Abstract
Objective
This study was performed to determine the healing effects of pentoxifylline on molecular responses and protection against severe ischemic damage in the small intestine.
Methods
Thirty-six Wistar albino rats were divided into six groups. The superior mesenteric artery was clamped for 120 minutes, and reperfusion was performed for 60 minutes. Saline (0.4 mL), pentoxifylline (1 mg/kg), and pentoxifylline (10 mg/kg) were intraperitoneally administered to the rats in the C1, P1, and P3 groups, respectively, 60 minutes before ischemia and to the rats in the C2, P2, and P4 groups, respectively, during reperfusion onset. Malondialdehyde, myeloperoxidase, tumor necrosis factor alpha, interleukin-1 beta, and interleukin-6 in serum and tissue were measured by enzyme-linked immunosorbent assay. Intestinal ischemic injury was histopathologically evaluated by the Chiu score and immunohistochemical staining.
Results
All serum and tissue molecular responses were significantly blunted in the pentoxifylline-treated groups compared with the controls. Significant improvement in ischemic damage was demonstrated in the pentoxifylline-treated groups by histological grading and immunohistochemical scoring.
Conclusions
The protective effects of pentoxifylline were confirmed by molecular responses and histopathological examination.
Keywords
Introduction
Ischemia–reperfusion (I/R) is a pathological process that gives rise to severe ischemic damage in the small intestine. Restoration of blood flow to ischemic tissue may prevent tissue necrosis and facilitate restoration of organ function.1,2 Several molecular markers associated with I/R have been identified.3–5 Pentoxifylline (PTF), a methylxanthine derivative, has a hemorheological effect that increases the elasticity of red blood cells and reduces blood viscosity. Decreased blood viscosity results in increased blood flow to the microcirculation and increased tissue oxygenation. The increased elasticity of the red corpuscles causes deformities in these structures. PTF has been used to treat intermittent claudication disorders in Europe since 1972. 6 PTF has also been shown to induce vasodilatation and inhibit neutrophil adhesion; these effects have stimulated interest in PTF as a potentially useful compound for treating various I/R-related conditions. In 1995, PTF was used for the first time as an anti-ischemic drug in a small bowel ischemia model in rats. 7 The discovery that PTF had an anti-tumor necrosis factor α (TNFα) effect resulted in its use in the treatment of organ ischemia. 8 PTF primarily inhibits phosphodiesterase enzymes, which increase the amount of cyclic adenosine monophosphate in polymorphonuclear leukocytes while decreasing free oxygen radical production. 9 Recent studies have shown that PTF inhibits xanthine oxidase, thus reducing superoxide and hydroxyl radicals and restoring capillary circulation and tissue oxygenation. Because PTF inhibits free oxygen radicals and phospholipase A2, prostacyclin release is increased. 10
In this study, we aimed to determine the influence of PTF on tissue healing by examining the molecular responses to and protective effects of PTF in a rat model of severe intestinal I/R. The role of PTF in severe small bowel ischemia was investigated by evaluating the molecular and pathological parameters in this experimental context. The timing and dose of PTF administration were studied to determine the effect of this molecule under different conditions. Our specific hypothesis was that PTF has a protective effect on small intestinal damage after I/R.
Methods
This study was approved by the Animal Experiments Local Ethics Committee of Istanbul University (process number 2015/80). The experiments were performed in adherence with the international guidelines for the care and use of laboratory animals at the Laboratory of Surgical Physiopathology.
Thirty-six male Wistar rats (250–350 g) were purchased from the Institute of Experimental Medicine at Istanbul University. Male rats were used because the menstrual cycle may affect blood test results. All rats were housed in metal cages and maintained in a 12-hour dark/light cycle at a controlled temperature of 22°C (±1°C). All rats were given food containing 21% protein and allowed free access to fresh tap water. All cages were cleaned daily. Each cage housed four rats, and rats that were in the same experimental group were housed together.
