Abstract
Objective
The present study aimed to identify Mollicutes infection in the reproductive system. We also examined the microbiological, biochemical, and antimicrobial profiles of Mollicutes infection, which are potentially associated with clinical reproductive abnormalities causing infertility in couples.
Methods
Thirty-seven couples who were attending an infertility clinic were enrolled. Detection of genital mycoplasmas was performed in cervicovaginal samples or male urethral swabs. Microbiological culture and biochemical and antimicrobial profiles were characterized using a Mycoplasma kit. The results were associated with reproductive abnormalities, as assessed by medical specialists from the infertility clinic.
Results
Up to 28.3% of all biological samples (n = 74) showed positive cultures. Bacterial isolates were Ureaplasma urealyticum (71.4%), Mycoplasma hominis (9.5%), or coinfections (19%). Most Mollicutes showed significant resistance to fluoroquinolones, macrolides, and tetracycline; and showed susceptibility to doxycycline, josamycin, and pristinamycin. The presence of resistant strains to any antibiotic was significantly associated with genital abnormalities (χ2 test, relative risk = 11.38 [95% confidence interval: 5.8–22.9]), particularly in women. The highest statistical association was found for macrolide-resistant strains.
Conclusion
The microbiological antibiotic resistance profile is epidemiologically associated with abnormalities of the reproductive system in couples attending an infertility clinic.
Keywords
Introduction
The World Health Organization, as well as the European Society of Human Reproduction and Embryology, define infertility as a disease of the reproductive system that leads to the inability to achieve a pregnancy after 12 months or longer with unprotected sex. Infertility is currently a problem of global distribution and increasing magnitude. An estimated 20% to 35% of all couples who wish to procreate have an infertility disorder. 1
The main causes of infertility in couples include female endocrine disorders, ovulatory dysfunction, tubal and peritoneal diseases, and male endocrine and testicular disorders and seminal obstruction caused by sexually transmitted infections. Notably, the male and female reproductive systems are suitable for growth of various pathogenic and non-pathogenic microorganisms. 2
Genital mycoplasmas belong to an independent taxonomic class called Mollicutes and Mycoplasmataceae. This bacterial family includes Mycoplasma spp., Mycoplasma hominis, Mycoplasma genitalium, and Ureaplasma urealyticum. These species may cause oligosymptomatic genital infections in women and men of reproductive age, such as urethritis, urinary tract infections, chorioamnionitis, pelvic inflammatory disease, and sperm cell disorders. Complications from these infections may lead to infertility.3,4
In Mexico, the National Health System has recognized the importance of infertility as a public health problem. However, only a few studies have addressed infertility causes, and mainly evaluated non-infectious etiologies. Therefore, evaluating the potential participation of sexually transmitted infections is important because Mollicutes, such as U. urealyticum and M. hominis, have been identified in semen samples and endocervical exudates from Mexican couples with infertility. 5 Furthermore, M. hominis has been reported in 24.2% of infertile males with sperm abnormalities, whereas U. urealyticum has been reported in 86.8% of infertile females with tubular abnormalities. 6
Antimicrobial therapy for Mollicutes genital mycoplasmas includes quinolones, tetracyclines, and macrolides as the drugs of choice. However, the indiscriminate use of antibiotics has promoted development of antibiotic-resistant strains. 7 Nonetheless, characterization of antibiotic resistance and its association with reproductive disorders in Mexican couples with infertility remain unclear. Therefore, this study aimed to identify Mollicutes infection in the reproductive system, as well as characterize its microbiological, biochemical, and antimicrobial profiles.
Patients and methods
Study population
In this cross-sectional study, 37 infertile couples were included from the Department of Assisted Reproduction National Medical Center “20 de Noviembre”, ISSSTE, Mexico City, Mexico between August 2015 and February 2016. Characteristics of the population, such as age, age at initiation of sexual activity, number of sexual partners, marital status, and infertility time, were collected from clinical records. Patients with anatomical alterations, genetic diseases, and endocrine disorders, and couples under antimicrobial treatment were excluded for at least 2 weeks before recruitment.
