Abstract
Desmoplastic trichilemmoma (DT) of the scalp is a rare disease. In this report, we describe a 67-year-old man who had a small papule lesion around the hairline of the left scalp for more than 40 years. The lesion had rapidly grown to become a solitary, round, 15-mm neoplasm within 6 months. Histopathological examination showed that the mass was composed of a peripheral layer of columnar cells with nuclear palisading resembling the outer root sheath; the mass was surrounded by a thickened glassy basement membrane. The stroma in the center of the mass displayed solid, hyaline degeneration and an eosinophilic pattern, streaked with epithelial cords or islands. Immunohistochemical evaluation showed diastase-resistant, periodic acid–Schiff-positive desmoplastic stroma at the center, and Ber-EP4-negative tumor cells (CD34-positive focally and Bcl-2-positive at the periphery). The tumor was completely resected, and no recurrence occurred during the follow-up period. In this report, we also summarize immunohistochemical features from previous reports of DT, facilitating differential diagnosis of DT from some invasive skin tumors.
Keywords
Introduction
Trichilemmoma, first described by Headington et al. 1 in 1962, is a benign tumor of cutaneous appendages with outer hair root sheath differentiation. 2 Trichilemmomas may occur as solitary or multiple tumors. Solitary trichilemmoma typically presents as a single, small, skin-colored papule on the face; in contrast, multiple trichilemmoma typically exhibits a perinasal and perioral distribution, and serves as a marker for Cowden's disease, an autosomal dominant inherited cancer syndrome.3,4 Desmoplastic trichilemmoma (DT) is a rare histological type of trichilemmoma. Thus far, only 10 cases have been reported in which DT affected the scalp.5–8 In the present report, we describe a man who exhibited DT of the scalp; we also summarize immunohistochemical features from previous reports of DT, which may aid in differential diagnosis of this disease.
Case report
A 67-year-old man reported the presence of a small papule lesion around the hairline of the left scalp for more than 40 years; it had not caused him any discomfort. He presented to our hospital for examination of the lesion, which had shown rapid growth over the past 6 months but had not exhibited itching, pain, or bleeding. The patient had been diagnosed with type 2 diabetes 15 years prior, and he denied any history of alcoholism or drug allergy. There were no similar cases in his family.
Upon physical examination, the only notable finding was a single, round, 15-mm neoplasm around the hairline of the left scalp, with hyperplasia in the surrounding area and a minimal brown pigmentation and crusting at the margin (Figure 1). The mass was tough without tenderness, and there was no enlargement of retroauricular or cervical lymph nodes. Histopathological examination of skin biopsies showed that the mass was surrounded by lobules of epithelial cells, some of which had clear cytoplasm (Figure 2a–e). At the periphery of the lobules was a layer of columnar cells with nuclear palisading that resembled the outer root sheath of hair follicles. These lobules were surrounded by a thickened glassy basement membrane. The stroma in the center of the mass displayed solid, hyaline degeneration and an eosinophilic pattern, which was streaked with epithelial cords or islands. Focal infiltration of lymphocytes, plasma cells, and eosinophilic granulocytes was observed in the dermis. Immunohistochemical evaluation showed periodic acid–Schiff (PAS)-positive, diastase-resistant, desmoplastic stroma in the center of the mass (Figure 2f, g). At the periphery, CD34 was present in the tumor cell membrane (Figure 2h); B-cell lymphoma 2 (Bcl-2) was present in the cell membrane and cytoplasm (Figure 2i), while Ber-EP4 staining was negative (Figure 2j). The patient was diagnosed with DT and the tumor was completely resected under local anesthesia. The patient did not experience recurrence of disease during the 8-month follow-up period after surgical removal of the primary tumor.

A mass lesion around the hairline of the left scalp of a 67-year-old man. A solitary, round, 15-mm neoplasm is present, with hyperplasia in the surrounding area and a small amount of brown pigmentation and crusting at the margin.
