Abstract
Objective
Carcinosarcoma consists of carcinomatous and sarcomatous tissues and is an aggressive malignant tumor. It is rarely reported in the hypopharynx.
Methods
A 72-year-old man presented with dysphagia and dyspnea. Laryngoscopy, computed tomography (CT), and 18F-fluorodeoxyglucose positron emission tomography/CT (18F-FDG PET/CT) showed a neoplasm on the left posterior hypopharyngeal wall. The patient underwent bilateral neck dissection and excision of the hypopharyngeal cancer followed by postoperative radiation therapy.
Results
Immunohistochemistry revealed carcinomatous cells with membrane positivity for cytokeratin, glucose transporter-1 (GLUT-1), phosphoinositide-3 kinase (PI3K), hypoxia-inducible factor-1α (HIF-1α), and hexokinase-II as well as sarcomatous cells with membrane positivity for smooth muscle actin, GLUT-1, HIF-1α, and PI3K. Histopathology and immunohistochemistry revealed a true carcinosarcoma of the hypopharynx (pT3N0M0, Stage III).
Conclusions
Thorough immunohistochemistry is required for a correct diagnosis of hypopharyngeal carcinosarcoma. 18F-FDG PET/CT may help to distinguish hypopharyngeal carcinosarcoma from benign tumors.
Keywords
Introduction
Carcinosarcoma consists of carcinomatous and sarcomatous tissues and is an aggressive malignant tumor. Carcinosarcoma has been reported in the respiratory tract, gastrointestinal tract, urogenital tract, and other sites. Although various reports have described spindle cell carcinoma in the head and neck,1–9 carcinosarcoma is rare in the larynx and hypopharynx. Of all malignant neoplasms, carcinosarcomas originating in the larynx or hypopharynx account for <1%. 10 Furthermore, this cancer is more rarely found in the hypopharynx (4.6%) than in the larynx (46.4%). 6 A recent report stated that the oral cavity (63.0%) is the most common site. Carcinosarcoma in the larynx (17.5%) and oropharynx/hypopharynx (11.7%) is relatively rare. 11 Only 16 cases originating in the hypopharynx have been described in the literature. Etiologic factors of this cancer include advanced age in men, a history of alcohol consumption, smoking, and a history of irradiation.
The histogenesis of carcinosarcoma remains unclear. Two antithetical hypotheses, the multiclonal hypothesis and the monoclonal hypothesis, have been proposed to explain the histogenesis of carcinosarcoma. The latter one is generally accepted. The monoclonal hypothesis states that the carcinosarcoma first originates from single totipotential stem cells, then differentiates in mesenchymal and epithelial directions. 12 However, the true histogenesis of carcinosarcoma is unknown. For a correct diagnosis, both histology and immunohistochemistry must show a malignant mesenchymal component (sarcomatous component) and an epithelial component (carcinomatous component) in both the primary and secondary sites.
Because hypopharyngeal carcinosarcoma is an extremely rare malignant tumor, all descriptions in the literature are presented as case reports. The prognosis of hypopharyngeal carcinosarcoma may vary according to the initial stage at presentation, tumor size, tumor invasion depth, metastasis of lymph nodes or distant areas, type of surgery, whether the patient undergoes radiotherapy or chemotherapy, and complications.1,13–15 Surgery is the proper treatment for hypopharyngeal carcinosarcoma. A reasonable therapeutic approach includes complete surgical resection and suitable neck dissection. Patients should undergo radiotherapy when a locally advanced, postoperative residual tumor is present or when the tumor has no clear safe surgical margins and exhibits extracapsular spread and vascular/perineural invasion. Furthermore, long-term follow-up is essential to identify recurrence or metastasis.1,13,14 However, some studies have not confirmed the therapeutic efficacy of radiotherapy on survival.2,3 Moreover, the impact of chemotherapy is unclear. 16 Thus, new treatments are required to improve long-term survival. Targeted therapies have recently been used to treat other cancers,17–19 but no such therapies have been developed for patients with hypopharyngeal carcinosarcoma. TU-ECS-1, a new human uterine carcinosarcoma cell line, was recently established to exhibit mutations of the tumor protein p53 gene (TP53) and KRAS 20 and represents a novel targeted therapy. In one study, overexpression of epidermal growth factor receptor (EGFR) and vascular endothelial growth factor C was found in patient-derived xenograft models of ovarian carcinosarcoma. Pathogenic mutations were also detected in TP53 and PIK3CA. However, treatment with a vascular endothelial growth factor (VEGF) inhibitor (BMS-690514) combined with carboplatin/paclitaxel did not inhibit tumor growth or improve animal survival. 21 Thus, better predictive markers of treatment response are needed.