The rats were randomly assigned to one of six study groups. They were fasted during the night before the surgery. On the day of surgery, the rats were anesthetized with ketamine hydrochloride (50 mg/mL) and xylazine hydrochloride (20 mg/mL) administered by intraperitoneal (IP) injection at a dose of 0.1 mL per 100 g. The rats were then placed in the supine position on a regularly disinfected and surgically draped operating table. Anesthesia and skin sterilization were performed under laboratory conditions for all animals. The animals were not mechanically ventilated. Next, a 4-cm median laparotomy was performed. The small intestine was examined, and the superior mesenteric artery (SMA) was identified and dissected. The SMA was then carefully isolated and clamped while keeping the superior mesenteric vein intact. The small intestine was repositioned into the abdominal cavity. The SMA was clamped for 120 minutes, and reperfusion was initiated by removing the clamp at the end of 120 minutes. Reperfusion was performed for 1 hour before collecting blood to measure the following inflammatory markers: lactic acid dehydrogenase (LDH), which is an indicator of small intestinal damage; tissue malondialdehyde (MDA), which is an indicator of lipid peroxidation and, later, of ischemic damage (it is also indicative of oxidative stress, which can lead to cellular damage after a shift to the pro-oxidant side of the pro-oxidant–anti-oxidant balance); and myeloperoxidase (MPO), which is a quantitative indicator of the presence of neutrophils in the small intestine (it is found in the granules of mammalian neutrophils and plays an important role in the killing abilities of phagocytes). Small bowel specimens were also obtained during blood collection for histological studies.
The rats were divided into the following six groups:
C1 control group (n = 6): 0.4 mL of normal saline (NS) was administered by IP injection 60 minutes before intestinal ischemia.
C2 control group (n = 6): 0.4 mL of NS was administered by IP injection during reperfusion onset.
P1 treatment group (n = 6): 0.4 mL of NS and 1 mg/kg of PTF were administered by IP injection 60 minutes before intestinal ischemia.
P2 treatment group (n = 6): 0.4 mL of NS and 1 mg/kg of PTF were administered by IP injection during reperfusion onset.
P3 treatment group (n = 6): 0.4 mL of NS and 10 mg/kg of PTF were administered by IP injection 60 minutes before intestinal ischemia.
P4 treatment group (n = 6): 0.4 mL of NS and 10 mg/kg of PTF were administered by IP injection during reperfusion onset.
Next, 6 mL of blood from each rat’s heart cavity was collected to quantify the levels of serum LDH, MDA, MPO, TNFα, interleukin-1 beta (IL-1β), and interleukin-6 (IL-6). The inflammatory cytokines TNFα, IL-1β, and IL-6 are indicative of an acute inflammatory response to bacteria and other infectious agents. Small intestine specimens were obtained for histological studies and to determine the concentrations of TNFα, IL-1β, IL-6, MDA, and MPO in wet tissue. Blood and tissue samples were taken at the end of reperfusion in all groups. The rats were killed immediately after collection of the blood and tissue samples. Tissue samples were fixed in 10% neutral formaldehyde.
All tissues were washed in phosphate-buffered saline (PBS) at 4°C, placed in a cryogenic tube, and stored at −80°C until analysis. Blood samples were taken from the heart apex while the heart was beating. Samples were then centrifuged at 3,000 to 35,000 rpm for 15 minutes. All serum samples obtained from each animal were placed into three separate Eppendorf tubes and stored at −80°C until analysis.
Preparation of tissue homogenates
Tissue samples were homogenized in PBS and formed a 10% (w/v) homogenate. Homogenization was performed with a tissue grinder fitted with a Teflon pestle at a speed of 1000 rpm for 10 minutes.
Serum and intestinal tissue tests
The serum LDH level was determined using a cobas 8000 e602 modular automatic analyzer (Roche Diagnostics, Mannheim, Germany). Serum and intestinal tissue levels of TNFα were measured using enzyme-linked immunosorbent assay (ELISA) kits (bms622, bms622TWO, and bms622TEN; Affymetrix eBioscience, San Diego, CA, USA). Serum and tissue levels of IL-6 were determined using ELISA kits (bms625, bms625TWO, and bms625TEN; Affymetrix eBioscience). Serum and tissue levels of IL-1β were determined using ELISA kits (bms630 and bms630TEN; Affymetrix eBioscience). Serum and tissue levels of MPO were determined using ELISA kits (201-11-0575; Shanghai Sunred Biological Technology Co., Ltd., Shanghai, China). Serum and tissue levels of MDA were determined using ELISA kits (201-11-0157; Shanghai Sunred Biological Technology Co., Ltd.).