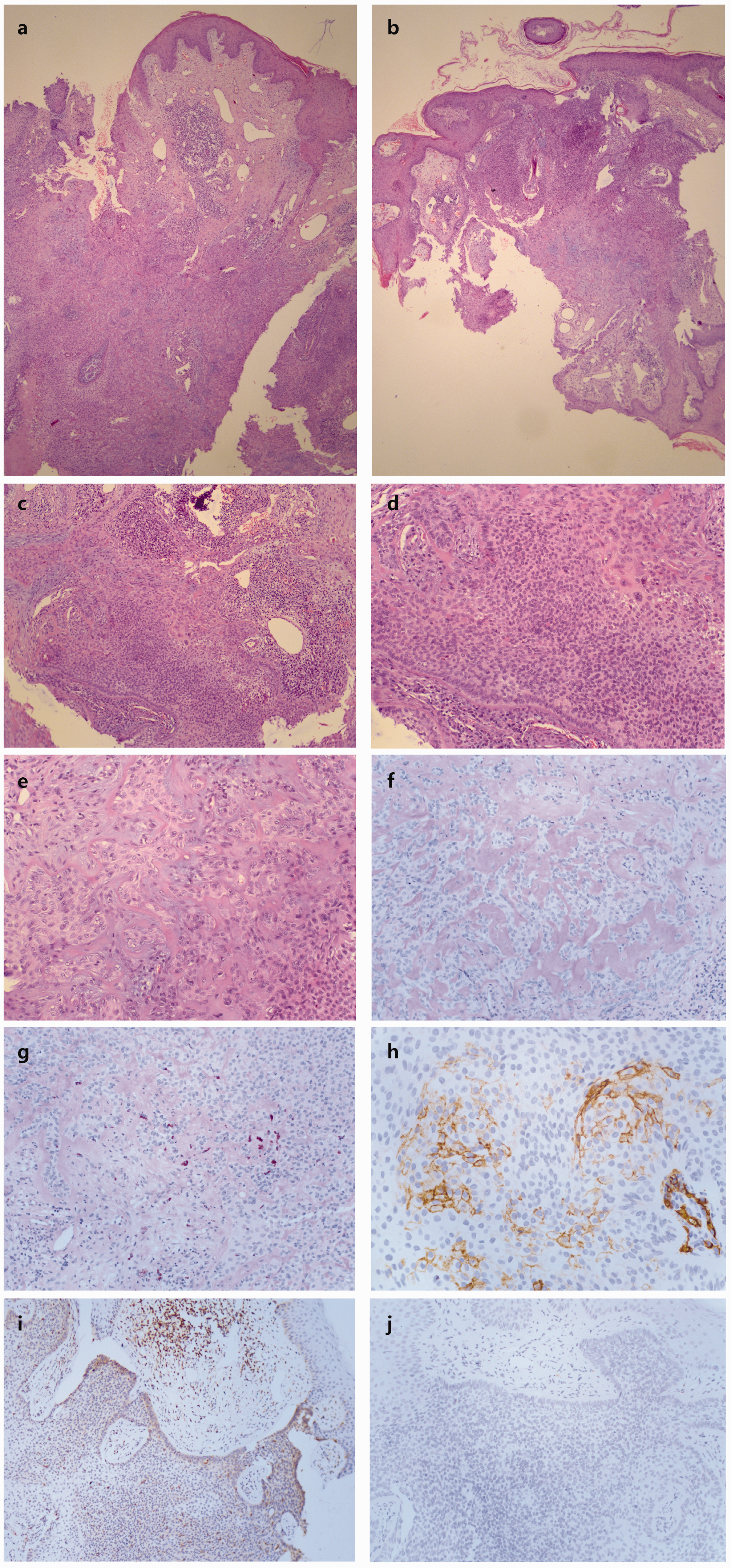
Histopathological examination of skin biopsies. a) The mass is centered with desmoplastic stroma, and surrounded by lobules of epithelial cells (hematoxylin and eosin [H&E] stain, 40×). (continued)b) The tumor is lobulated and continuous with the epidermis (H&E stain, 40×). c) The tumor is composed of small cells and hyaline degeneration in stroma, with focal infiltration of lymphocytes, plasma cells, and eosinophilic granulocytes in the dermis (H&E stain, 100×). d) A few of the tumor cells showing clear cytoplasm with palisading of low columnar cells in the periphery of a tumor lobule surrounded by a thickened basement membrane (H&E stain, 200×). e) Stroma in the center of the mass displaying solid, hyaline degeneration and an eosinophilic pattern, streaked with epithelial cords or islands (H&E stain, 200×). f) Desmoplastic stroma with periodic acid–Schiff (PAS)-positive staining (PAS stain, 200×). g) Desmoplastic stroma with PAS diastase-positive staining (PAS diastase stain, 200×). h) CD34-positive expression on peripheral tumor cell membrane, but not in desmoplastic stroma (immunohistochemistry, 400×). i) Bcl-2 positive expression in tumor cell cytoplasm and membrane (immunohistochemistry, 100×). j) Ber-EP4-negative staining (immunohistochemistry, 200×).