The Warburg effect (aerobic glycolysis) is a phenomenon in which cancer cells consume a large amount of glucose and undergo aerobic glycolysis, although the oxygen is sufficient. Reprogramming the Warburg effect in a tumor by targeting its mitochondria improves the radiosensitivity of the tumor. 22 Several hypoxic markers are involved in the Warburg effect, including phosphatidylinositol-3 kinase (PI3K), hypoxia-inducible factor-1α (HIF-1α), hexokinase-II (HK-II), and glucose transporter-1 (GLUT-1). Thus, we propose that targeting hypoxic markers may be useful for treating hypopharyngeal carcinosarcoma.
18F-Fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET/CT) is performed for whole-body examination of glucose metabolism and can show the distribution of tumor cells of various malignancies. This imaging technique has been used to provide prognostic information regarding prostate, primary liver, and uterine carcinosarcomas in clinical practice.23–25 Because hypopharyngeal carcinosarcoma is rare, only one report to date has described the use of 18F-FDG PET/CT for this type of tumor. 15
We herein present a case of a true carcinosarcoma (in situ squamous cell carcinoma and leiomyosarcoma) in the hypopharynx. We also review its clinical manifestations, pathogenesis, clinicopathology, immunohistochemistry, diagnosis, prognostic factors, and therapeutic approaches; identify the presence of PI3K, HIF-1α, HK-II, and GLUT-1 within the carcinosarcoma; and discuss the role of 18F-FDG PET/CT in diagnosing carcinosarcoma. This case is being reported to help physicians ensure a correct diagnosis and seek effective targeted therapies for hypopharyngeal carcinosarcoma.
Case report
This study protocol was approved by the Research Ethics Committee of the First Affiliated Hospital, College of Medicine, Zhejiang University. Verbal informed consent was obtained from the patient.
A 72-year-old man presented to our institution in April 2015 with a 2-month history of cough, progressive dysphagia, and occasional pharyngalgia. He also had a 1-month history of slurred speech, chest pain, dyspnea, and the sensation of a foreign body in his hypopharynx without chest pain, nausea, vomiting, hemoptysis, dizziness, headache, chills, or fever. The patient reported no weight loss. He had a 50-year history of daily moderate alcohol consumption and a 50-year history of smoking 20 cigarettes per day. He had stopped drinking and smoking for 1 year. He denied previous exposure to radiation. The patient had undergone surgery for carcinoma of the rectum 1 year previously with good postoperative recovery. He reported no history of hypertension, diabetes, or heart disease. He was taking no medications and reported no allergies.
Physical examination revealed a giant neoplasm toward the laryngeal cavity of the posterior hypopharyngeal wall. The tumor was compressing a vocal cord and had a rough surface without ulceration or hemorrhage. Several 0.5- × 0.5-cm lymph nodes were found at bilateral level II of the neck; the nodes were of moderate hardness and showed poor mobility and no tenderness.
