Abstract
Objective
MicroRNA-590-5p (miR-590-5p) has been reported to stimulate osteoblast differentiation; however, its effect in diabetic osteoporosis remains unknown. This study investigated the effect of miR-590-5p on high glucose (HG)-suppressed osteoblast differentiation.
Methods
The effect of HG on MC3T3-E1 cell survival was assessed using the MTT assay. The expression levels and activities of osteoblastic proteins were evaluated by quantitative reverse transcription polymerase chain reaction (qRT-PCR), alkaline phosphatase (ALP) assay, and immunoblotting assay. Tumor growth factor-β (TGF-β) signaling in MC3T3-E1 cells was assessed using luciferase assay, qRT-PCR, and immunoblotting. Mineralized nodule formation in MC3T3-E1 cells was examined by using the mineralization assay.
Results
When MC3T3-E1 cells were exposed to HG conditions, there was significant downregulation of miR-590-5p and osteoblastic proteins (e.g., collagen I, Runx2, and ALP); in contrast, Smad7 was upregulated. Furthermore, miR-590-5p targeted Smad7 and inhibited its expression. Additionally, overexpression of miR-590-5p significantly promoted osteoblast growth and differentiation by upregulating TGF-β signaling in HG-treated MC3T3-E1 cells.
Conclusions
Collectively, the results showed that miR-590-5p was involved in osteogenesis; moreover, miR-590-5p may represent a potential target for the treatment of diabetic osteoporosis.
Keywords
Introduction
Osteoporosis is a bone disease characterized by reduced bone quantity and quality or increased fragility. 1 Complications from debilitated bone, such as reduced bone mineral density (BMD), bone quality, and strength, can contribute to an increased risk of fragility fractures. 2 Diabetes mellitus (DM) is a chronic medical disease that may impact osteoporosis, and may cause long-term microvascular and macrovascular complications. 3 DM reportedly patients tend to exhibit an increased incidence of osteoporosis, such that high glucose (HG) could inhibit osteogenic differentiation. 4 Recent studies have shown that, in HG conditions, reduced tumor growth factor-β (TGF-β) signaling in bone tissue is critical in the reduction of bone mass and fragility of diabetic osteoporosis (DO). 5 TGF-β signaling can activate two serine threonine kinase receptors (type I and type II), which phosphorylate intracellular Smad proteins. 5 Receptor Smads (e.g., Smad1, 2, 3, 5, and 8) can bind to Smad4 and undergo nuclear translocation, to initiate target gene transcription. 5 Conversely, TGF-β signaling can be inhibited by Smad6 and Smad7.
Many studies have extensively assessed the regulation of osteoblast differentiation by microRNAs (miRNAs) to evaluate their therapeutic potential in osteoporosis. 6 Notably, miRNAs target specific mRNAs involved in mediating multiple biological processes and pathologies, such as cell proliferation, differentiation, growth, and development.7,8 microRNA-590-5p (miR-590-5p) has been reported to stimulate osteoblast differentiation by indirectly protecting and stabilizing the runt-related transcription factor 2 (Runx2) protein through targeting of Smad7. 9 However, whether miR-590-5p mediates osteoblast growth and differentiation under diabetic conditions (e.g., HG conditions) remains unknown. In this study, we examined the expression levels of miR-590-5p in MC3T3-E1 osteoblasts exposed to HG conditions, and investigated the effects of miR-590-5p on osteoblast growth and differentiation, as well as the underlying mechanisms mediating these effects, in MC3T3-E1 osteoblasts exposed to HG conditions.
Materials and methods
Cells and chemicals
Mouse osteoblastic cell line MC3T3-E1 was purchased from American Type Culture Collection (Manassas, VA, USA). Cells were cultured in α-minimum essential medium (MEM) supplemented with 10% fetal bovine serum (FBS), and 1% penicillin/streptomycin (Beyotime, Nantong, China) in 5% CO2 at 37°C. Glucose was purchased from Sigma-Aldrich (St. Louis, MO, USA).