This report was approved by the ethics committee of Beijing Friendship Hospital, Capital Medical University, in accordance with the ethical standards of the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Written informed consent was obtained from the patient for publication of this report.
Discussion
DT is a benign adnexal tumor of the outer sheath of the pilosebaceous follicle, which comprises superficial lobular growth with central desmoplastic stroma. This tumor was first described by Hunt et al. 9 in 1990. DT is regarded as a rare lesion: Tellechea et al. 10 identified only seven cases of DT in 21,198 consecutive skin biopsies, and Crowson et al. 11 identified only 28 cases of DT among 120,000 accessioned biopsies over 18 months. The disease typically occurs in men at a mean age of 59.5 years (range: 8‒89 years), 7 and primarily affects the face (particularly in or around the lips, as well as on the eyelids); 12 it sometimes occurs on the scalp, neck, or chest. 9 DT typically comprises a single dome-shaped papule <1 cm in diameter, which is smooth or crusted; it may exhibit pearly edges. 9 The lesion develops slowly over months to years. 7 However, sometimes DT develops from nevus sebaceous.5,13–15 DT can develop concomitantly with a variety of other adnexal tumors, such as trichoblastoma; 5 it also may occur in combination with basal cell carcinoma (BCC).11,15,16
Histologically, a DT lesion is lobulated with a clear boundary that extends into the dermis and is continuous with the epidermis. 10 It is composed chiefly of small cellular clusters with uniform nuclei and clear cytoplasm, which contain variable glycogen vacuolation. There is a clear palisadic arrangement of the cells at the periphery of the lobules, surrounded by a thick basement membrane. 17 The architectural pattern of epithelial cell cords and small nests merges with a dense eosinophilic hyaline stroma at the central area. The proportion of desmoplasia varies, but is generally between 20% and 60% of the lesion. 9 Although 10 cases have been reported in which DT developed on the scalp,5–8 the patient in our case had some unique clinical features: (1) the lesion was 1.5 cm in diameter, larger than the typical size of 1 cm; and (2) the lesion showed different histological characteristics, with less glycogen in the cytoplasm, as well as tumor cells with PAS-negative staining that were only focally positive for CD34. These findings differed from those in previous reports. For a more comprehensive understanding of the histological features of this disease, we reviewed the previously reported immunohistochemical results by searching the literature in PubMed, Web of Science, EBSCO, and MEDLINE electronic databases. The following keywords were searched in all four databases: “desmoplastic trichilemmoma,” “trichilemmoma,” and “adnexal neoplasm.” As shown in Table 1, 95 cases of DT have been reported, of which 49 underwent immunohistochemical staining.
Summary of previous results of immunohistochemical staining.

CEA, carcinoembryonic antigen; CK, cytokeratin; EMA, epithelial membrane antigen; GFAP, glial fibrillary acidic protein; HPV, human papilloma virus; PAS, periodic acid–Schiff; SMA, smooth muscle actin.
