Abstract
Objective:
To model the estimated disease-modifying effect of etanercept over sulphasalazine in patients with ankylosing spondylitis.
Methods:
A post hoc analysis of data from the Ankylosing Spondylitis Study Comparing ENbrel and Sulfasalazine Dosed Weekly (ASCEND) study was performed using the Natural History Staggered Start (NHSS) method. A mixed model with a linear effect over time was fitted to the ASCEND data and resampling was performed to generate confidence intervals.
Results:
At week 16, the total additional improvement in Bath Ankylosing Spondylitis Metrology Index of the etanercept arm over the sulphasalazine arm was 0.62 points, of which 31% (0.19 points) was estimated to be due to disease-modifying effect.
Conclusions:
The analysis of ASCEND data suggests that etanercept may have a larger disease-modifying effect than sulphasalazine. Further research is needed with more objective measures such as magnetic resonance imaging or X-radiography to confirm these results.
Introduction
The Bath Ankylosing Spondylitis Metrology Index (BASMI), an objective measure of spinal mobility, assesses a combination of structural damage and disease activity in patients with active ankylosing spondylitis.1,2 In the Ankylosing Spondylitis Study Comparing ENbrel and Sulfasalazine Dosed Weekly (ASCEND) study, etanercept treatment resulted in significantly better improvement in spinal mobility symptoms than sulphasalazine. 3 This may be due to a symptomatic effect, a disease-modifying effect (resulting in symptom improvement), or a combination of both. There is a strong interest in estimating the disease-modifying effect of treatment in rheumatic diseases such as ankylosing spondylitis4–6 as well as other debilitating progressive disorders.7–10
Disease-modifying effects result from effects on the underlying disease process or pathology that do not depend on continued treatment,8,9,11,12 with clinical outcomes reflecting the true disease state rather than being a temporary state due to a drug effect. This is a ‘slope’ effect, since the clinical benefit potentially accumulates as treatment continues. Treatments that have such an effect would be expected to have long-lasting (carry-over) benefits that accumulate over time, with the size of the effect depending on the duration of treatment.
Symptomatic clinical effects result from a treatment directly affecting symptoms rather than the underlying disease process or pathology.8,9,11,12 A symptomatic treatment effect improves the patient's symptoms while they are receiving treatment, but the true disease state continues to progress as if the patient were untreated. A symptomatic effect wears off after treatment is removed, such that patients who stop receiving treatment deteriorate until their symptoms reflect the true disease state.
The Natural History Staggered Start (NHSS) analysis has been proposed to differentiate between symptomatic and disease modification treatment effects.13,14 This method assumes that treatment effects that occur quickly are symptomatic effects and those that accumulate over a longer time are disease-modifying effects. NHSS analysis estimates the cross-sectional relationship between baseline score (or disease severity) and the symptomatic effect of treatment, and then estimates the slope separation that is due to patients in the study progressing to different disease severities. This slope separation is then subtracted from the total slope separation in order to calculate the natural history estimator (NHE), an estimate of the disease-modification effect.
After the initial stage and adjustment for different symptomatic effects at different disease severities, the differences between slopes in the linear model reflect differences in disease-modifying effects in treatment arms (active treatment versus active control or placebo).
In this report, we apply the NHSS method to the ASCEND clinical trial, to determine the disease-modifying effect of etanercept over sulphasalazine in patients with ankylosing spondylitis.
Patients and methods
ASCEND
ASCEND was a randomised double-blind study to evaluate the safety and efficacy of etanercept and sulphasalazine in subjects with ankylosing spondylitis (ClinicalTrials.gov identifier: NCT00247962).
3
Details of the study design and protocol have been reported previously.
3
In brief, subjects were randomly assigned to the study treatment at the allocation rate of 2:1 for either 50 mg etanercept once weekly or 3 g sulphasalazine daily. A total of 566 subjects were enrolled in the study (
NHSS analysis
Based on knowledge of the pharmacokinetics and pharmacodynamics of etanercept, its symptomatic effect was assumed to be fully evident by 2 weeks post baseline. Thus, the model was fitted for the 2–16 week period. As reference, and to test the model assumptions, the results were estimated with extended initial periods by performing separate analyses for weeks 4–16, weeks 8–16, and weeks 12–16.