Cell viability
MC3T3-E1 cells were seeded into 96-well plates and incubated for 24 hours. Cells were subsequently starved overnight with 0.5% FBS, then cultured in 10% FBS containing normal glucose (5.5 mM) or HG (25.5 mM). Fourteen days later, cells were examined by MTT assay, in accordance with the manufacturer's instructions.
Quantitative reverse transcription polymerase chain reaction (qRT-PCR)
Total RNA was extracted using RNAiso Plus (Takara Bio Inc., Kusatsu, Japan), and cDNA was synthesized from equal quantities of total RNA using the PrimeScript™ RT reagent Kit (Takara Bio Inc.). To determine mRNA levels, qRT-PCR was performed using the SYBR Green qPCR Master Mix (Takara Bio Inc.). The primers used were as follows: collagenI sense: 5′-CATGGCCAAGAAG ACATCC-3′, antisense: 5′-CTCGGGTTT CCACGTCTC-3′; Runx2 sense: 5′-CCT TCCAGACCAGCAGCAG-3′, antisense: 5′-TCCGTCAGCGTCAACACCA-3′; Smad7 sense: 5′-TAGCCGACTCTGCGA ACTA-3′, antisense: 5′-AGATAATTCGT TCCCCCTGT-3′; and Gapdh sense: 5′-GCACCGTCAGGCTGAGAAC-3′, antisense: 5′-ATGGTGGTGAAGACGCCAG T-3′.
For evaluation of mature miRNA, miRNA was extracted using the MiRNeasy Mini Kit (Qiagen, Hilden, Germany), and the miRNA bulge-loop was reverse transcribed with a micro Script II RT kit (Qiagen). The primers used were as follows: miR-590-5p sense: 5′- TAGCCAGTCAGA AATGAGCTT-3′, antisense: 5′-TGCTG CATGTTTCAATCAGAGA-3′; and U6 sense: 5′- CTCGCTTCGGCAGCACA-3′, antisense: 5′-AACGCTTCACGAATTTGC GT-3′.
Immunoblotting
Immunoblotting was performed as described previously. 10 Whole-cell lysates were extracted by using RIPA lysis buffer (Beyotime). Equal amounts of total proteins were subjected to SDS-PAGE separation, followed by immunoblotting with specific antibodies. Anti-Collagen I and Runx2 antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA). Anti-Smad7, p-Smad2/3, Smad2/3, and GAPDH antibodies were purchased from Santa Cruz Biotechnology (Dallas, TX, USA). Anti-Myc tag antibody was purchased from Sigma-Aldrich.
Alkaline phosphatase (ALP) assay
The activity of ALP in culture supernatants was detected by examination of hydrolysis of p-nitrophenyl phosphate, in accordance with the manufacturer's instructions (Beyotime).
Luciferase assay
MC3T3-E1 cells were co-transfected with TGF-β-specific luciferase reporters (CAGA-Luc) and renilla plasmids by Lipofectamine® 2000 (Invitrogen, Carlsbad, CA, USA), in accordance with the manufacturer's instructions. Then, cells were prepared for luciferase assay using the Dual-Luciferase® Reporter Assay System, in accordance with the manufacturer’s instructions (Promega, Madison, WI, USA).
The predicted binding sites of miR-590-5p in the 3’UTR of Smad7 mRNA were identified by using microRNA.org and Pictar; fragments containing predicted miR-590-5p binding sites or mutants were amplified and subcloned into the pGL3 vector. Then, these plasmids were transfected into MC3T3-E1 cells along with pre-miR-590-5p or precon, using the Lipofectamine® 2000 Reagent (Invitrogen) in accordance with the manufacturer’s instructions, followed by luciferase assay.