DT tumor cells predominately have a glycogen-rich, clear cytoplasm, which is positively stained by PAS; in some tumors, the cells are diastase-resistant.9,14,18,19 Most cases also exhibit positive staining for CD34, a surface marker expressed in tumor cells during complete or partial differentiation toward outer root sheath cells.2,6,16,19–22 Normal outer root sheath cells reportedly express cytokeratin (CK)1/10, CK5/14, CK6/16, and CK17. 23 In DT, tumor cells are often positive for CK5/6, CK34βE12 (a high-molecular-weight CK), and CK14.17,24 p63 is expressed in more than 90% of benign and malignant adnexal tumors, including DT; 17 conversely, Ki67 has been found in fewer than 10% of DT tumor cell nuclei, indicating low cell proliferation and a benign nature of the tumor. 17 Carcinoembryonic antigen (CEA), epithelial membrane antigen (EMA), CK7, and α-lactalbumin are typically not expressed in DT,10,14,17 indicating that DT tumors do not include a true glandular component. Several additional markers are helpful for differential diagnosis. For instance, negative staining for S100, smooth muscle actin, and glial fibrillary acidic protein may help to exclude the possibility of a myoepithelial lesion; 17 negative staining for BerEP4 excludes the possibility of BCC.16,22
Desmoplastic stroma is rich in glycogen and acidic mucopolysaccharides, indicated by positive staining using both PAS and Alcian blue methods.2,9,10,20 Positive staining for vimentin10,20,24 and type I collagen, 24 as well as positive Masson staining, 3 is indicative of interstitial collagen components; these are predominately produced by fibroblasts, a cell of mesenchymal origin. Massi and Franchi reported that the central extracellular matrix showed diffuse and intense positivity for tenascin. 24 Factor XIII subunit a (XIIIa) is a marker for distinguishing dendritic cells in a wide range of dermatological diseases. 25 Factor XIIIa expression is reportedly absent in stromal cells of DT, 10 indicating that the development of this stroma may occur independently of dendritic cells. Negative CD34 staining has been reported in hyalinized stroma. 17
DT can easily be misdiagnosed as one of several invasive tumors (most frequently sclerosing BCC and desmoplastic squamous cell carcinoma [SCC]) because of the random pattern in the center of DT lesions, where irregular cords and epithelial nests are entrapped in a dense, sclerotic, collagenous stroma.9,10,20 Sclerosing BCC lacks the circumscription of DT, as well as the preference of DT for the central area of the lesion. 22 Furthermore, in contrast to DT, BCC expresses minimal levels of CD34, but often demonstrates expression of BerEP4. 7 Desmoplastic SCC exhibits polygonal epithelioid morphology and peripheral neuroinvasiveness, 19 with PAS-negative staining and absence of CD34 expression, which differ from DT. In some cases, DT must be differentiated from malignant trichilemmoma, which exhibits heterogeneity and high Ki67 expression. In addition, immunohistochemical staining can be used to differentiate DT from some neoplasms that arise from sweat glands and sebaceous glands; the latter often express both CEA and EMA.7,10,14 In the present case, the patient was diagnosed with DT, based on focal CD34-positive staining and on Ber-EP4-negative staining in tumor cells, as well as PAS-positive staining and CD34-negative staining in desmoplastic stroma. Bcl-2 is a proto-oncogene that inhibits apoptosis, and is diffusely expressed throughout BCC tumor nodules; in trichoepithelioma, Bcl-2 is present only in the outermost layer of the cells. 26 In the present case, Bcl-2 was expressed solely at the periphery of the tumor, with a degree of staining between that of BCC and trichoepithelioma.
In a few cases, the dermoscopic features of trichilemmomas have been reported. Based on dermoscopic images, trichilemmomas have been described as keratin masses with perivascular whitish halos, 27 hyperkeratotic central areas with peripheral erythematous radial lines in a red iris-like structure, 28 non-pigmented nodules with peripheral vessels arranged in a radial and centered manner, 29 and linear vessels arranged in a radial manner with surrounding reflective whitish areas, as well as central crusted and hyperkeratotic areas. 30 In contrast, DT has been described as focal crown vessels with unspecific poorly defined white areas and hyperkeratotic central depression. 29 Therefore, the presence of white structures, a hyperkeratotic central area, and a radial arrangement of peripheral vessels may support a diagnosis of DT. Indeed, dermoscopy is a promising assistive tool for diagnosis of DT.
The clinical manifestations of DT are nonspecific. Histopathologically, it may be necessary to differentiate DT from multiple types of invasive skin tumors. Immunohistochemical staining can serve as a useful auxiliary tool in the diagnosis of DT.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