Histopathological analyses
Cross-sections (4-µm-thick) of small bowel tissue were stained with hematoxylin and eosin after paraffin embedding. The samples were examined and photographed under a light microscope. Histopathological changes were evaluated in a double-blind manner (blinded assessment of outcomes by two independent assessors). Each section of ischemic intestinal tissue was scored on a 6-point scale as described by Chiu et al. 11
Immunohistochemical studies
Intestinal tissues were dissected, fixed in 10% neutral buffered formalin, embedded in paraffin wax, and then cut into 4-µm-thick sections. The sections were placed onto adhesive slides, deparaffinized in xylene, and rehydrated in a graded alcohol series. Immunoperoxidase staining was performed with an UltraVision Large Volume Detection System: Anti-Polyvalent, HRP (Cat. No. TP-125-HL; Thermo Fisher Scientific, Waltham, MA, USA). Endogenous peroxidase activity was inactivated by incubation in 3% hydrogen peroxide for 10 minutes. The sections were incubated with blocking solution for 5 minutes at room temperature. Next, they were incubated with anti-IL-6 antibody (dilution 1:100; BIOZOL Diagnostica Vertrieb GmbH, Eching, Germany), anti-TNFα antibody (dilution 1:100; BIOZOL), anti-MPO antibody (dilution 1:100; BIOZOL), and anti-IL-1β antibody (dilution 1:100; BIOZOL) overnight at 4°C and washed with PBS the next day. Antibody detection was performed using a biotinylated universal secondary antibody, horseradish peroxidase streptavidin-complex, and amino-ethyl-carbazole as the chromogen. The sections were then counterstained with Mayer’s hematoxylin. Normal rabbit IgG (Cat. No. sc-2027; Santa Cruz Biotechnology, Dallas, TX, USA) was used for the negative control.
Histological score analysis
A Leica DM2500 light microscope was used for histological score analysis, and the samples were photographed with a Leica DFC280 digital camera system (Leica Microsystems, Wetzlar, Germany). The total tissue area of each slide was examined under 400× magnification by two researchers in a blinded manner. The intensity of immunostaining was semiquantitatively evaluated using the following scores: 0 (no staining), 1+ (weak but detectable staining), 2+ (moderate staining), 3+ (distinct staining), and 4+ (intense staining).
Statistical analyses
The Kolmogorov–Smirnov test was used for normally distributed data. The K-sample Kruskal–Wallis test, chi-square test, and analysis of variance were performed to compare the results. Differences were considered statistically significant at a P value of < 0.05. Statistical analyses were performed using SPSS software, version 23.0.1 (IBM Corp., Armonk, NY, USA).
Results
Biochemical results of blood and small bowel tissue analysis
The TNFα, IL-6, IL-1β, MPO, MDA, and LDH levels measured in blood are shown in Table 1. The TNFα, IL-6, IL-1β, MPO, and MDA levels measured in small bowel tissue are shown in Table 2. Significant differences in serum and tissue molecular responses were found between the P1 and C1 groups and between the P3 and C1 groups (both the P1 and P3 groups were treated with PTF 1 hour prior to ischemia) (P < 0.05 for all) (Table 3). Similarly, significant differences in serum and tissue molecular responses were observed between the P2 and C2 groups and between the P4 and C2 groups (both the P2 and P4 groups were treated with PTF at the beginning of reperfusion) (P < 0.05 for all) (Table 3). These significant differences were the result of elevated molecular responses in the control groups. However, some significant differences were also found between the groups treated with PTF. The blood IL-6 levels were significantly different between the P1 and P3 groups (P = 0.002) and between the P2 and P4 groups (P = 0.009) (Figure 1). Furthermore, the blood IL-1β levels were significantly different between the P1 and P3 groups (P = 0.002) and between the P2 and P4 groups (P = 0.002) (Figure 2). The blood MDA and LDH levels were significantly different between the P1 and P3 groups (P = 0.002 for both) (Figures 3 and 4). The tissue IL-6 and MDA levels were significantly different between the P2 and P4 groups (P = 0.004 and P = 0.41, respectively) (Figures 5 and 6).
TNF-α, IL-6, IL-1β, MPO, MDA, and LDH values in blood

