Abstract
Bony erosion and dural ectasia rarely occur in the posterior part of the vertebral body in ankylosing spondylitis (AS). Because of the compression of nerve tissue that adheres to the inflamed dura mater, a range of neurologic symptoms may develop. Few studies have investigated therapies for this condition, and surgical intervention might alleviate the neurologic dysfunction. Here, we report an uncommon case of lumbar spinal cord herniation secondary to anterior dural ectasia in a patient with AS. The patient had a 31-year history of AS and presented with progressive numbness and weakness of his left leg that had lasted for nearly 1 month. Magnetic resonance imaging (MRI) revealed a dural defect in the posterior aspect of the L1 and L2 vertebral bodies, with displacement of the nerve tissue adjacent to the lytic lesion. The patient declined surgery and was prescribed oral meloxicam. The neurological symptoms of the left leg remained stable during 2 years of follow-up. The dural enhancement on MRI resolved, although the erosion-like lesion in the vertebral bodies persisted. This unusual case supports the central role of inflammation in anterior dural ectasia of patients with AS. Anti-inflammatory treatment may be an alternative therapy for patients who decline surgery.
Keywords
Introduction
New bone formation in the spine and sacroiliac joints is a hallmark of ankylosing spondylitis (AS), a chronic inflammatory disease. Bony erosion in the posterior part of the vertebral body is rare in patients with AS. 1 The spinal cord may be compressed and adhere to the dura mater in the erosion-like lesion, which is known as spinal cord herniation (SCH) through the anterior dural ectasia. This complication of AS leads to a wide range of neurologic comorbidities. 2
Surgical treatment might alleviate the neurologic dysfunction that results from SCH through the dural ectasia. 3 Herein, we present an uncommon case of anterior dural ectasia at the L1 and L2 vertebrae in a patient with AS. Interestingly, despite radiographic changes in the vertebral lesion, neurological deficits of our patient remained stable throughout the 2-year follow-up without surgical intervention.
Case report
The ethics committee of Guangdong Second Provincial General Hospital approved this study (No. 2018-FSKWZ-011). Written informed consent was obtained from the patient for publication of this report and any accompanying images before submission.
A 44-year-old man with a 31-year history of AS was admitted to our rheumatology service because of progressive numbness and weakness of his left leg that had lasted for approximately 1 month. After being diagnosed with AS at the age of 13, our patient had seldom visited his physician but he used painkillers as needed. The patient denied any history of trauma or surgery.
Upon physical examination, the patient had a positive chest expansion test, with chest expansion of 1 cm (normal range: ≥5 cm), and motion of the thoracic and lumbar spine was severely decreased. The patient also had a positive Schober’s test, with only 0.5 cm of extension (normal range: ≥5 cm), with dorsal kyphosis. Neurological examination showed significant wasting of the muscles in his left leg but normal muscle tone. A strength exam revealed weakness in the left lower extremity (grade 4/5 of the iliopsoas and quadriceps muscles and grade 1/5 of the dorsiflexors and plantar flexors) upon manual muscle testing. Knee jerk and ankle jerk reflexes were normal on the left side, and the Babinski response was negative. Sensation to pinprick and touch was impaired in the left L5–S1 dermatomal distribution. These findings were suggestive of predominantly left-sided L5–S1 radiculopathy with some proximal extension.
The patient subsequently underwent a series of diagnostic tests. Whole blood count, serum creatinine, transaminases, calcium, and alkaline phosphatase were within normal limits. Systemic inflammation was not evident, with an erythrocyte sedimentation rate of 11 mm/hour and C-reactive protein concentration of 1.6 mg/L. Both the human leukocyte antigen B27 and tuberculosis-specific enzyme-linked immunospot assays were negative.
A plain radiograph of the thoracolumbar spine revealed bony bridging and squaring of the vertebral bodies, which are typical features of AS (Figure 1). A computed tomography (CT) scan of the lumbar spine displayed well-defined erosion-like lesions in the posterior margin of the L1 and L2 vertebral bodies (Figure 2). The volume of the cystic lesion was 68 × 44 × 27 mm, and the longitudinal and transverse diameters of the hernia orifice were 65 and 38 mm, respectively. Additionally, magnetic resonance imaging (MRI) of the lumbar spine revealed a lytic lesion involving the posterior part of the L1 and L2 vertebral bodies, with hyperintense signals on T2-weighted images. In addition, the spinal cord and part of the nerve roots were found to be dislocated and adhered to the dural defect in the vertebral bodies. The dorsal subarachnoid space was enlarged. MRI with gadopentetate dimeglumine (a gadolinium-based contrast agent) showed enhancement, thickening, and extension of the dura mater in the cyst. No thickening, clumping, or postcontrast enhancement of the spinal cord and nerve roots was noted (Figure 3).