Laryngoscopy revealed a neoplasm in the hypopharynx (Figure 1). A CT scan confirmed the presence of a large, irregular submucosal mass occupying the left pyriform sinus (Figure 2). An 18F-FDG PET/CT scan confirmed a mass with high 18F-FDG uptake on the left posterior hypopharyngeal wall (Figure 3). The maximum standardized uptake value (SUVmax) was 10.3, indicating the presence of high glycometabolism and malignancy. The scan also revealed several small lymph nodes at bilateral level II of the neck, the SUVmax of which was 2.5. Other imaging examinations and routine blood examinations showed no severe abnormalities. The patient was diagnosed with hypopharyngeal carcinosarcoma, cT3N2M0, Stage IVA based on the 2002 American Joint Committee on Cancer TNM classification of tumors of the hypopharynx.

Laryngoscopy revealed a neoplasm in the hypopharynx (arrows).
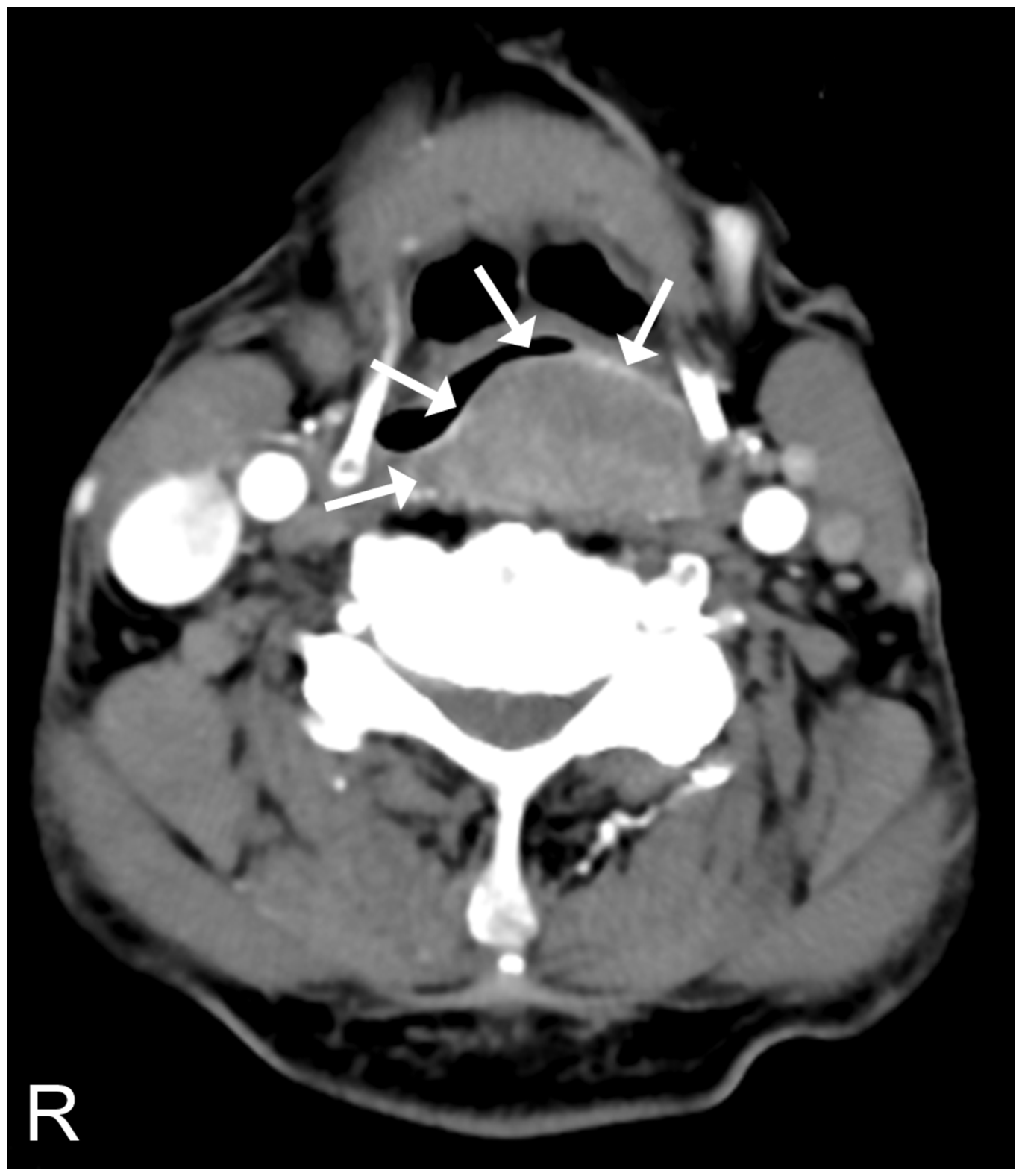
An axial computed tomography scan revealed the presence of a large, irregular submucosal mass occupying the left pyriform sinus (arrows).

