Abstract
Objective
This case is reported to introduce an advanced surgical technique and share our experience with surgeons.
Methods
A 53-year-old man was admitted to the Department of Urology of The Second Xiangya Hospital with complaints of frequent urination and dysuria in June 2018. He had been diagnosed with genitourinary tuberculosis and left renal dysfunction at another hospital and had undergone left nephrectomy 6 months previously. At our institution, intravenous urography showed a normally functioning right solitary kidney. Computed tomography indicated a high-density mass in the abdominal cavity. At the patient’s request, robot-assisted laparoscopic augmentation ileocystoplasty and excision of the intraperitoneal mass was performed.
Results
The surgery was performed using a completely intracorporeal technique. The procedure was completed in 240 minutes, and the patient was discharged on postoperative day 11. At the 2-month follow-up, ultrasound examination showed that the maximal bladder capacity had increased to 1000 mL. Intravenous urography revealed no urine leakage, and filling of the bladder was satisfactory.
Conclusions
With the development of robot-assisted surgery, robotic surgery can be successfully used for less frequently performed operations.
Introduction
Tuberculosis (TB) is a leading public health problem, and genitourinary TB is a common extrathoracic form of TB. Thimble bladder is the characteristic late stage of genitourinary TB. Augmentation cystoplasty is necessary to relieve the irritative lower urinary tract symptoms and prevent upper tract deterioration. 1 The most widely used bowel segment for augmentation cystoplasty is a detubularized patch of ileum. Ideally, this procedure can reduce the risk of metabolic disturbance. 2 Augmentation ileocystoplasty was initially described in humans in 1889. The technique was accepted in the 1950s for the management of tuberculous bladders and was further popularized by Bramble in the 1980s. 3
Although commonly performed using the open technique, minimally invasive approaches such as laparoscopy and robot-assisted laparoscopy are more frequently being used for augmentation ileocystoplasty. Robot-assisted laparoscopic augmentation ileocystoplasty not only offers a cosmetic advantage but also involves a shorter hospital stay, less postoperative pain, and lower perioperative complication rates. However, limited information about the surgical technique is available in the published literature.
We herein report a case of robot-assisted laparoscopic augmentation ileocystoplasty and excision of an intraperitoneal mass. To the best of our knowledge, this is the first reported experience of robot-assisted laparoscopic augmentation ileocystoplasty in China and the second reported experience of robotic augmentation cystoplasty in a tubercular small contracted bladder. This case is being reported to introduce an advanced surgical technique and share our experience with surgeons.
Case report
A 53-year-old man with a 1-year history of frequent urination and dysuria was admitted to the Department of Urology of The Second Xiangya Hospital of Central South University (Changsha, China) in June 2018. He had been diagnosed with genitourinary TB and left renal dysfunction in another hospital. Therefore, he had been administered anti-tubercular therapy and had undergone left nephrectomy 6 months previously to treat the genitourinary TB and loss of left renal function. About 2 months previously, the frequent urination had become more obvious; therefore, he presented to our hospital for examination and was diagnosed with bladder contracture.
Intravenous urography showed a normally functioning right solitary kidney. Computed tomography indicated a high-density mass in the abdominal cavity. Ultrasound examination showed a maximal bladder capacity of 25 mL, and the post-void residual urine volume was about 19 mL. At the patient’s request, robot-assisted laparoscopic augmentation ileocystoplasty and excision of the intraperitoneal mass were performed (Figures 1–3). The operative technique is outlined below.

Preoperative intravenous urography.