Plain radiographs of the thoracolumbar spine. (a) Anteroposterior view shows bony fusions in the lumbar spine (arrows). (b) Lateral view shows squaring of the vertebral bodies (arrowheads).

Bone window computed tomography (CT) scan. (a) Axial CT image was obtained at the L1 level, displaying a lytic lesion in the dorsal vertebral body (arrow). (b) Sagittal CT image showing cystic lesions located in the posterior of the L1 and L2 vertebral bodies (arrows).
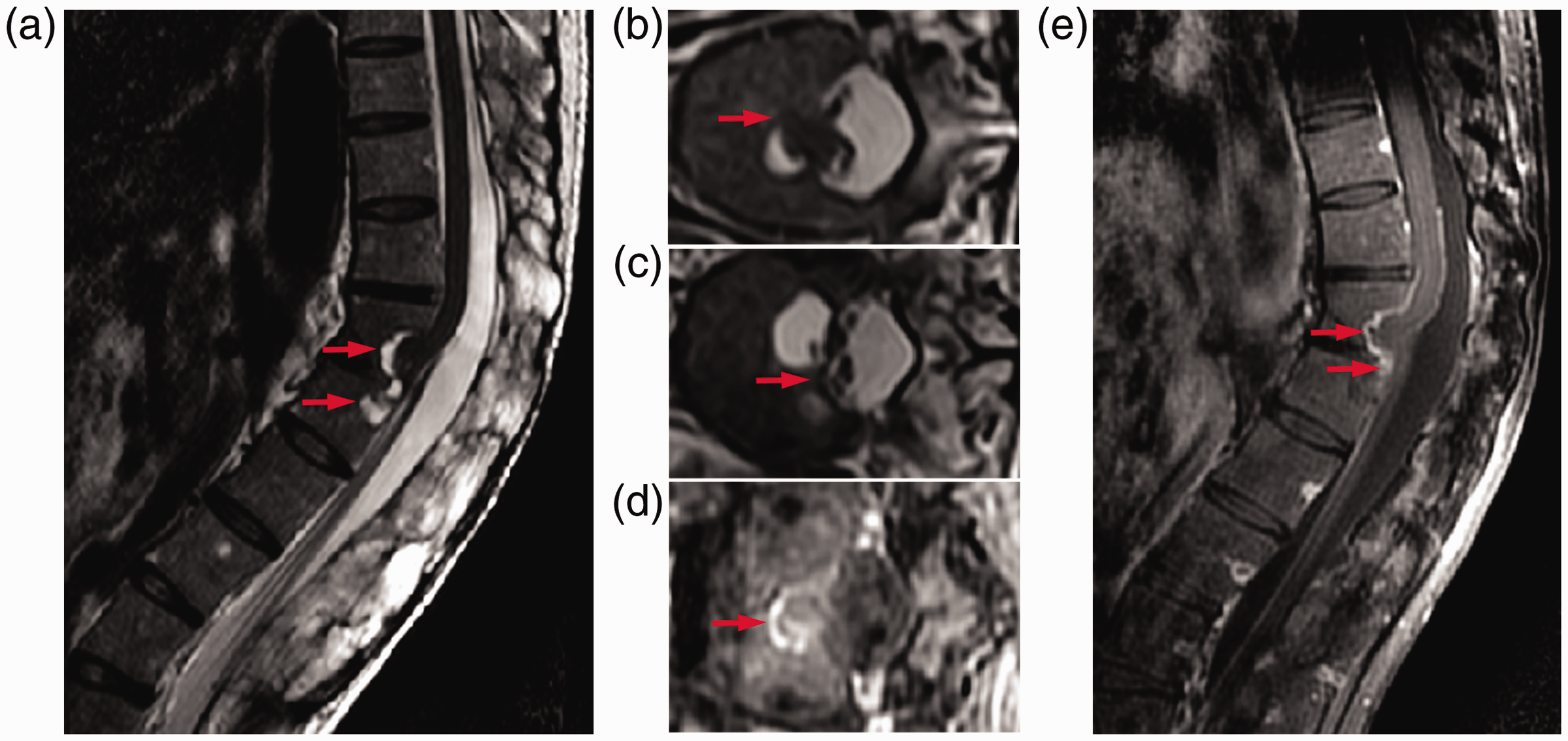
Magnetic resonance imaging (MRI) scan. Sagittal (a) and axial (b) T2-weighted MRI, exhibiting ventral displacement of the spinal cord, which was adhered to the dural ectasia (arrows). (c) Axial T2-weighted MRI at a lower level, showing dislocation of the nerve roots and attachment to the dural defect (arrow). Axial (d) and sagittal (e) T1-weighted MRI with gadopentetate dimeglumine (a gadolinium-based contrast agent), revealing dural thickening and enhancement in the cyst (arrows).