Plasmid construction and gene transfection
The mouse Smad7 gene was amplified by PCR and cloned into the pcDNA3.1 vector with a Myc tag, as previously described. 11 The primers for Smad7 amplification were as follows: forward, 5′-ATGTTCAGGACCAAACGATCTGC-3′; reverse, 5′-CTACCG GCTGTTGAAGATGACCTC-3′. The plasmids were transfected into MC3T3-E1 cells by using Lipofectamine® 2000 (Invitrogen), in accordance with the manufacturer’s instructions.
The miR-590-5p mimic and a negative control (miR-NC) were purchased from Ambion (Carlsbad, CA, USA). miRNAs were transfected into MC3T3-E1 cells by Lipofectamine RNAiMAX Reagent (Invitrogen) in Opti-MEM (Invitrogen), in accordance with the manufacturer’s instructions.
Mineralization assay
Mineralized nodule formation was measured by using Alizarin Red staining, as described previously. 9 In brief, cells were rinsed twice with phosphate-buffered saline, then fixed with 4% paraformaldehyde for 30 minutes at room temperature. Subsequently, cells were stained with 2% Alizarin Red-S (Sigma-Aldrich) for 20 minutes, then extensively rinsed with distilled water.
Statistical analysis
Data are recorded as mean ± standard deviation of at least three independent experiments. All statistical analyses were conducted with Prism 7.0 (GraphPad, La Jolla, CA, USA). Differences between groups were examined by using Student's t-test; differences with p < 0.05 were considered to be statistically significant.
Results
miR-590-5p is downregulated in MC3T3-E1 osteoblasts in HG conditions
To investigate the effect of HG on cell survival and osteoblastic differentiation in MC3T3-E1 cells, cells were cultured in HG conditions or normal medium for 14 days. As shown in Figure 1a, the survival of MC3T3-E1 cells was remarkably reduced when cells were cultured in HG conditions (p < 0.01). Concurrently, expression levels and activities of osteoblastic proteins (e.g., Collagen I, Runx2, and ALP) were downregulated in HG-treated cells (p < 0.01), which indicated that HG suppressed cell differentiation in MC3T3-E1 cells (Figure 1b–1e). In addition, miR-590-5p was prominently reduced in MC3T3-E1 cells cultured in HG conditions (p < 0.01) (Figure 1f).

miR-590-5p is downregulated in MC3T3-E1 osteoblasts in high glucose conditions. (a) MC3T3-E1 cells were cultured in high glucose (HG) conditions (25.5 mM glucose) or normal medium (5.5 mM glucose) for 14 days, followed by MTT assay. (b, c) qRT-PCR was performed to evaluate expression of osteoblastic genes, including (B) collagen I (COL-I), and (C) runt-related transcription factor 2 (Runx2). (d) Culture supernatants of the above cells were prepared for ALP assay. (e) The above cells were lysed for immunoblotting against COL-I, Runx2, and GAPDH. (f) The indicated cells were prepared for qRT-PCR against miR-590-5p. U6 was used as an internal control. **p < 0.01.
HG suppresses TGF-β signaling by upregulating Smad7 expression in MC3T3-E1 osteoblasts
As shown in Figure 2a, a construct of signaling pathway-selective TGF-β firefly luciferase (CAGA-Luc) was made and transfected into MC3T3-E1 cells, which then underwent HG treatment. The luciferase assay showed that HG significantly suppressed TGF-β reporter activity (p < 0.01) (Figure 2a). Furthermore, an immunoblotting assay was performed to examine the expression levels of related proteins in the TGF-β signaling pathway. As shown in Figure 2b, the phosphorylation of Smad2/3 was significantly inhibited in HG-treated cells. However, Smad7, a negative mediator of TGF-β signaling, showed upregulated protein and mRNA levels in HG-treated cells (p < 0.01) (Figure 2b, 2c). Finally, the effect of Smad7 on TGF-β signaling inhibition was confirmed by immunoblotting and luciferase assay (p < 0.01) (Figure 2d, 2e).