This study was designed and performed according to ethical guidelines of the 1975 Declaration of Helsinki. The study was approved by the Local Committees of Research, Ethics in Research, and Biosafety of the Centro Médico Nacional “20 de Noviembre”, ISSSTE, Mexico City (Protocol ID No. 033.2013). All participants provided written informed consent.
Cervicovaginal samples
Samples were collected by specialized and experienced medical personnel according to the routine procedure in the Department of Reproductive Biology. For cervicovaginal exudate, the sample was taken by placing the patient in a gynecological position, and a sterile mirror was inserted into the vagina, which was lubricated with sterile water and heated to 37°C if necessary. The use of commercial lubricants or an antiseptic was avoided. Once the cervix was located, the mirror was fixed by opening the valves and a vaginal sac or cervix sample was taken, as appropriate. During routine sampling, two swabs were obtained for replicates of the samples. With the first swab, direct smears were stained with the Gram technique (to evaluate the Nugent criteria). The second swab was used for transport media R1 broth (Mycoplasma Gallery IST2) to detect the presence of mycoplasmas. 8
Urethral swabs
Patients were asked to retract the penis foreskin and keep it in this position throughout the procedure. The medical personnel used sterile cotton or gauze to clean the opening of the urethra at the tip of the penis. To collect the sample, a cotton applicator (cotton swab) was gently inserted approximately 2 cm into the urethra and turned. To obtain a good sample, the test was performed at least 2 hours after urinating. The swab was placed in R1 broth (Mycoplasma Gallery IST2) to initiate isolation of mycoplasmas.
Microbiological culture
The liquid media for U. urealyticum and M. hominis were inoculated from the transport medium R1 (Mycoplasma IST2 kit, Biomérieux, Marcy l'Etoile, France) A total of 150 µL of the broth transport medium R1, containing the clinical sample, were inoculated. To perform phenotypic identification of U. urealyticum, urea broth was used, which contained medium base (pleuropneumonia-like organism broth), yeast extract, horse serum, and urea. To determine growth of this microorganism, phenol red was added to the culture medium because it turns from red to intense raspberry red in the presence of urease and ammonium production. Similarly, culture medium specific for M. hominis had arginine added. When metabolized, arginine produces an alkaline compound that turns phenol red to raspberry red. Culture media were incubated at 37°C until the phenol red indicator changed color. Solid cultures were performed using a rich medium supplemented with horse serum, yeast extract, and cysteine, and either urea substrates for U. urealyticum or arginine for M. hominis was added. Culture media were inoculated by adding three drops of 5 µL each. The inoculated plates were incubated at 37°C in 5% CO2, and were checked daily for colonial morphology of a “fried egg” for M. hominis or a “sea urchin” for U. urealyticum. A culture was considered negative if no colony growth was obtained after 15 days of incubation. 9 Strategies to minimize the chance of non-specific results included the following: 1) addition of polymyxin B, amphotericin B, and penicillin to cultures; 2) parallel assays using positive control of Mollicutes, and 3) all of the growth obtained was further characterized by the IST2 gallery to evaluate specificity.
Biochemical characterization
The Mycoplasma IST2 gallery was used according to the manufacturer’s instructions as follows. A swab with the sample was placed in the transport medium R1 broth (3 mL). The broth was mixed with the lyophilisate R2 provided by the gallery, and this contained the substrates necessary for development of microorganisms. A volume of 50 µL was distributed to each of the domes of the 22 tests that were divided into three sections. In the first section, phenotypic detection of M. hominis and U. urealyticum was performed. In the second section, microorganisms were quantified, with determination of whether the sample contained a concentration > 104 change in color units (CCU)/sample. This indicated the importance of the presence of these microorganisms. In the last section, sensitivity tests were performed for different antimicrobials, and sensitivity was indicated by a change in color from yellow to red. Finally, after inoculation of each of the domes, two or three drops of sterile mineral oil were added. The gallery was incubated at 37°C and the results were recorded at 24 and 48 hours. 10
Clinical evaluation
Hysterosalpingography was performed in a radiology room. The patients lay on a table under an X-ray machine and placed their feet in stirrups. An instrument called a speculum was inserted into the vagina. After cleaning the cervix, the doctor passed a thin tube (catheter) through it. A dye (contrast medium) was passed through this tube, and it filled the uterus and fallopian tubes. X-rays were then taken. This contrast medium enabled these areas to be viewed on X-rays. 11 In spermatobioscopy, the patient was asked for at least 3 days of sexual abstinence and not to perform intense physical exercise days before the analysis. The patient was required to masturbate in a clean room near the laboratory to process the sample and thus avoid thermal shock. A macroscopic study of the semen was performed to evaluate liquefaction, appearance, volume, pH, and viscosity. A microscopic study was performed later to evaluate sperm concentrations, presence of leukocytes, motility, vitality, and sperm morphology. 12 All of the study population was routinely searched for other genital pathogens, such as Neisseria and Chlamydia, with negative results.