The patient was diagnosed with anterior dural ectasia–related lumbar SCH. Surgery for dural repair was proposed after consultation with an orthopedic surgeon, but the patient declined because of the potential adverse effects and financial concerns. The patient agreed to regular follow-up in the rheumatology clinic, and he was prescribed continuous oral meloxicam (7.5 mg twice per day) for AS. During the 2-year follow-up period, the motor and sensory deficits of the patient’s left leg remained the same and he showed a normal erythrocyte sedimentation rate and C-reactive protein concentration. A CT scan was performed after 2 years and demonstrated that the lytic lesion had grown to 83 × 46 × 33 mm (Figure 4). The hernia orifice had also expanded, to 72 × 39 mm. Furthermore, MRI demonstrated enlargement of the bony erosion and stable attachment of the nerve tissue to the dura mater in the cystic lesion. However, contrast-mediated enhancement was no longer observed in the lesion and the thickening of the dura mater had resolved (Figure 5).

Bone window computed tomography (CT) scan at the 2-year follow-up. (a) Axial CT scan showing the erosion-like lesion enlarged at the L1 level (arrow). (b) Sagittal CT image demonstrating expansion of the lytic lesion in the posterior aspect of the L1 and L2 vertebral bodies (arrows).

Magnetic resonance imaging (MRI) at the 2-year follow-up. Sagittal (a) and axial (b, c) T2-weighted MRI corresponding to the computed tomography scan and displaying the enlargement of the lesion and spinal cord and nerve roots remaining adhered to the dural ectasia (arrows). Axial (d) and sagittal (e) T1-weighted MRI with gadopentetate dimeglumine (a gadolinium-based contrast agent), showing resolution of the dura mater thickening and absence of gadolinium enhancement.
Discussion
Ankylosing spondylitis is associated with a variety of neurological disorders that contribute to morbidity and mortality, but data on the prevalence of neurologic involvement in patients with AS remain limited. 4 Compression of the spinal cord or nerve root is the most common cause of neurologic complications in patients with AS. 5 Although our patient also suffered from paresthesia and paresis due to SCH, MRI revealed displacement of nerve tissue in the posterior part of the L1 and L2 vertebral bodies, which is uncommon in AS patients.
Spinal syndesmophyte and ankylosis are highly prevalent in patients with long-standing AS.6,7Bony erosion tends to occur in the corner of the vertebra, but few reports have focused on the lytic lesion and enlarged dural sac in the posterior aspect of the vertebral body. Inflammation has been shown to play a critical role in the adhesion of nerve tissue and the formation of erosion-like lesions in vertebral bodies. 8 The available evidence suggests that local enthesitis and ligamentitis around the spinal canal contribute to arachnoiditis, which leads to inflammation, degeneration, fibrosis, adhesion, and tethering of nerve tissue. 9 Liu et al. 10 described fibrous tissue with scant lymphoplasma cell infiltration in the dura mater of an AS patient with dural ectasia, indicating a chronic inflammatory process. Local inflammation establishes a slowly progressive process that leads to bony erosion accompanied by compression due to expansive arachnoid diverticula. 2 Furthermore, meningeal fibrosis causes impaired resorption of cerebrospinal fluid. The fibrotic dura mater is inelastic and compromised in its response to the pressure fluctuations of cerebrospinal fluid that occur secondary to arterial pulsation and breathing; these factors induce dural sac enlargement, diverticula formation, and bony erosion. 11
Bony erosion in vertebral bodies can be caused by lumbar spine tuberculosis, especially in a geographic region with a high prevalence of
Surgical treatment of dural ectasia might improve the neurologic symptoms, but the literature regarding management of anterior dural ectasia–related lumbar SCH is scarce. 3 Bele et al. 2 reported an AS patient with anterior dural ectasia but the patient was lost to follow-up. Cornec et al. 13 reported on an AS patient with posterior dural ectasia that was unexpectedly cured by treatment with a tumor necrosis factor-α inhibitor. Our patient declined surgical intervention partly because of financial concerns, so we chose to treat the condition with an oral nonsteroidal anti-inflammatory drug, which is a first-line therapy for AS. 14 Neurological deficits in our patient remained stable, consistent with the disappearance of the dural enhancement in the lytic lesion of the vertebral bodies. Our case supports the essential role of inflammation in anterior dural ectasia formation and confirms the effectiveness of anti-inflammatory therapy. In addition, expansion of the cyst and hernia orifice led to decompression of the nerve tissue, which might have contributed, in part, to the preservation of neural function in our patient.
In conclusion, SCH through the anterior dural ectasia in a lumbar bony erosion is a rare neurological comorbidity of AS and is closely related to inflammation. Therefore, anti-inflammatory treatment is a potential alternative for patients who refuse surgical intervention.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