(a) An 18F-fluorodeoxyglucose PET/CT scan revealed a mass on the left posterior hypopharyngeal wall (arrows). (b) Transaxial view of the mass along the lesser curve of the body of the hypopharynx were observed on an overlay of the CT, PET, and PET/CT images (arrows). MIP images showed a large mass on the left posterior hypopharyngeal wall measuring 5.0 × 3.5 cm with a maximum standardized uptake value of 10.3. PET, positron emission tomography; CT, computed tomography; MIP, maximum intensity projection.
We first performed a tracheotomy under local anesthesia and a biopsy of the hypopharyngeal neoplasm during direct laryngoscopy under general anesthesia. Examination of the biopsy specimen revealed a soft tissue tumor composed of spindle cells with cellular pleomorphism; thus, malignancy could not be excluded. Next, the patient underwent bilateral dissection of the neck including levels IIA, IIB, III, IV, and V with preservation of the jugular vein, carotid artery, and cranial nerves X and XI. The hyoid bone was partially resected with preservation of both the superior laryngeal nerve and artery. An incision was made in the thyrohyoid membrane, and a giant oval neoplasm was observed on the posterior hypopharyngeal wall. The tumor did not involve the epiglottis, inlet of the esophagus, bilateral pyriform sinuses, or bilateral arytenoid cartilages. Next, the hypopharyngeal tumor was excised with 2-cm surgical margins under direct vision. Intraoperative surgical margin frozen section examination revealed severe hyperplasia of the focal squamous epithelium on the superior and right surgical margins. The left and esophageal surgical margins were negative. Extended resection of the superior and right surgical margins was then performed. Finally, we inserted hypopharyngeal prosthetics with pedicled muscle flaps and a HeaL rehabilitation membrane (Yantai Zhenghai Biotechnology Co., Ltd., Yantai Economic and Technological Development Zone, Shandong, China). No complications were observed during the postoperative period. Following surgery, the patient underwent radiotherapy at Taizhou Hospital (Taizhou, China).
The tumor of the posterior hypopharyngeal wall was 5.0 × 3.5 × 3.0 cm, was nodular in shape (Figure 4(a) and (b)), and possessed a gray-yellow section (Figure 4(c)). The tumor was biphasic in appearance, consisting of bundles of spindle cells that were vacuolar, abnormally mitotic, basophilic, hyperchromatic, and pleomorphic in appearance. Obvious giant cells were present in the tumor. Furthermore, several areas of severe hyperplasia of squamous epithelium and in situ squamous cell carcinoma were observed (Figure 5(a)–(c)). The superior surgical margin revealed moderate to severe hyperplasia of the squamous epithelium. The right surgical margin was free of tumor cells. Interestingly, no lymph node metastasis was present. Immunohistochemistry revealed carcinomatous cells with membrane positivity for cytokeratin (CK) (Figure 5(d)) and sarcomatous cells with membrane positivity for smooth muscle actin (SMA) (Figure 5(f)) and negativity for CK (Figure 5(e)). Furthermore, GLUT-1, HIF-1α, and PI3K were positive in both the carcinomatous and sarcomatous cells, whereas HK-II was negative in the sarcomatous cells and positive in the carcinomatous cells (Figure 6). Reactivity for CK, desmin, CD34, SMA, Ki-67, MyoD1, myogenin, CD117, S-100, and CD68 was detected at various levels. The Ki-67 index was approximately 25%. Immunoreactivity was positive for CD68 and negative for desmin, MyoD1, myogenin, CD117, S-100, and CD34 (data not shown).