Statistical analysis
Data were analyzed using the Statistical Package for Social Sciences v.18.0. (SPSS Inc., Chicago, IL, USA) for Windows. A P value ≤ 0.05 (two-tailed) was considered to be statistically significant. Qualitative variables are described as mean and standard deviation, and were compared using the Student’s t-test. The chi-square test was calculated in the independence test of qualitative random variables.
Results
The study population consisted of 74 patients (37 men and 37 women) who were recruited from the infertility clinic, aged 31 to 34 years old. Most of the patients had begun an active sexual life from the age of 18 years old. The most frequent number of sexual partners was more than three. There was a significant difference between sexual practices of men and women (p < 0.001). A significantly higher proportion of men showed asymptomatic infections then did women (p < 0.001). The most prevalent genitourinary symptom was secretion (women) and dysuria (men). For the time of infertility, most patients showed longer than 5 years. The detailed sociodemographic characteristics are shown in Table 1.
Demographic and clinical characteristics of couples with infertility.

Quantitative data are shown as mean ± standard deviation. Qualitative data are shown as number (%). #Determined by the number of couples. Statistical analysis was performed with Fisher’s test. *Symptomatic was considered as any combination of secretion, itching, and/or burning when urinating.
Characterization of specimens
Seventy-four samples of cervical and urethral exudates were collected, and the analysis included two methods of characterization: microbiological (culture conditions, Nugent criteria occurred in < 10%, and related to M. hominis) and biochemical (by the Mycoplasma IST2 kit) (Figure 1). By using these approaches, we observed a 100% diagnostic agreement between both methods, where genital mycoplasmas were isolated from 21 (28.3%) samples, either as a single microorganism or as a coinfection with both types of genital mycoplasmas. Notably, U. urealyticum was found in 15 of 21 samples as a single infective bacterium. U. urealyticum was found as a coinfection with M. hominis in four samples. M. hominis was found in two samples.

Characterization of clinical specimens. Left panels: bacteria growing in pleuropneumonia-like organism medium. (a) Mycoplasma hominis shows a characteristic “fried egg” shape. (b) Ureaplasma urealyticum shows a “sea urchin” shape. Right panels: Biochemical characterization using the Mycoplasma IST2 kit. The first three domes on the left are intended for identification purposes. The next two domes are intended for quantification purposes. The last nine domes are intended for characterization of antibiotic resistance, according to color development: yellow (susceptible) or red (resistant).
The presence of genital mycoplasmas, as a shared infection between couples, was observed in 76% of patients. Notably, genital mycoplasmas showed a threshold concentration of ≥104 CCU, which indicated active infection in all of the patients.
Additionally, biochemical characterization was performed to detect antibiotic resistance profiles of the microorganisms. These microorganisms included fluoroquinolone-resistant strains of U. urealyticum, particularly to ciprofloxacin (86.6%) and ofloxacin (80%), whereas they were highly sensitive to doxycycline, josamycin, and pristinamycin. M. hominis strains were completely resistant to macrolides (erythromycin, tetracycline, clarithromycin, and azithromycin). However, fluoroquinolone resistance was found in 50% of cases and the strains were highly susceptible to doxycycline, josamycin, and pristinamycin. When U. urealyticum and M. hominis were found as coinfection, resistance to most antibiotics was observed, mainly to fluoroquinolones and macrolides (Table 2).