A mixed-model analysis on the change of BASMI, with time of visit as a continuous covariate in the model, was run using PROC MIXED in SAS® version 9.2. 15 A compound symmetry covariance structure was used. Scheduled time of visit (categorical variable) was included in the repeated statement for modeling the covariance structure. A linear effect over time was included in the model. NHE was calculated based on parameter estimates from this model.
Terms correspond to the model shown below:



NHE can be interpreted as the number of points of treatment-related benefit (relative to control) due to disease modification based on the linear model. The standard error of this estimate was obtained by using a bootstrap procedure: re-sampling with replacement from the original dataset and calculating the standard error of the distribution of the NHE from this re-sampling procedure. Then, 90% confidence intervals for the NHE were obtained using the 5th and 95th percentiles of the bootstrap distribution of the NHE. Observing whether 0 is contained in the confidence interval is equivalent to performing a two-sided test using a 10% significance level. The percentage of the treatment effect that is due to disease modification was calculated as the ratio of the estimated disease-modifying effect at the end of the study to the total treatment effect at the end of the study.
The predicted means over time were calculated using the linear model from SAS® PROC MIXED. The estimated disease-modifying effect decline over time is assumed to have the same intercept as the control arm, since any early dramatic effects are defined as symptomatic. Observed means over time were also calculated based on grouped time values. If 0 was not covered by the 90% confidence interval, the parameter was deemed significantly different from 0 at one-sided α = 0.05 or two-sided α = 0.10.
Results
The NHSS analysis revealed that a proportion of the total treatment effect of etanercept over sulphasalazine was due to a positive disease-modifying effect. This additional treatment effect was 0.62 BASMI points at week 16 (90% CI 0.044, 0.342), of which 0.19 points (31%) represented a disease-modifying effect. The disease-modifying effect was significant at the 90% confidence level.
Figure 1 shows the disease-modifying effect of the study drugs in terms of change in BASMI score. The piece-wise linear curves are the means of the change of BASMI at each time point. The two fitted straight lines display the fitted BASMI change by the model, and the NHE line shows the disease-modifying effect of the sulphasalazine arm plus the incremental disease-modifying effect of etanercept. The adjustment to the slope estimates was minimal, indicating that the slopes over time are a good approximation to the disease-modifying effect for etanercept over sulphasalazine.
Mean change in Bath Ankylosing Spondylitis Metrology Index (BASMI) of etanercept versus sulphasalazine in patients with ankylosing spondylitis included in the ASCEND trial.
3
etn, etanercept; obs, observed data; nhe, natural history estimator; ssz, sulphasalazine.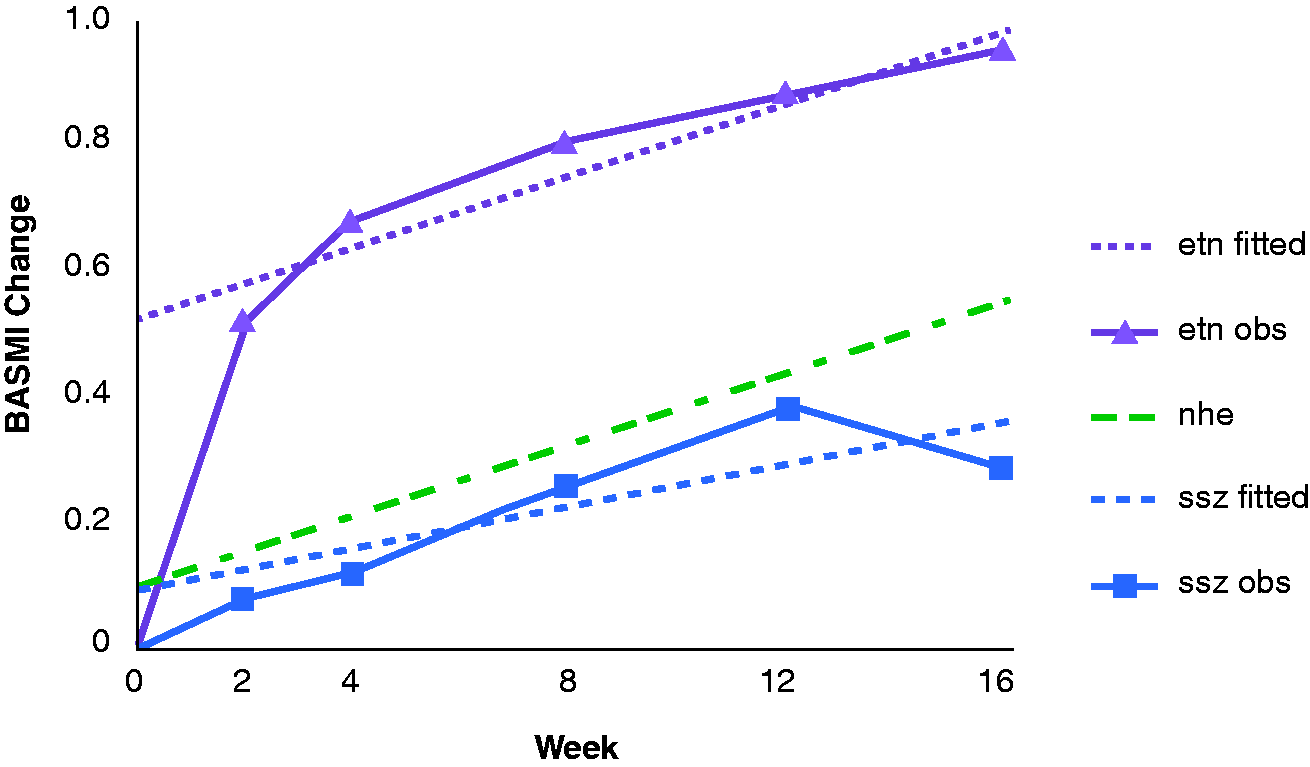