High glucose exposure suppressed TGF-β signaling by upregulating Smad7 expression in MC3T3-E1 osteoblasts. (a) Luciferase constructs of signaling pathway-selective tumor growth factor-β (TGF-β) (CAGA-Luc) or empty vector (PGL3-Luc) were transfected into MC3T3-E1 cells for 24 hours under normal or high glucose (HG) conditions, followed by luciferase assay. (b) MC3T3-E1 cells were cultured in HG or normal conditions for 14 days, followed by immunoblotting against Smad7, p-Smad2/3, Smad2/3, and GAPDH. (c) The above cells were prepared for qRT-PCR against Smad7. GAPDH was used as an internal control. (d) Myc-Smad7 or empty vector plasmids were transfected into MC3T3-E1 cells for 48 hours, followed by immunoblotting against Myc, p-Smad2/3, Smad2/3, and GAPDH. (e) CAGA-Luc or PGL3-Luc, along with empty vector or Myc-Smad7, were transfected into MC3T3-E1 cells for 48 hours, followed by luciferase assay. **p < 0.01.
miR-590-5p targets Smad7 and regulates TGF-β signaling in MC3T3-E1 osteoblasts
As stated above, HG downregulated miR-590-5p, but upregulated Smad7 in MC3T3-E1 cells. Thus, we evaluated whether a relationship existed between miR-590-5p and Smad7. As shown in Figure 3a and 3b, we confirmed that Smad7 was a target of miR-590-5p. In HG conditions, mRNA and protein levels of elevated Smad7 were significantly attenuated by overexpression of miR-590-5p in MC3T3-E1 cells (p < 0.01) (Figure 3c, 3d). In addition, immunoblotting and luciferase assays clearly showed that overexpression of miR-590-5p enhanced the activity of the TGF-β signaling pathway in HG-treated MC3T3-E1 cells (p < 0.01) (Figure 3d, 3e).

miR-590-5p targets Smad7 and regulates TGF-β signaling in MC3T3-E1 osteoblasts. (a) Predicted binding sites of miR-590-5p in the 3’UTR of Smad7 mRNA. (b) miR-590-5p or mock vector (miR-NC), along with wild-type (WT) or mutant (Mut.) Smad7 3’UTR were co-transfected into MC3T3-E1 cells, followed by luciferase assay. (c) miR-NC and miR-590-5p mimic were transfected into MC3T3-E1 cells for 24 hours in high glucose (HG) conditions, followed by qRT-PCR against Smad7. GAPDH was used as an internal control. (d) The above cells were prepared for immunoblotting against Smad7, p-Smad2/3, Smad2/3, Runx2, Collagen I (COL-I), and GAPDH. (e) CAGA-Luc or PGL3-Luc, along with miR-NC or miR-590-5p mimic, were transfected into MC3T3-E1 cells for 48 hours, followed by luciferase assay. **p < 0.01.
miR-590-5p attenuates the inhibitory effect of HG on osteoblast growth and differentiation in MC3T3-E1 osteoblasts
miR-NC or miR-590-5p mimic constructs were used to detect whether miR-590-5p affected osteoblast growth and differentiation in HG-treated MC3T3-E1 osteoblasts. As shown in Figure 4a, overexpression of miR-590-5p attenuated the inhibitory effect of HG on osteoblast growth in MC3T3-E1 osteoblasts (p < 0.01). In addition, Alizarin Red staining clearly showed that HG suppressed the formation of mineralized nodules in MC3T3-E1 osteoblasts; however, the cells exhibited an increased number of nodules after overexpression of miR-590-5p (p < 0.01) (Figure 4b, 4c). Thus, we examined the expression levels of osteogenic genes to evaluate the status of osteoblast differentiation in MC3T3-E1 cells. As shown in Figure 4d and 4e, ALP assay and qRT-PCR revealed that overexpression of miR-590-5p attenuated the inhibitory effect of HG on ALP activity and Runx2 expression (p < 0.01); this indicated that miR-590-5p promoted osteoblast differentiation in HG-treated MC3T3-E1 cells.