Ureaplasma urealyticum and Mycoplasma hominis antimicrobial profile of all samples.
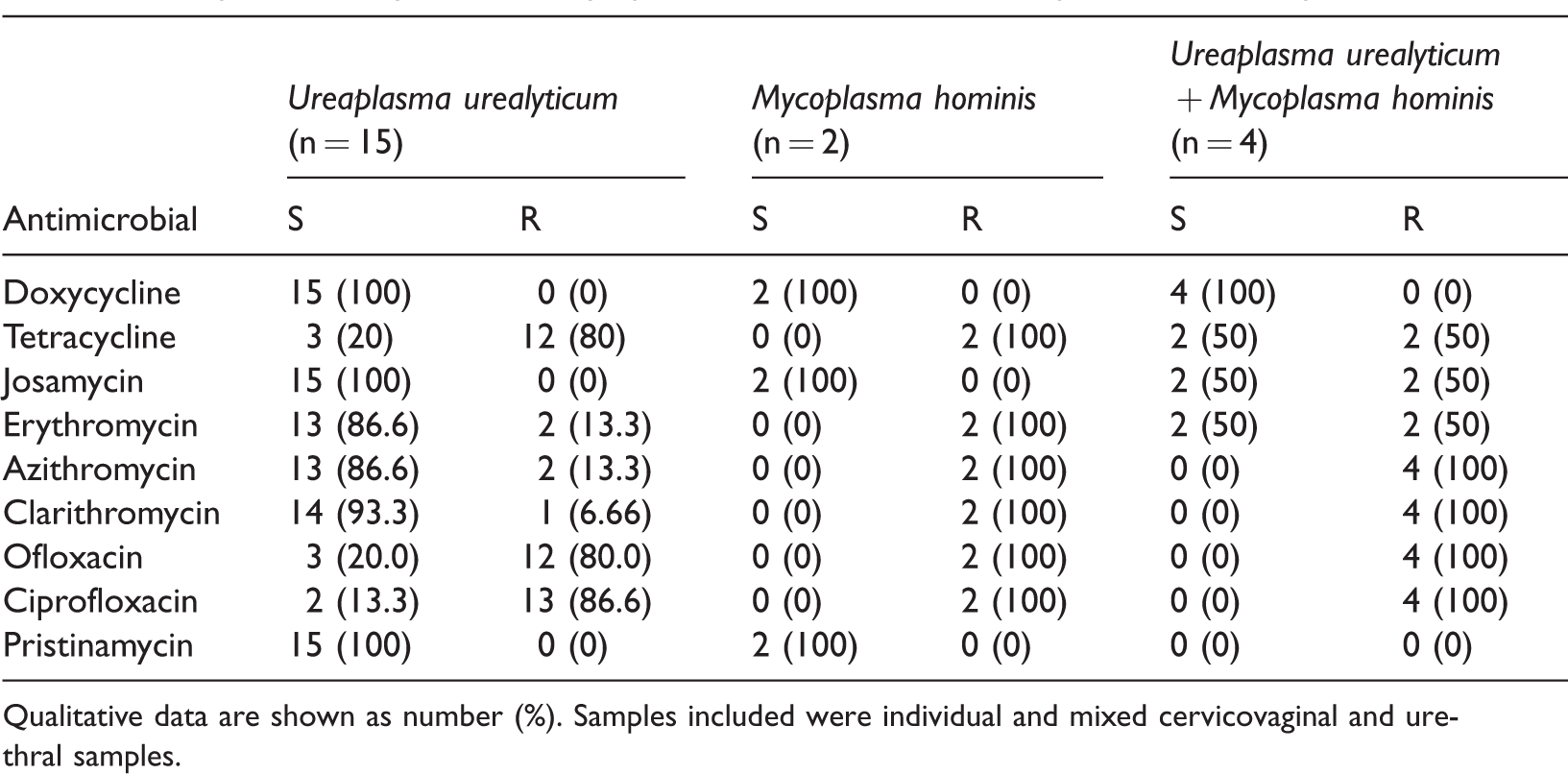
Qualitative data are shown as number (%). Samples included were individual and mixed cervicovaginal and urethral samples.