TNF-α, tumor necrosis factor alpha; IL-6, interleukin-6; IL-1β, interleukin-1 beta; MPO, myeloperoxidase; MDA, malondialdehyde; LDH, lactic acid dehydrogenase; SD, standard deviation.
TNF-α, IL-6, IL-1β, MPO, and MDA values in small bowel tissue

TNF-α, tumor necrosis factor alpha; IL-6, interleukin-6; IL-1β, interleukin-1 beta; MPO, myeloperoxidase; MDA, malondialdehyde; SD, standard deviation.
Significant improvement in molecular markers between control and pentoxifylline treatment groups

TNF-α, tumor necrosis factor alpha; IL-6, interleukin-6; IL-1β, interleukin-1 beta; MPO, myeloperoxidase; MDA, malondialdehyde; LDH, lactic acid dehydrogenase.
aKruskal–Wallis test, bMann–Whitney U test.

Mean IL-6 (pg/mL) levels in blood (statistically significant differences between the groups are given in Table 3). IL-6, interleukin-6.

Mean IL-1β (pg/mL) levels in blood (statistically significant differences between the groups are given in Table 3). IL-1β, interleukin-1 beta.

Mean MDA (nmol/mL) levels in blood (statistically significant differences between the groups are given in Table 3). MDA, malondialdehyde.

Mean LDH (IU/L) levels in blood (statistically significant differences between the groups are given in Table 3). LDH, lactic acid dehydrogenase.

Mean IL-6 (pg/g wet tissue) levels in wet tissue (statistically significant differences between the groups are given in Table 3). IL-6, interleukin-6.