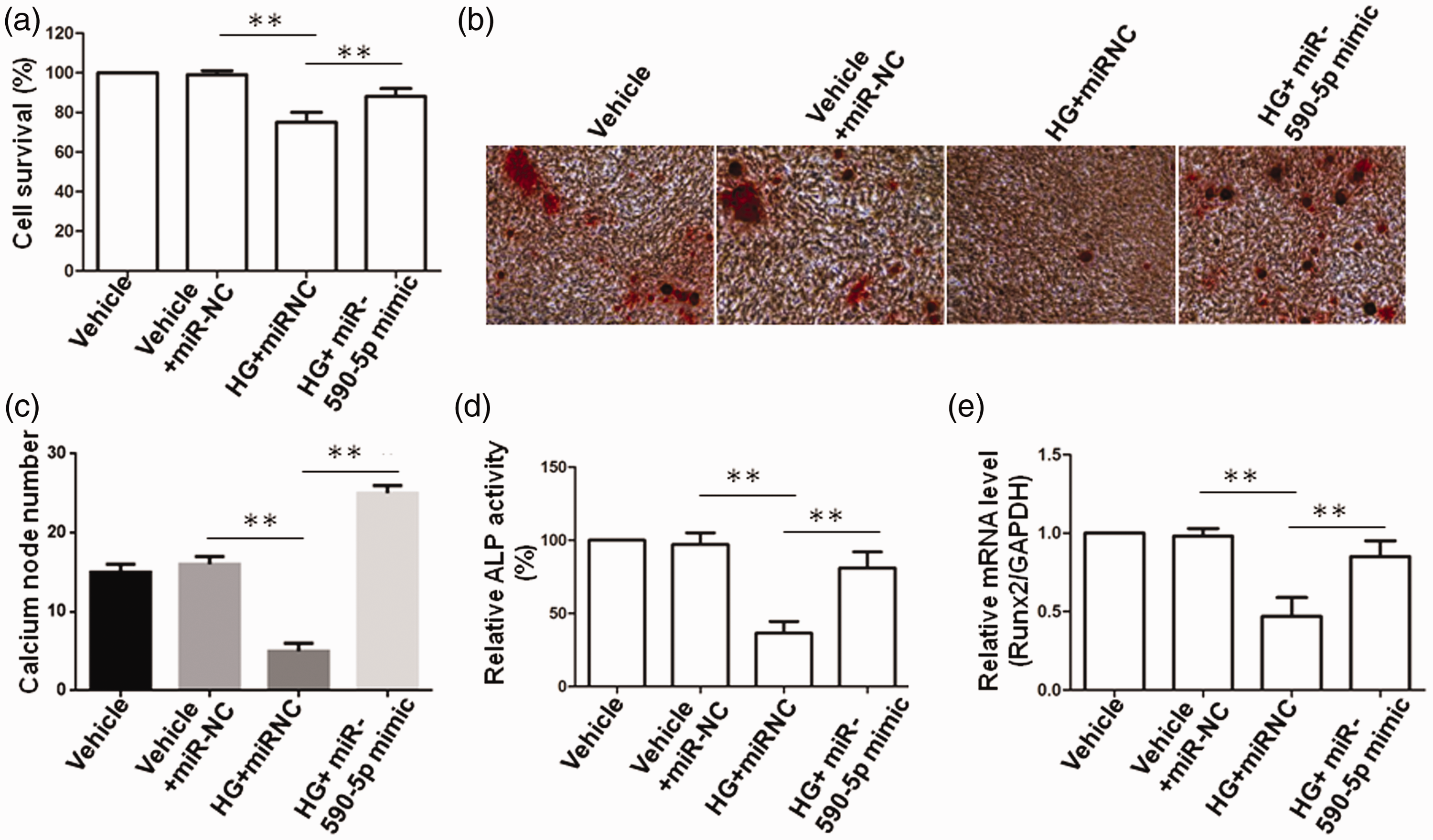
miRNA-590-5p attenuates the inhibitory effect of high glucose on osteoblast growth and differentiation in MC3T3-E1 osteoblasts. (a) MC3T3-E1 cells were cultured in high glucose (HG) conditions or normal medium for 14 days after the transfection of miR-NC or miR-590-5p mimic, followed by MTT assay. (b) MC3T3-E1 cells transfected with miR-NC or miR-590-5p mimic were cultured in HG for 21 days; then, mineralization of osteoblasts was depicted by Alizarin Red staining. (c) Statistical analysis of the data shown in (b). (d) MC3T3-E1 cells transfected with miR-NC or miR-590-5p mimic were cultured in HG conditions or normal medium for 14 days; then, culture supernatants were prepared for ALP assay. (e) The above cells were prepared for qRT-PCR against Runx2. GAPDH was used as an internal control. **p < 0.01.