Association of antibiotic resistance in Mollicutes with the clinical condition of patients with infertility
Clinical conditions that are associated with infertility in males, such as asthenospermia and azoospermia, showed a higher prevalence in the population who was infected with genital mycoplasmas (both p < 0.001). However, those without genital mycoplasma infection did not present with any abnormalities. In women, unilateral and bilateral tubular obstruction, premature rupture of the fetal membranes, and ectopic pregnancies occurred significantly more frequently in patients who were positive for genital mycoplasma infection than in those who were not positive for genital mycoplasma infection (all p < 0.001) (Table 3).
Clinical condition associated with genital mycoplasma infection (n = 37).

Data are shown as number (%). The symbols indicate conditions associated with infection of genital mycoplasmas in men (*) and women (#), p < 0.001. AS, asthenospermia; AZ, azoospermia; OL, oligoasthenozoospermia; OL1, oligoasthenoteratozoospermia; TO, tubular obstruction; EP, ectopic pregnancy; PRM, premature rupture of the membranes; NCE, nonspecific chronic endocervicitis; C, cervicitis; U., Ureaplasma; M., Mycoplasma.
Furthermore, a higher prevalence and significant association (chi-square test, p < 0.0001) was found between microorganisms that were resistant to any antibiotic and the presence of any genital abnormality (relative risk [RR] = 11.38, 95% confidence interval [CI]: 5.8–22.9, p < 0.0001, Appendix 1). This finding was also observed for most of the antibiotic groups tested, including tetracyclines (RR = 6.5, 95% CI: 2.1–19.5, p < 0.0001), macrolides (RR = 37.7, 95% CI: 5.25–271, p < 0.0001), quinolones (RR = 1.1, 95% CI: 0.81–1.6, p = 0.25), and pristinamycin (RR = 9, 95% CI: 0.85–94.9, p = 0.19).
Discussion
The main finding of the present study was the higher prevalence of Mollicutes that was resistant to any antibiotic within the group of patients with genital abnormalities among couples who were attending an infertility clinic. The study population was characterized by couples aged 31 to 34 years. There was a significant difference between sexual practices of men and women. More than two thirds of men showed asymptomatic infections and approximately three quarters of the women were symptomatic. The higher prevalence of infection in women with identifiable clinical conditions related to infertility is consistent with previous reports.13,14 This finding supports the epidemiological association between Mollicutes infection and infertility in women.
Similar to previous studies, we observed that U. urealyticum was the most prevalent (71%) species that was isolated in samples of the cervix and urethra from the studied couples. The reported prevalence of U. urealyticum cervical infection in similar populations varies from 50% to 80%, 15 whereas its prevalence may reach up to 90% in women who are sexually active. M. hominis infection was the next most prevalent infection in our study. The presence of M. hominis is relevant because it contributes to polybacterial infection known as “bacterial vaginosis”. This condition is considered a dysbiosis related to the absence of Lactobacilli and an increase in vaginal pH that favors further infections. In fact, synergism between M. hominis and other microorganisms causing bacterial vaginosis has been suggested. Similarly, studies in the Italian population have reported a higher prevalence of U. urealyticum and M. hominis, and a possible association of M. hominis with bacterial vaginosis.16–20
In some patients, we observed coinfection of Ureaplasma spp. with M. hominis, which is expected. Ureaplasma spp. and M. hominis may become opportunistic agents under immunocompromising situations, and rapidly increase their bacterial load (determined by quantitative PCR and/or microbiological culture). This is considered a useful marker of clinically significant infection. 21 Furthermore, coinfection with other microorganisms is considered important information because of potential interactions with Mollicutes. An example of this situation is that coinfection of human papilloma virus and Mollicutes has been suggested to accelerate tissue damage, leading to organ dysfunction.