(a, b) The tumor of the posterior hypopharyngeal wall was 5.0 × 3.5 × 3.0 cm and exhibited a nodular shape. (c) The tumor was gray-yellow upon sectioning.
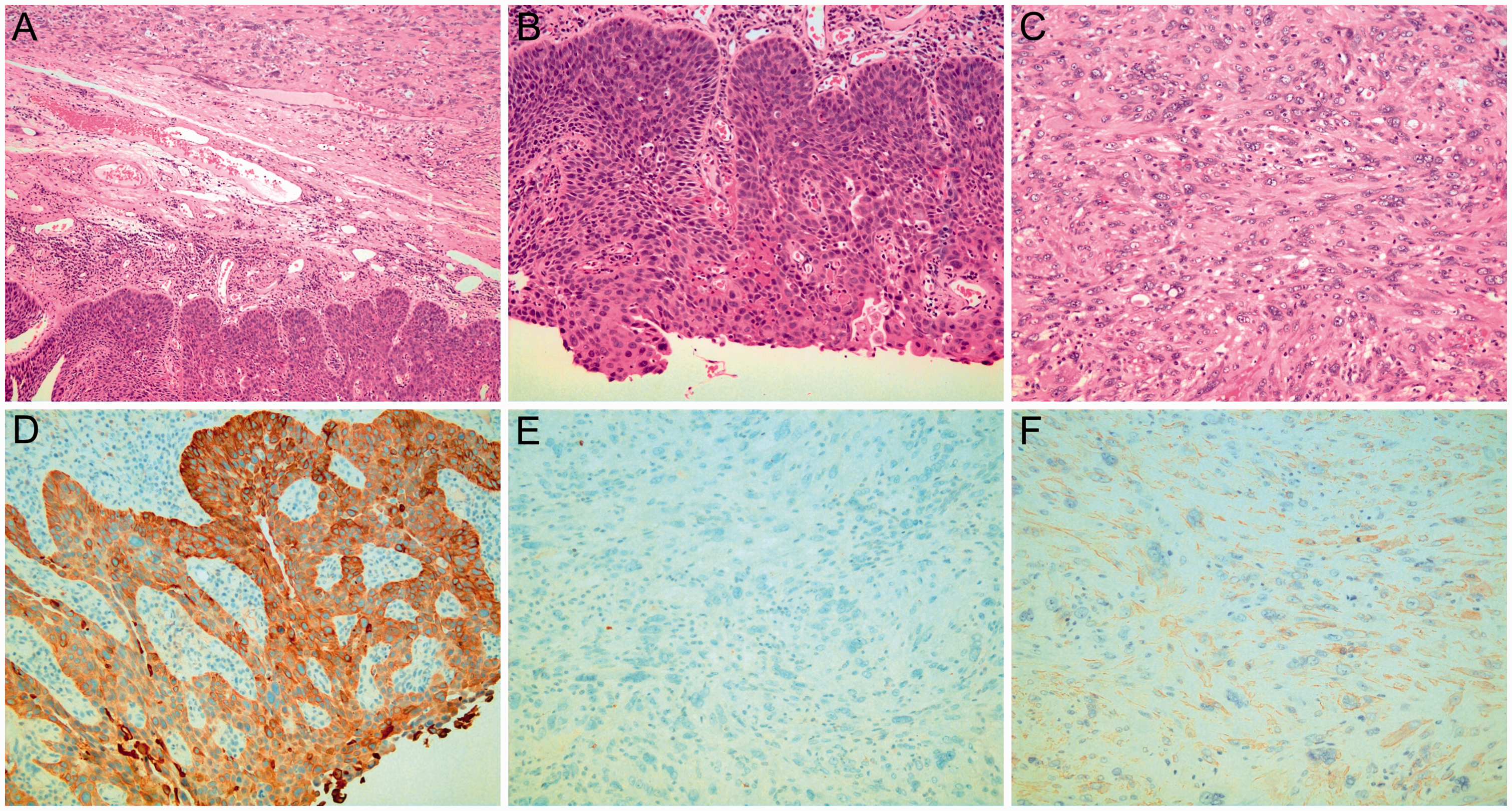
Definitive histological examination showed a carcinosarcoma with carcinomatous and sarcomatous components. Immunohistochemistry with monoclonal antibodies for cytokeratin (CK), desmin, CD34, smooth muscle actin (SMA), Ki-67, MyoD1, myogenin, CD117, S-100, and CD68 was performed. The tumor showed strong positivity for CK and SMA. (a) Hematoxylin–eosin (original magnification, ×100). In situ squamous cell carcinoma with cells infiltrating the subepithelial corium. Upper right: Undifferentiated spindle cell carcinoma cells. (b) Hematoxylin–eosin (original magnification, ×200). In situ squamous cell carcinoma with atypical cells at the basal layers. (c) Hematoxylin–eosin (original magnification, ×200). A sarcomatous component with a vacuolar, abnormal mitotic, basophilic, hyperchromatic, and pleomorphic appearance and obvious giant cells within the tumor. (d) CK (original magnification, ×200). Immunohistochemistry revealed carcinomatous cells with membrane positivity for CK. (e) CK (original magnification, ×200). Immunohistochemistry revealed sarcomatous cells with membrane negativity for CK. (f) SMA (original magnification, ×200). Immunohistochemistry revealed sarcomatous cells with membrane positivity for SMA.