Preoperative computed tomography. M, mass.
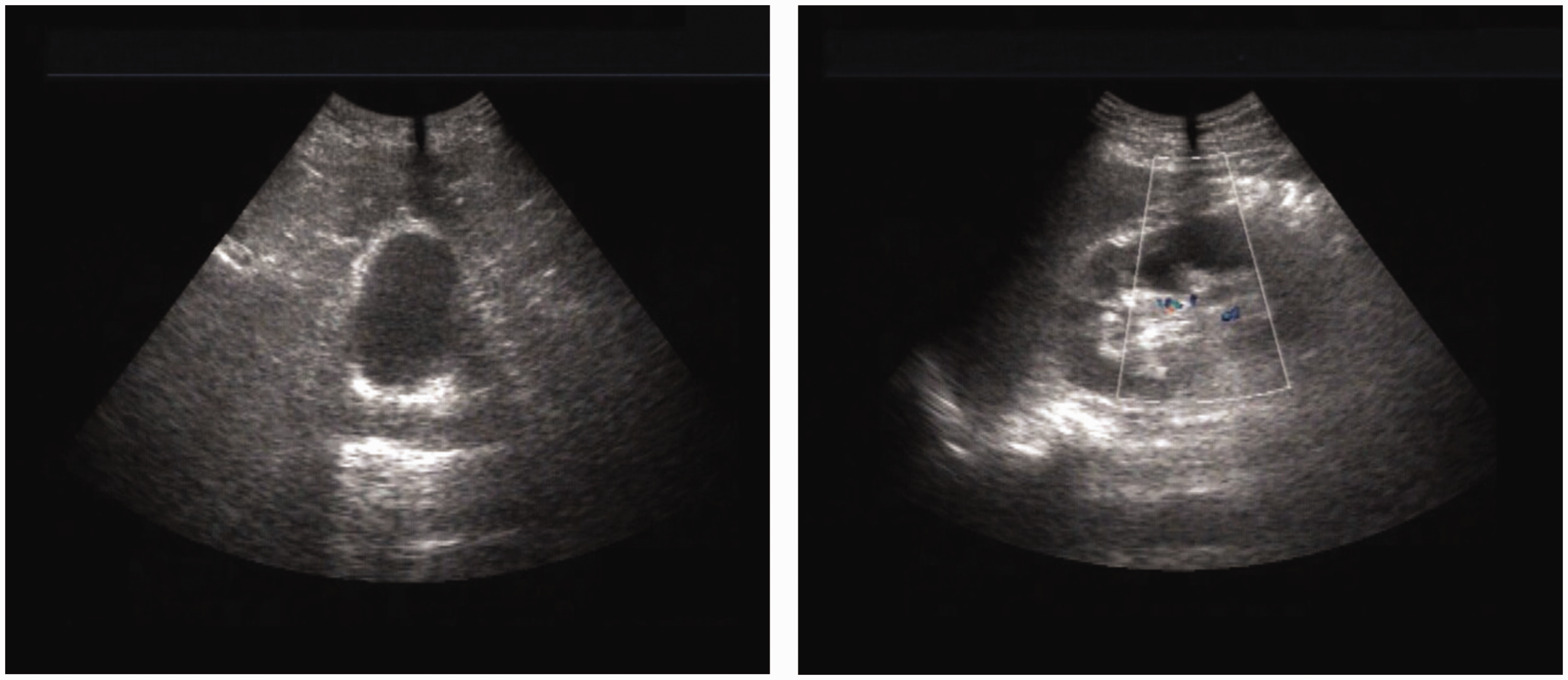
Preoperative ultrasound.

Port positions: A, assistant ports; R, robotic ports; C, camera port.
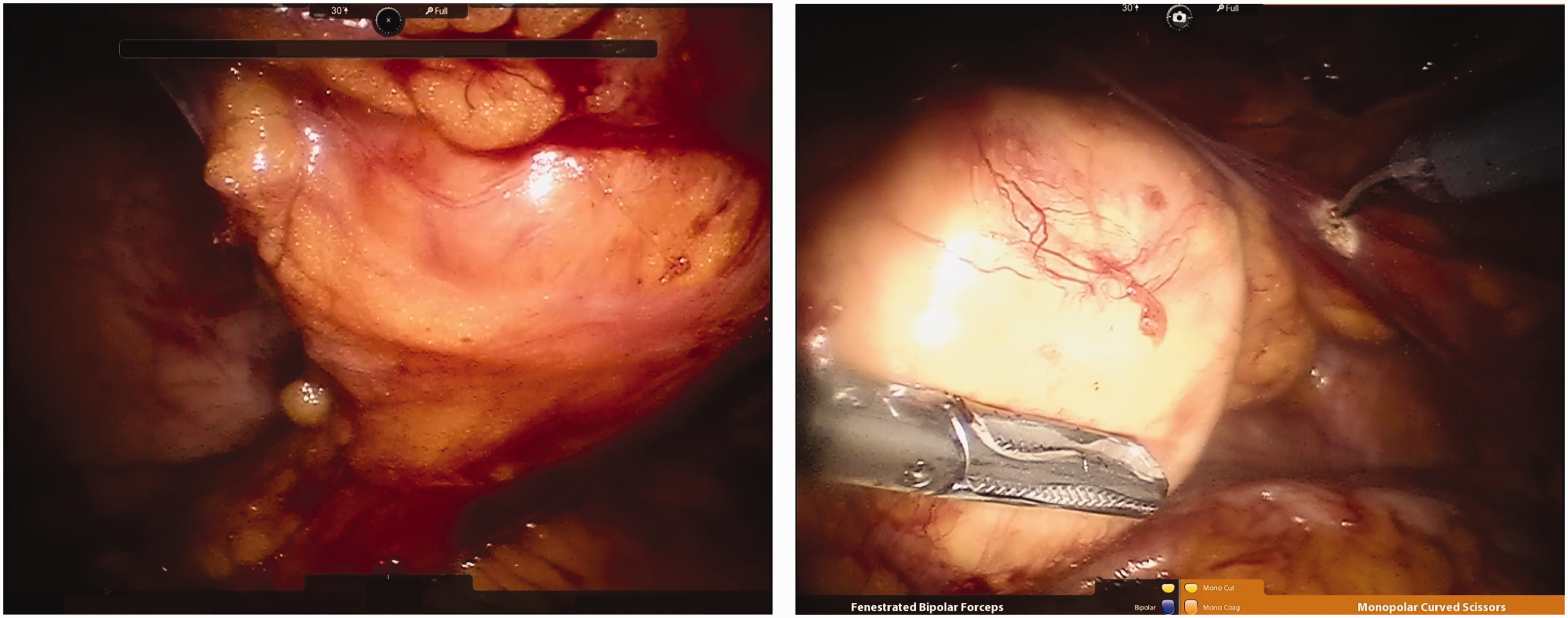
Excision of intraperitoneal mass.