Mean MDA (nmol/g wet tissue) levels in wet tissue (statistically significant differences between the groups are given in Table 3). MDA, malondialdehyde.
Histopathological results
Ischemic damage was significantly reduced in the groups treated with PTF as demonstrated by histological staging. Statistically significant differences were found between the C1 group and the P1–P3 groups as well as between the C2 group and the P2–P4 groups (P = 0.004 and P = 0.002, respectively). There were significant differences between the P1 and C1 groups and between the P3 and C1 groups (P = 0.002 and P = 0.026, respectively), but not between the P1 and P3 groups. Similarly, there were statistically significant differences between the P2 and C2 groups and between the P4 and C2 groups (P = 0.002 for both), but not between the P2 and P4 groups. The results of the histological staging of I/R injury according to Chiu et al. 11 are presented in detail in Table 6.
Immunohistochemistry results
Immunohistochemical scores in all groups
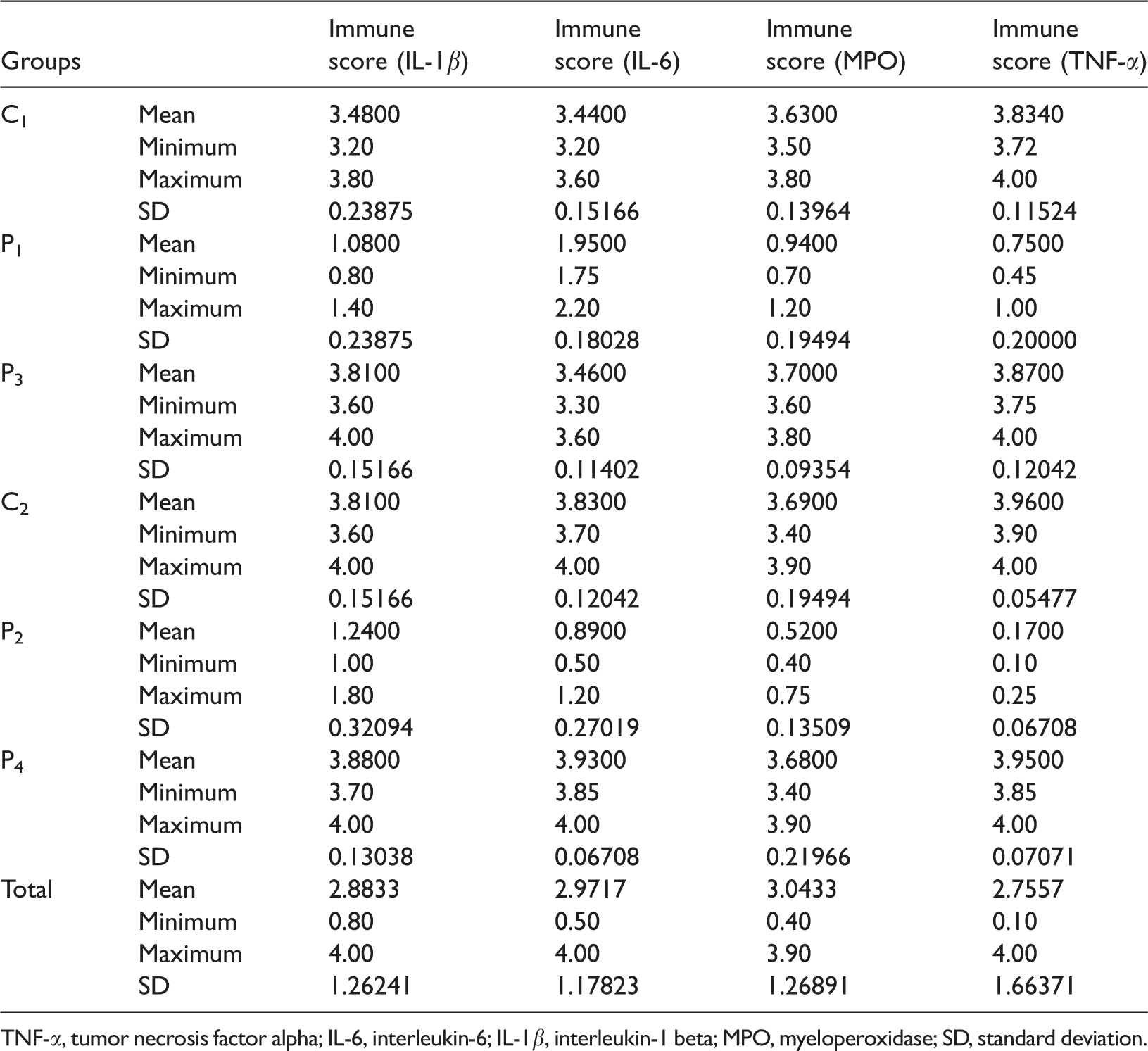
TNF-α, tumor necrosis factor alpha; IL-6, interleukin-6; IL-1β, interleukin-1 beta; MPO, myeloperoxidase; SD, standard deviation.
A, B Immunohistochemical score of IL-1β, IL-6, MPO, and TNF-α expression in control and pentoxifylline treatment groups

Values are presented as mean ± standard deviation; All of italic entries emphasize that the value of P is statistically significant. aP = 0.008 versus C2 and P4 groups. TNF-α, tumor necrosis factor alpha; IL-6, interleukin-6; IL-1β, interleukin-1 beta; MPO, myeloperoxidase.
Results of histological staging of ischemia–reperfusion injury according to Chiu et al. 11