M. hominis and Ureaplasma spp. have been implicated in a wide variety of infections that may lead to infertility. Female chronic genital infection might induce adhesions to the inside and outside of fallopian tubes. This results in obstruction of the fallopian tube and is an obstacle for union of the sperm and egg. Infection is also related to ectopic pregnancies and premature rupture of the membranes, leading to a lower possibility of pregnancy. This is consistent with our finding of a higher prevalence of genital Mollicutes in women who had any reproductive abnormality. Similarly, a Danish study showed a significant correlation between the presence of M. hominis and U. urealyticum with tubal abnormalities in a population of 304 infertile women. 22 Genito-urinary infections, such as urethritis, prostatitis, and other inflammatory processes, are also related to 15% of male infertility. 23 Mollicutes is particularly associated with sperm abnormalities, such as aberrant motility, deficient mitochondrial function, and loss of DNA integrity.24–27 Increased formation of hydrogen peroxide and reactive oxygen species might induce peroxidation of sperm lipids, which causes an internal alteration that makes them incapable of moving. 28 U. urealyticum has been described as the most frequent species related to male reproductive system abnormalities.29–31 Consistently, U. urealyticum is the most frequent species in men with infertility; although most of our male affected population did not show any genital infections. This finding is in contrast to a previous report, which showed that the presence of U. urealyticum and M. hominis was related to impaired density of semen, and abnormal sperm vitality and motility in infertile men.8,32 Such differences in observations may be explained by methodological variations in the tests used to evaluate sperm.
With regard to the antibiotic profile, U. urealyticum isolates showed a high rate of fluoroquinolone resistance; while M. hominis showed resistance to macrolides. These profiles are consistent with previous reports9–33 and may be related to the widespread use of fluoroquinolones within the Mexican population, especially as therapy for urinary infections. 33 Moreover, Mexican official therapeutic guidelines 32 recommend that symptomatic sexually transmitted infections (where Neisseria gonorrhoeae, Chlamydia trachomatis, M. hominis, U. urealyticum, and other facultative anaerobic bacteria have been identified by culture media or quantitative PCR) be treated with fluoroquinolones or clindamycin for ambulatory patients, or cephalosporin and doxycycline in case of in-hospital treatment. This may affect prognosis of the ambulatory population because of the antibiotic resistance profile, which may lead to additional resistance to antimicrobials, therapeutic failure, and chronic infection with potential complications. Interestingly, U. urealyticum and M. hominis isolates were highly sensitive to agents, such as doxycycline, josamycin, and pristinamycin, which is consistent with the pattern of antibiotic resistance found in other populations.11,12,18,34 Generally, our findings support the benefit of routine isolation of microorganisms as part of a comprehensive evaluation and therapy during infertility studies.
Interestingly, we found that the presence of antibiotic-resistant strains of genital Mollicutes were associated with reproductive abnormalities in the study population, particularly in women. Previous reports have described the distribution of antibiotic-resistant strains of genital Mollicutes in populations with infertility.35–38 However, to the best of our knowledge, this is the first study to evaluate the role of bacterial antibiotic resistance in clinical reproductive abnormalities in couples attending an infertility clinic. Although the precise mechanism remains unclear, we believe that a relation of antibiotic-resistant strains with reproductive abnormalities and infertility is feasible. This is because novel mechanisms of antibiotic-resistant bacteria suggest induction of microbiota dysbiosis, which facilitates bacterial migration and translocation, and is potentially associated with higher damage in reproductive organs.39,40
Antimicrobial resistance profile and genital abnormalities.

Qualitative data are shown as number (%). Samples included were individual and mixed cervicovaginal and urethral samples. U., Ureaplasma; M., Mycoplasma; S, sensitive; R, resistant.
Footnotes
Acknowledgments
We would like to thank Sofia Mulia for kindly correcting the style of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the E-015 institutional program and Secretaría de Investigación y Posgrado del Instituto Politécnico Nacional (SIP 20171099 and 20180060) (IPN). Brenda Maldonado Arriaga had a scholarship from CONACyT. Graciela Castro Escarpulli received support from Estímulos al Desempeño en Investigación, Comisión y Fomento de Actividades Académicas (Instituto Politécnico Nacional), and Sistema Nacional de Investigadores (SNI, CONACyT).