(a) Cystostomy. (b) Isolation of ileal segment. (c, d) Creation of ileal pouch.

(a) Completion of the ileal pouch. (b) Suturing of the ileal segment and urinary bladder. (c) Completion of the ileocystoplasty. (d) Ileo–ileal anastomosis. UB, urinary bladder; I, ileal pouch.
Pneumoperitoneum and port insertion: Pneumoperitoneum was created using a Veress needle. Six ports, including three robotic ports, were inserted. Two assistant ports were used on the left side (12 and 5 mm). The robot was docked, and the procedure was begun (Figure 4).
Excision of intraperitoneal mass: The mass, about 4 cm in diameter, could be seen near the abdominal wall along the median line of the abdominal cavity. Space was present between the mass and its surrounding tissues. The mass was resected completely and sent for postoperative pathological examination of paraffin sections (Figure 5).
Cystostomy: The space of Retzius was developed, and the top wall of the bladder was fully freed. The bladder was distended by filling it with saline through the urethral catheter and then incised from the top wall.
Preparation of ileal patch: After locating the ileocecal junction, a 20-cm ileal segment was isolated by an ENDOPATH stapler (Ethicon Inc., Somerville, NJ, USA) about 15 cm from the ileocecal junction. The isolated ileal segment was incised vertically along the boundary of the opposite side of the mesentery. A U-shaped ileal pouch was formed by consecutive sutures of the medial and lateral sides of the incised phase.
Augmentation cystoplasty and enteric anastomosis: The ileal patch was sutured to the edges of the urinary bladder using 3-0 polyglactin sutures, starting from the posterior corner of the opened bladder and then progressing to the anterior corner in a continuous fashion from either side. The new bladder was reinforced by 3-0 absorbable interrupted sutures. Saline was injected into the new bladder through the urethral catheter, and no saline exosmosis was observed after filling the bladder. Ileo–ileal anastomosis was performed using the same endoscopic stapler, and the mesenteric window was closed with interrupted 3-0 silk sutures. An 18-French drain was kept in the pelvis behind the anastomosis (Figures 6 and 7).
No surgery-related intraoperative complications occurred. The operative time was 240 minutes, and the estimated blood loss was 100 mL. The length of hospital stay was 11 days. Microscopic examination of the biopsy specimen revealed fibrous tissue proliferation, vitreous degeneration, cholesterol crystallization, hemosiderosis, and calcification in the cyst wall. A large amount of coagulative necrotic tissue was seen in the cystic mass. Anti-acid staining suggested a tuberculous granuloma. At the 2-month follow-up, ultrasound examination showed that the maximal bladder capacity had increased to 1000 mL. Intravenous urography revealed no urine leakage, and filling of the bladder was satisfactory. The patient had no irritative urinary symptoms and was voiding with an insignificant post-void residual urine volume. The patient did not require intermittent catheterization and could effectively control his urination. At the 6-month follow-up, the patient’s symptoms of frequent urination and dysuria had greatly improved (Figures 8 and 9).

