Abstract
Sweet’s syndrome is a rare, acute febrile neutrophilic dermatosis sometimes associated with inflammatory diseases, infections, malignancies or the administration of pharmacotherapeutics. Very seldom, it occurs as an extraintestinal manifestation of Crohn’s disease. We present a unique combined diagnosis of recurrent Sweet’s syndrome associated with colovaginal fistulization, subsequently diagnosed as Crohn’s disease.
Keywords
Introduction
The association between Sweet’s syndrome and Crohn’s disease has rarely been observed since the original classification of this acute febrile neutrophilic dermatosis by Sweet in 1964. 1 Sweet’s syndrome classically presents as painful, erythematous, violaceous plaques or nodules on the upper torso and limbs. It is typically accompanied by signs of systemic inflammation such as fever, weakness, leukocytosis and elevated erythrocyte sedimentation rate. Further testing of patients with concomitant gastrointestinal symptoms can exceptionally lead to the diagnosis of inflammatory diseases such as Crohn’s disease. Understanding this association is crucial when first evaluating patients exhibiting skin lesions and associated gastrointestinal complaints. Systematic investigations optimize diagnosis, treatment planning and final prognosis of Sweet’s syndrome declaring with Crohn’s disease.
Case report
A 58-year-old Caucasian female displayed painful and pruritic, erythematous cutaneous lesions involving her palms, fingers, knees and calves. They occurred sporadically over the previous five years. She also complained of painful ulcerations on her lower lip, sore eyelids and photophobia. In addition, she suffered from intermittent abdominal pain with hematochezia, accompanied by loose stool vaginal discharge and generalized arthralgia for close to five years. She endorsed a significant loss of appetite, maintaining a liquid diet for nourishment. Otherwise, she described no other constitutional symptoms and denied any recent travels, sick contacts or consumption of unusual foods. She had no significant medical history and was prescribed naproxen for 14 days by her family physician with no resolution of the lesions.
Examination revealed a patient with Fitzpatrick skin phototype 2, warm to the touch although afebrile. Several regular, edematous, violaceous erythematous nodules and plaques, varying in size from 0.5 to 1.5 cm, with peripheral excoriations were noted on the volar and dorsal surfaces of both hands, including all fingers (Figure 1(a) and (b)). These lesions were accompanied by tenderness on palpation. Similar lesions were visible on both knees, calves and pretibial regions (Figure 1(c)). Multiple ulcerations coalesced on the mucosal aspect of her lower lip. Medial portions of both bulbar conjunctivae were erythematous with associated inflammation on the superior palpebral regions, bilaterally. Histopathology of both punch biopsies of the left thigh showed diffuse, dense dermal inflammatory neutrophilic infiltrates with sparse lymphocytes (Figure 2(a) and (b)), pathognomonic for Sweet’s syndrome. There was no evidence of atypia, vasculitis or granuloma formation.
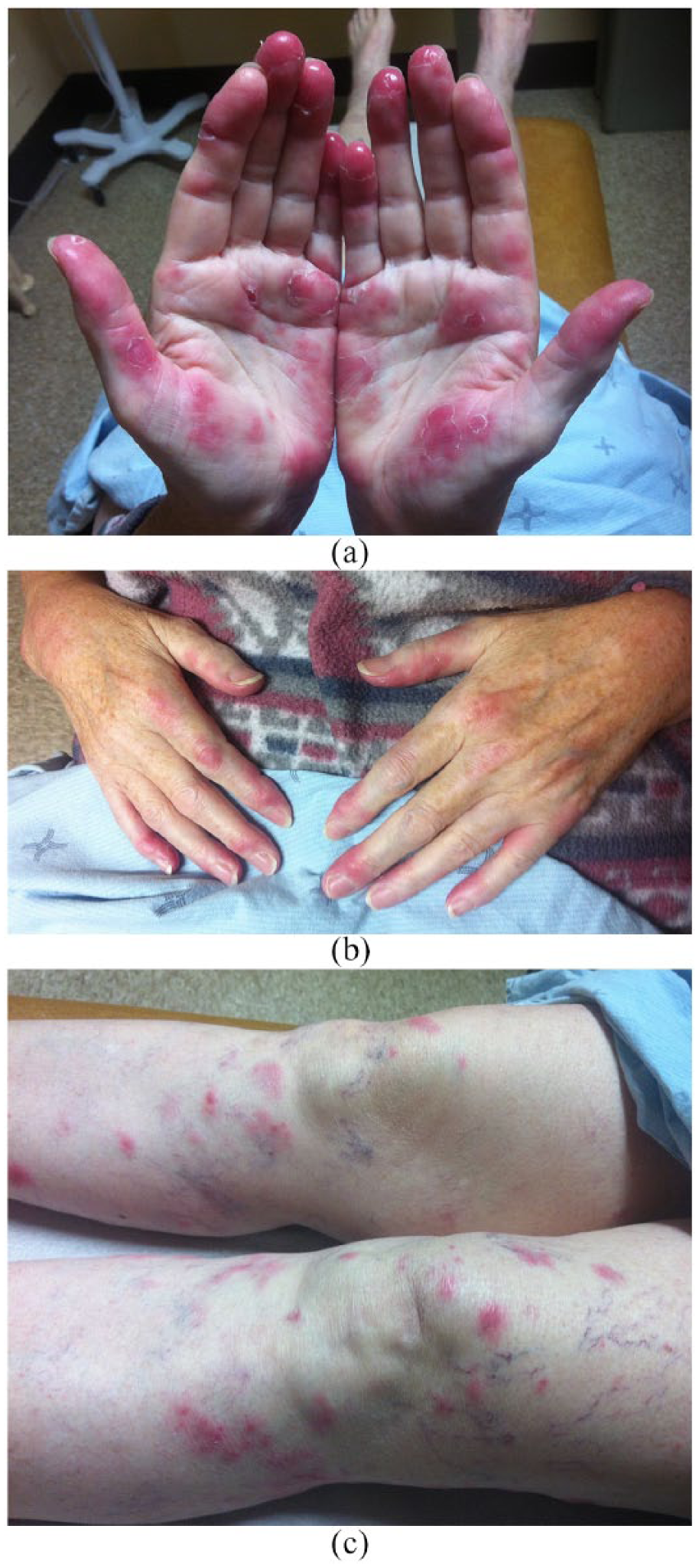
Erythematous, violaceous nodules and plaques (varying in size from 0.5 to 1.5 cm) on (a, b) hands and (c) knees/pretibial regions.

