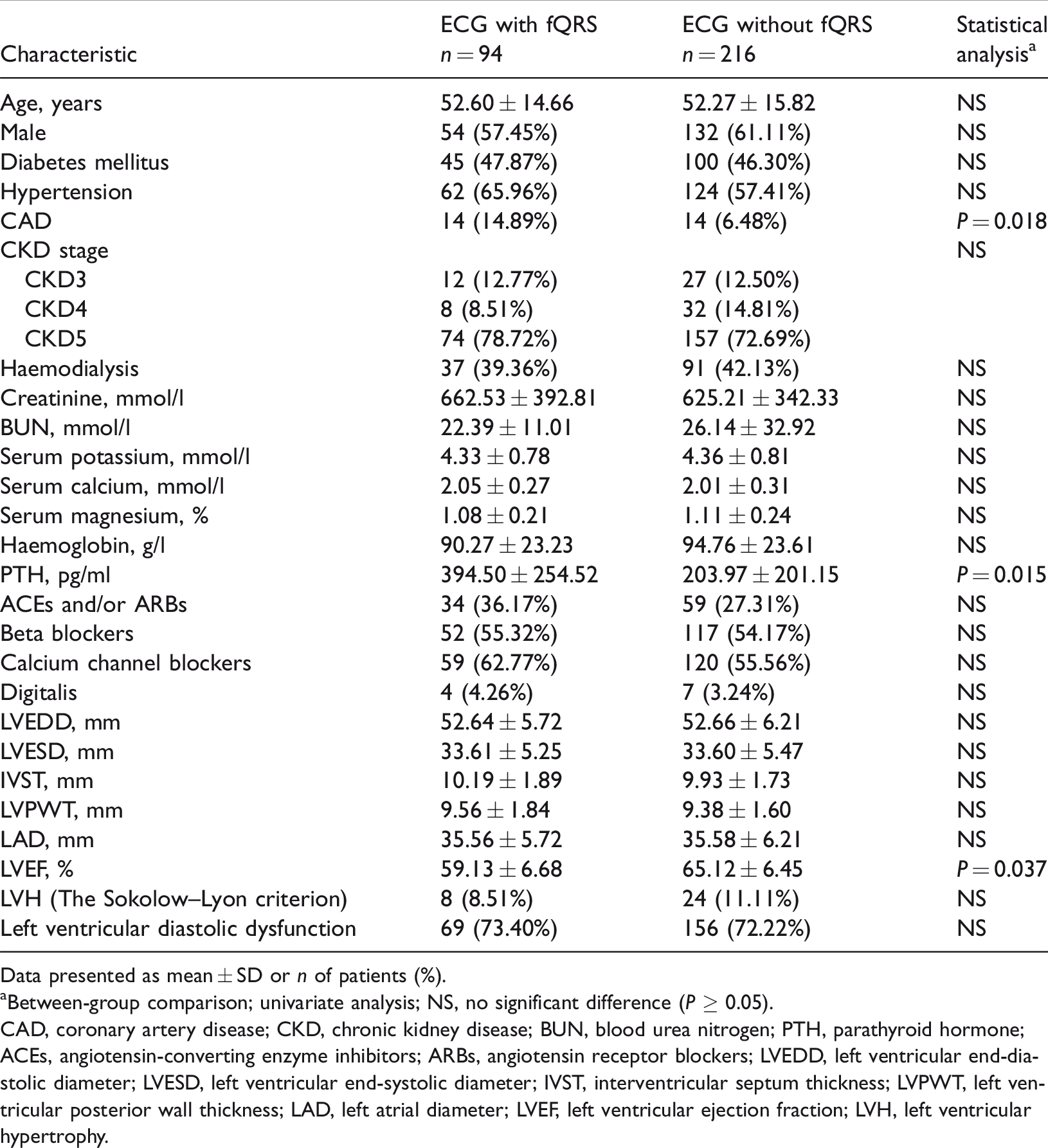
Data presented as mean ± SD or n of patients (%).
aBetween-group comparison; univariate analysis; NS, no significant difference (P ≥ 0.05).
CAD, coronary artery disease; CKD, chronic kidney disease; BUN, blood urea nitrogen; PTH, parathyroid hormone; ACEs, angiotensin-converting enzyme inhibitors; ARBs, angiotensin receptor blockers; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; IVST, interventricular septum thickness; LVPWT, left ventricular posterior wall thickness; LAD, left atrial diameter; LVEF, left ventricular ejection fraction; LVH, left ventricular hypertrophy.

Left ventricular ejection fraction (LVEF) in patients with fragmented QRS (fQRS) in different electrocardiogram (ECG) leads. Between-group comparison; univariate analysis.
Multivariate logistic regression analysis of the association between left ventricular ejection fraction (LVEF), coronary artery disease (CAD) and parathyroid hormone (PTH) with fragmented QRS (fQRS) in patients with chronic kidney disease.

OR, odds ratio; CI, confidence interval; NS, no significant association (P ≥ 0.05).