Difference in disease-modifying effect (change in Bath Ankylosing Spondylitis Metrology Index) of etanercept versus sulphasalazine in patients with ankylosing spondylitis included in the ASCEND trial, 3 as determined by Natural History Staggered Start analysis of different modeling periods.

Obtained by a bootstrap method with 1000 simulations.
The total 16-week difference differs slightly owing to differences in data modeling.
NS, not statistically significant (
Discussion
The present analysis found a significant positive disease-modifying effect of etanercept over sulphasalazine in the treatment of ankylosing spondylitis. As the half-life of etanercept is about 70 h 16 and that of sulphasalazine is 5–10 h, 17 it is reasonable to expect them to reach steady state and full pharmacodynamic effect within 2 weeks. Thus, their symptomatic effects were likely to be fully established by week 2, and any further improvements in symptoms were therefore mostly due to a disease-modifying effect.
In the ASCEND clinical trial, the efficacy of etanercept was compared against an active control (sulphasalazine) rather than a placebo control. 3 Thus, the calculated NHE is the difference in disease-modifying effect of etanercept versus sulphasalazine. As the active control may also have a disease-modifying effect, the difference between arms is more difficult to detect and less likely to be significant. Nonetheless, there were significant positive differences when modeling the week 2–16 period. The magnitude of the disease-modifying effect based on the 2–16 week time interval was consistent with effects based on 4–16 weeks and 8–16 weeks. The 12–16 week model was based on the period of decline in the sulphasalazine group, and may reflect longer term disease modification estimates if this pattern were to continue beyond 16 weeks.
The NHSS method relies on the assumption that symptomatic effects are fully established prior to collection of data included in the model. Since the current results were similar when based on data from 12–16 weeks and from 2–16 weeks, this assumption seems reasonable. The additional assumption (that the cross-sectional relationship between baseline score and symptomatic effect is the same as the longitudinal relationship between true score [without symptomatic effects] and symptomatic effect) is untestable however, rendering the current findings not necessarily conclusive. Further research with more objective measures such as magnetic resonance imaging or X-radiography will be needed to confirm these results. The NHSS method provides a straightforward approach to estimate disease-modifying effects when comparing an active treatment and a placebo or active control.
Footnotes
Declaration of conflicting interest
Suzanne Hendrix is an employee of Pentara Corporation, who were paid consultants to Pfizer Inc. in connection with the development of this manuscript. Andrew Koenig and Amitabh Singh are employees of and own stock in Pfizer Inc. Wenzhi Li is an employee of Quintiles, who were paid contractors to Pfizer Inc., providing statistical and writing support for the development of this manuscript.
Funding
The ASCEND study was funded by Wyeth, which was acquired by Pfizer Inc. in October 2009. Editorial support was provided by Donna McGuire and Patricia McChesney of UBC Scientific Solutions, and was funded by Pfizer Inc.