Discussion
DM and osteoporosis are prevalent metabolic diseases, and an increasing number of individuals have been diagnosed with diabetic osteoporosis in the past several years. 12 Osteoblasts exert vital influence on bone formation, including synthesis and secretion of bone matrix.13,14 Increasing evidence indicates that ALP plays an important role in the development of calcification in vivo; moreover, its activity can reflect osteoblast maturity.15,16 In this study, to imitate the process that occurs in diabetic osteoporosis, HG was added during osteogenic differentiation of MC3T3-E1 osteoblastic cells. We found that ALP activity, as well as expression of various osteogenic genes, were reduced when MC3T3-E1 osteoblastic cells were cultured in HG conditions, indicating that osteoblast function was reduced.miR-590-5p has been shown to promote progression of several types of cancers (e.g., breast and cervical cancers); furthermore, TGFβR2 and collagen I are known target genes of miR-590-5p. 17 However, the expression level of miR-590-5p in the presence of HG is not yet known. In the present study, HG was used to stimulate MC3T3-E1 osteoblastic cells, which resulted in reduction of miR-590-5p; this suggested that, in HG conditions, miR-590-5p may play an important role in osteoblast function. In addition, TGF-β signaling and expression of Smad7 were downregulated in MC3T3-E1 osteoblastic cells exposed to HG. To identify the potential molecular mechanism by which miR-590-5p mediates osteogenic differentiation in HG conditions, the relationship between miR-590-5p and Smad7 was examined. The present study confirmed that miR-590-5p targeted Smad7 and regulated TGF-β signaling in MC3T3-E1 osteoblastic cells, which was consistent with the findings of previous studies.9,17
Runx2 is an important marker for early differentiation of osteoblasts, and is primarily expressed in osteogenic and multi-differentiated interstitial cells. 18 Smad7 is known to inhibit osteoblast differentiation via Smurf2-mediated Runx2 degradation. 9 Collagen I is another crucial indicator in the primary stages of osteoblast differentiation, which can also be regulated by Runx2. 19 In the present study, Runx2 and Collagen I were significantly reduced while Smad7 was increased in HG conditions; however, these expression patterns were reversed upon the addition of miR-590-5p mimic. Further studies demonstrated that miR-590-5p attenuated the inhibitory effect of HG on osteoblast growth and differentiation in MC3T3-E1 osteoblastic cells.
Collectively, the results of this study indicated that miR-590-5p attenuated the inhibitory effect of HG on osteoblast differentiation by suppressing the expression of Smad7 in MC3T3-E1 osteoblastic cells. This constitutes a novel insight into the function of upregulated miR-590-5p in the prevention and treatment of diabetic osteoporosis.
Footnotes
Acknowledgements
We wish to thank Dr. Jane Han for her discussion on this project.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