A high degree of damage was detected in the immunohistochemical images of samples from the C1 group (Figure 7). The number of goblet cells and amount of secretion they produced were substantially increased. The morphology of the goblet cells had deteriorated, and the expression of cell type-specific markers was elevated, especially in the lamina propria. Cell damage was more intense in the C2 than C1 group (Figure 8). Severe breakage and deletions were observed in the intestinal villi. Increased IL-1β expression was observed between damaged cells. Abnormal morphology and expression in the lamina propria were also increased. Tissue samples from the P1 and P2 groups were preserved without deterioration of the tissue integrity and with minimal immunoreactivity (Figures 7 and 8). The expression intensity was fairly low. Images showing normal small intestinal morphology were obtained. Although expression of goblet cell markers was somewhat evident in the P3 and P4 groups, tissue damage was prevented in a similar manner in the P3 and P2 groups (Figure 8). However, partial tissue damage was sustained in the P4 group. The morphology of the lamina propria was impaired, and the expression intensity from the lamina propria cells was increased.

Immunostaining detection images for the C1, P1, and P3 groups. Decreased immunopositivity of IL-1β, IL-6, MPO, and TNF-α expression was observed in both the P3 and C1 groups but was lower in the P3 group. Although the immunopositivity was decreased in the P3 group, the damage continued at the tissue level. Streptavidin–biotin peroxidase method. Counterstain: Mayer’s hematoxylin. Magnification is ×10 for all photographs. IL-1β, interleukin-1 beta; IL-6, interleukin-6; MPO, myeloperoxidase; TNF-α, tumor necrosis factor alpha.