Diffuse, dense dermal inflammatory neutrophilic infiltrates with sparse lymphocytes and no evidence of vasculitis (hematoxylin–eosin stain; (a) ×4, (b) ×40).
Laboratory results revealed microcytic anemia (109 g/L with mean corpuscular volume (MCV) of 78.9 fL), leukocytosis (13,500/mL) with neutrophilia (11,400/mL), thrombocytosis (578,000/mL) and elevated erythrocyte sedimentation rate (ESR; 68 mm/h). Additional investigations including hepatitis B, hepatitis C, HIV, serum and urine protein electrophoresis with immunofixation, rheumatoid factor, thyroid function and urinalysis were within normal limits.
The patient was initially treated with 50 mg of oral prednisone daily which resolved her cutaneous eruption. However, the nodules and plaques recurred with titration to a 5-mg dose of oral prednisone.
Further investigations revealed a normal chest X-ray and mammogram. Pelvic ultrasound identified two small myometrial fibroids. Optometric evaluation determined the presence of a moderate Meibomian gland dysfunction with bilateral nasal conjunctivitis. There were no signs of ocular ulcerations or scarring. Computed tomography scan of the abdominal–pelvic region showed a possible intersphincteric fistula-in-ano which prompted an urgent gastroenterology consult. The colonoscopy revealed penetrating Crohn’s colitis with an underlying complex colovaginal fistula. This diagnosis was confirmed on histopathology of the colonic biopsy, which showed chronic, diffuse colitis with crypt abscesses (Figure 3). Prednisone was substituted for 50 mg oral azathioprine daily with complete resolution of the cutaneous lesions along with most of the gastrointestinal symptoms. Due to a significant risk of complications, the patient declined surgical treatment of her colovaginal fistula.

Chronic, diffuse colitis with crypt abscesses (×4).
Discussion
Sweet’s syndrome is a rare, acute febrile neutrophilic dermatosis typically affecting adults between the age of 30 and 50 years, with a slight predilection for women. 2 It can be categorized into three major groups: classical (idiopathic), malignancy-associated and drug-induced. 2 The classical variant can be associated with infections (upper respiratory or gastrointestinal), pregnancy and inflammatory diseases such as Crohn’s disease. Diagnosis is based on fulfilling both major criteria and two of four minor criteria 3 (Table 1). Sweet’s syndrome’s association with inflammatory bowel diseases such as Crohn’s disease is particularly rare.4,5 Historically, the diagnosis of Sweet’s precedes that of Crohn’s disease. 6 A biopsy-proven, concurrent diagnosis of Sweet’s with Crohn’s has not been documented to our knowledge. Although cases of fistulization have exceptionally occurred with Sweet’s associated inflammatory bowel disease, the presence of a colovaginal fistula has not been confirmed in the literature.
Diagnostic criteria for classical Sweet’s syndrome.

Adapted with permission by Elsevier publications and RightsLinkTM.
Fulfillment of both major criteria and two of four minor criteria is necessary to establish a diagnosis of Sweet’s syndrome.
Sweet’s syndrome can occasionally be associated with malignancies such as breast or genitourinary tumors and hematological disorders (acute myeloid leukemia and myelofibrosis). 7 Therefore, thorough investigations are justified, especially in patients exhibiting systemic symptoms. A mammogram and chest X-ray (to exclude lung malignancy and tuberculosis) should complement exhaustive blood work, including serology. A differential of rheumatoid neutrophilic dermatosis can be ruled out with a normal anti-rheumatoid factor antibody screening test. In patients complaining of gastrointestinal symptoms and loose stool vaginal discharge, an abdominal–pelvic computed tomography scan and colonoscopy should be performed to exclude inflammatory bowel disease, colorectal cancer or urogenital pathology. For patients experiencing mouth ulcers and inflamed erythematous conjunctivae, an urgent optical assessment is crucial in avoiding permanent vision loss attributed to scarring. Typically, patients respond well to corticosteroid treatment. With recalcitrant manifestations or combined Sweet’s/Crohn’s disease, the use of immunosuppressants such as azathioprine has shown promising results.
We present a unique case of a concurrent diagnosis of Sweet’s syndrome and Crohn’s disease featuring colovaginal fistulization. Clinicians should consider inflammatory bowel disease as a leading differential in patients exhibiting concurrent cutaneous and gastrointestinal complaints. Sweet’s associated Crohn’s disease may closely precede the diagnosis of malignancies. Therefore, rigorous investigations are warranted to reduce morbidity and mortality.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of the article.
Informed consent
The patient gave written informed consent for publication of the content and photos.