Discussion
This current retrospective analysis aimed to determine the prevalence of fQRS and investigate its relationship with left cardiac function in Chinese patients with CKD. The current study demonstrated that the fQRS was prevalent in patients with CKD regardless of the CKD stage. Furthermore, although fQRS in the inferior leads accounted for more than half of all of the ECG leads, only fQRS in the lateral leads was significantly associated with a lower LVEF. To the best of our knowledge, this is the largest study on the prevalence of fQRS in patients with CKD and its relationship with left ventricular function.
Chronic kidney disease is a growing public health issue for a number of reasons. For example, a reduced eGFR is a risk factor for cardiovascular mortality caused by acute myocardial infarction, heart failure, thromboembolic disease and sudden cardiac death (SCD). 15 Patients with CKD are more likely to die from cardiovascular disease than to develop end-stage renal disease. 16 Myocardial fibrosis caused by uraemic toxins with cardiovascular toxicity is thought to be one of the contributors to this deterioration.3–5,17 Fragmented QRS on the ECG is a simple, inexpensive and readily available marker of myocardial fibrosis and myocardial scarring.7,18,19 Fragmented QRS is associated with heart failure and is a predictor of mortality and arrhythmic events in various diseases.9,20–22
A previous study in a Finnish middle-aged general population found that the prevalence of fQRS was 19.7% (n = 2147), including 15.7% in the inferior leads, 0.8% in the lateral leads and 2.9% in the anterior leads. 23 The prevalence of fQRS was 50% in the inferior leads, 30% in the lateral leads and 43% in the anterior leads in patients that experienced exercise-related SCD. 20 Furthermore, the prevalence of fQRS is 21.9% in myocardial infarction, 24 26.1% in metabolic syndrome 22 and 60% in CKD. 21 This current study found that the prevalence of fQRS in patients with CKD was 30.32% and there were no significant differences in prevalence among the three different CKD stages. Furthermore, the fQRS was most prevalent in the inferior leads, followed by the lateral leads and the lowest was in the anterior leads. This current study was the first report of fQRS in Chinese patients with CKD and the prevalence was lower than the previous study. 21 There is no obvious explanation for this difference between the two studies except for ethnicity of the study population, 21 so more evidence is needed to understand the prevalence of fQRS in CKD.
The fQRS on a 12-lead ECG is a marker of a prior myocardial infarction, defined by regional perfusion abnormalities, which has a substantially higher sensitivity and negative predictive value compared with the Q wave. 6 In acute coronary syndrome, fQRS appeared within 48 h (especially within 24 h) from the onset of symptoms and persisted thereafter. 25 The fQRS is an independent predictor of mortality in patients with CAD. A previous study demonstrated that all-cause mortality (34.1% versus 25.9%) and cardiac event rate (49.5% versus 27.6%) were higher in CAD patients with fQRS than in patients without fQRS, respectively. 26 Furthermore, fQRS can also reflect intracardiac conduction abnormality and will represent a substrate for ventricular arrhythmia. 27 The conclusion from this current study was consistent with previous studies that showed that CAD is an independent risk factor for the fQRS.
Previous studies have reported the relationship between fQRS and left ventricle systolic functions. For example, there was a significant inverse correlation between the number of fQRS and left ventricle systolic functions.21,22,24,28 Moreover, fQRS was independently associated with subclinical left ventricle dysfunction in patients with CKD. 21 Pathophysiological fQRS is generally accepted to derive from regional myocardial fibrosis/scarring and ischaemia, which cause nonhomogeneous myocardial electrical activation. 7 Myocardial fibrosis/scarring contributes to left ventricle dysfunction. 29 In the current study, the LVEF was significantly lower in the patients with fQRS and only fQRS in the lateral leads was associated with significantly lower LVEF compared with the other leads. The fQRS in the lateral leads represent abnormal structures in the left ventricle, which is also associated with a worse outcome in patients. 23 Previous research showed that fQRS was related to left ventricular systolic dysfunction. 28 In this current study, the prevalence of left ventricular diastolic dysfunction was 72.58%, which was similar to that shown in a previous study. 30 Although there was no significant relationship between fQRS and left ventricular diastolic dysfunction, the left ventricular diastolic dysfunction was more prevalent in patients with fQRS than those without although the difference was not significant. Hyperphosphataemia is prevalent in patients with CKD, which is associated with CAD, peripheral vascular disease, endothelial dysfunction, disorders of mineral and bone metabolism, and a higher mortality rate. 31 Hyperphosphataemia stimulates PTH release. 32 Evidence from basic research has shown that PTH contributes to four major cardiovascular effects: contractile disturbance, cardiomyocyte hypertrophy, cardiac interstitial fibrotic and vasodilator effect. 33 There was no clinical evidence of a significant relationship between PTH and fQRS in the current study.
This current study had several limitations. First, it was a single-centre retrospective study and the sample size was small. Secondly, myocardial structural changes were not observed using imaging modalities. Thirdly, the study did not assess right ventricular function.
In conclusion, this current study found that fQRS was prevalent in Chinese patients with CKD regardless of the CKD stage. Furthermore, fQRS in the inferior leads was the most prevalent but only fQRS in the lateral leads was significantly associated with a lower LVEF.