Immunostaining detection images for the C2, P2, and P4 groups. Decreased immunopositivity of IL-1β, IL-6, MPO, and TNF-α expression was observed in both the P4 and C2 groups but was lower in the P4 group. Although the immunopositivity was decreased in the P4 group, the damage continued at the tissue level. Streptavidin–biotin peroxidase method. Counterstain: Mayer’s hematoxylin. Magnification is ×10 for all photographs. IL-1β, interleukin-1 beta; IL-6, interleukin-6; MPO, myeloperoxidase; TNF-α, tumor necrosis factor alpha.
Discussion
Recent studies have shown that PTF reduces superoxide and hydroxyl radicals by inhibiting xanthine oxidase in patients in the clinical setting; PTF can result in increased tissue oxygenation and improved capillary filling in cases of strangulation by small bowel closed loop obstruction, ischemic colitis, or intestinal I/R injury.12–14 In the present study, our aim was to determine the effects of pentoxifylline on free oxygen radicals and oxidative damage in an I/R model in rats. Our study had three components. First, we collected blood samples to quantify the serum levels of LDH, MDA, MPO, TNFα, IL-1β, and IL-6. We also collected tissue samples from the small bowel to quantify the tissue levels of MDA, MPO, TNFα, IL-1β, and IL-6. Second, we performed histopathological injury scoring for the small bowel. Third, we performed immunohistochemical injury scoring and acquired immunohistochemical images of the small bowel.
A similar experimental study performed by Lloris-Carsí et al. 15 in 2013 suggested that PTF protected the small intestine after severe I/R. TNFα, IL-1β, IL-6, MDA, and MPO in both blood and intestinal tissue were investigated in our study, but TNFα, IL-1β, and IL-6 in blood and MDA and MPO in intestinal tissue were investigated in the study by Lloris-Carsí et al. 15 Moreover, we added immunohistochemical analyses to the present study to enrich the results of previous studies.
Prior to 2013, the PTF dose range applied in intestinal I/R rat models was approximately 20 to 300 mg/kg body weight. However, in the study performed by Lloris-Carsí et al., 15 PTF doses of 1 and 10 mg/kg were administered. PTF resulted in significant protection against severe ischemic small intestinal damage, even when used during reperfusion onset and at levels not previously reported (10 mg/kg). These effects were evidenced by an improved biochemical and histologic profile. Lower doses of PTF (1 mg/kg) also demonstrated a significant protective effect against inflammation and tissue markers of I/R damage. 15 Because PTF doses of 1 and 10 mg/kg had a significant protective effect on severe ischemic small intestinal damage as well as on inflammation and tissue markers of I/R damage, we also used low doses in our study.
Both tissue and blood levels of TNFα, IL-6, and IL-1β (which are indicative of inflammation) improved in all groups treated with PTF before ischemia and at the onset of reperfusion. These results suggest that PTF protects against inflammatory processes in the small bowel. Both tissue and blood levels of MPO, which is a marker of oxidative stress, improved in all groups treated with PTF before ischemia and at the onset of reperfusion. MDA is an indicator of lipid peroxidation levels, and MDA levels are always elevated following tissue damage. 16 In an isolated canine gracilis muscle model of I/R, PTF inhibited platelet-activating factor and thus had protective effects. 17 In the present study, both the tissue and blood levels of MDA improved in all groups treated with PTF before ischemia and at the onset of reperfusion. The blood levels of LDH, which is indicative of cell damage, improved in all groups treated with PTF before ischemia and at the onset of reperfusion. Cumulatively, these results indicate that PTF protects against cell damage.
The histopathological injury scores for the small bowel in all groups treated with PTF were significantly lower than those in the control group. These results suggest that PTF has a significant role in the prevention of oxidative stress, inflammatory processes, and cell damage in the small bowel.
When we examined the immunohistochemical staining results, we found that tissue integrity with low immunoreactivity was preserved in the P1 and P2 groups (treated with 1 mg/kg of PTF before ischemia and at the beginning of reperfusion). The immunohistochemical images were similar to the normal small intestinal morphology, indicating that tissue damage was prevented in these groups. We found that tissue damage was partly prevented in the P3 group (treated with 10 mg/kg of PTF before ischemia). However, tissue damage was more extensive in the P4 group (treated with 10 mg/kg of PTF at the onset of reperfusion), suggesting that a high dose of PTF was not protective against tissue damage. A PTF dose of 10 mg/kg during reperfusion onset produced worse outcomes than a dose of 1 mg/kg. Thus, 10 mg/kg may not provide an adequate blood current or tissue oxygenation in the microcirculation of the small intestine compared with 1 mg/kg. We found that tissue damage was more intense in the C2 than C1 group, although the same intensity of tissue damage was expected in both control groups. In the C1 group, 0.4 mL of NS administered by IP injection 3 hours before the same treatment in the C2 group may have induced less tissue damage because of the increased microvascular current in the small intestinal wall. Given our findings of the differences between the P2 and P4 groups and between the C1 and C2 groups, we believe that larger sample sizes are necessary to draw a clear conclusion.
This is the first study to use immunohistochemical analyses in a rat model of intestinal I/R for evaluation of the systemic and local expression of inflammatory cytokines and oxidative stress markers of ischemic small bowel injury.7,15,18–22 Few studies reported in the literature have addressed the role of PTX in small intestinal I/R.7,15,18–23 The present study on the mechanisms underlying the protective effect of PTF in small intestinal ischemia specifically suggests that the anti-inflammatory action of PTF is very important. This study indicates that PTX reduces the levels of TNFα, IL-1β, and IL-6 after severe organ ischemia in rats, which may have important implications for ischemia outcomes because these three proinflammatory cytokines are regulators of the inflammatory response in human and animal ischemic cells. Our findings are similar to those of other studies that investigated the effects of PTF on ischemic small bowel injury.7,15,18–22
The present findings regarding tissue MPO as a marker of neutrophil infiltration and organ responses to PTF are similar to the findings from the previous study performed by Lloris-Carsí et al. 15 However, we also examined blood MPO levels and found statistically significant differences. Our findings regarding other markers that were examined to evaluate ischemic damage, such as MDA and LDH, are also similar to the findings from their study. 15 Furthermore, we examined MDA in blood and found statistically significant differences.
The main limitation of our research approach was the small number of animals in each group (n = 6), which was suggested by the ethics committee. We believe that as the sample size increases, the statistics will prove to be more significant.
In conclusion, our biochemical, histopathological, and immunohistochemical findings suggest that a low dose of PTF may effectively improve ischemic damage in experimental I/R in rats. We plan to confirm the findings of this study using larger sample sizes in the future.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