Postoperative intravenous urography.

Postoperative ultrasound.
The present study was approved by The Second Xiangya Hospital Bioethics Committee. The patients provided consent to publish the details and photographs of his case.
Discussion
With the development of laparoscopic technology, laparoscopic surgery has been increasingly applied in the field of urology. The first report of laparoscopic bladder augmentation was by Docimo et al., 4 who described a 17-year-old girl who underwent augmentation using the stomach in 1995. In 2000, Gill et al. 5 reported laparoscopic bladder augmentation in three patients in whom the pouch was prepared extracorporeally. Additionally, el-Feel et al. 6 reported that laparoscopic augmentation ileocystoplasty is a safe and technically feasible procedure; in their cases, bladder dissection and reconstruction of the ileovesical anastomosis were performed laparoscopically, whereas the ileal pouch was prepared extracorporeally. In 2002, Meng et al. 7 and Elliott et al. 8 reported complete laparoscopic enterocystoplasty; the procedure was completed in 9 h, and the bladder capacity measured 250 mL after 4 weeks and 550 mL after 8 months. Rebouças et al. 9 recently reported a case of pure laparoscopic augmentation ileocystoplasty in a patient with Guillain–Barre syndrome. The total operative time was 335 minutes. At the 6-month follow-up, a cystogram showed a significant improvement in bladder capacity. 9
With the advancements that have been made in technology, robotic surgery is now more widely used in urology. Pal and Koupparis 10 reported that robot-assisted surgery appears to be associated with reduced morbidity, less blood loss, a reduced length of hospital stay, and comparable clinical outcomes in comparison with the corresponding open procedures and a shorter operative duration and learning curve than the equivalent laparoscopic techniques. To the best of our knowledge, Al-Othman et al. 11 was the first to describe robot-assisted laparoscopic augmentation ileocystoplasty in which a completely intracorporeal technique was used to perform the procedure in 2008. In 2010, Kang et al. 12 reported a case of robot-assisted laparoscopic augmentation ileocystoplasty that was conducted in a patient with a neurogenic bladder after spinal cord injury. The procedure was completed in 300 minutes, the estimated blood loss was about 225 mL, and the bladder capacity increased to 350 mL during the seventh postoperative month. 12 In 2014, Dogra et al. 13 first reported a case of robot-assisted laparoscopic augmentation in a tubercular small contracted bladder. The total operative time was 420 minutes, and the estimated blood loss was 200 mL; the patient had no irritative urinary symptoms and was voiding with an insignificant post-void residual urine volume during the sixth postoperative month. 13 Caputo et al. 14 recently reported their experience with an adult with a low capacity bladder due to neurogenic bladder dysfunction. The operative time was 286 min, and the estimated blood loss was 50 mL. 14
Wiestma et al. 15 also reported a case of robot-assisted laparoscopic augmentation ileocystoplasty in a 6-year-old boy (18.5 kg body weight), demonstrating the feasibility and safety of an entirely intracorporeal approach in a pediatric patient. 15 Murthy et al. 16 and Barashi et al. 17 further proved the feasibility of robot-assisted laparoscopic augmentation ileocystoplasty in pediatric patients.
When compared with these previous cases of robot-assisted laparoscopic augmentation ileocystoplasty, the present case is unique in two notable ways. First, the preoperative maximal bladder capacity was 25 mL, which might be the smallest preoperative bladder capacity of all the cases ever reported. Second, this is the first case in which augmentation ileocystoplasty and excision of an intraperitoneal mass were combined with robot-assisted laparoscopy.
Conclusions
With advancements in technology and increases in surgeons’ experience in robotic surgery, it can be successfully used for less frequently performed procedures. In this case, we successfully performed robot-assisted laparoscopic augmentation ileocystoplasty and excision of an intraperitoneal mass. This case report contributes to the better characterization of this advanced surgical technique, which can be used by surgeons when necessary. More studies and reports of experiences are needed to further validate this advanced surgical technique.
Declaration of conflicting interest
The authors declare no conflicts of interest.
Footnotes
Funding
This study was supported by a grant from the Technological Innovation Guidance Plan (no. 2017sk50123) of Hunan Province. The funder had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