Immunohistochemistry with monoclonal antibodies for glucose transporter-1 (GLUT-1), hexokinase-II (HK-II), hypoxia-inducible factor-1α (HIF-1α), and phosphatidylinositol 3-kinase (PI3K). GLUT-1, HIF-1α, and PI3K were positive in both carcinomatous and sarcomatous cells, whereas HK-II was positive in carcinomatous cells and negative in sarcomatous cells. (a) GLUT-1 (original magnification, ×100). Carcinomatous cells (upper right) and sarcomatous cells (lower left) were positive for GLUT-1. (b) GLUT-1 (original magnification, ×200). Carcinomatous cells were positive for GLUT-1. (c) GLUT-1 (original magnification, ×200). Sarcomatous cells were positive for GLUT-1. (d) HK-II (original magnification, ×100). Carcinomatous cells (upper right) were positive for HK-II, whereas sarcomatous cells (lower left) were negative for HK-II. (e) HK-II (original magnification, ×200). Carcinomatous cells were positive for HK-II. (f) HK-II (original magnification, ×200). Sarcomatous cells were negative for HK-II. (g) HIF-1α (original magnification, ×100). Carcinomatous cells (upper right) and sarcomatous cells (lower left) were positive for HIF-1α. (h) HIF-1α (original magnification, ×200). Carcinomatous cells were positive for HIF-1α. (i) HIF-1α (original magnification, ×200). Sarcomatous cells were positive for HIF-1α. (j) PI3K (original magnification, ×100). Carcinomatous cells (upper right) and sarcomatous cells (lower left) were positive for PI3K. (k) PI3K (original magnification, ×200). Carcinomatous cells were positive for PI3K. (l) PI3K (original magnification, ×200). Sarcomatous cells were positive for PI3K.
Definitive histological examination showed a carcinosarcoma (in situ squamous cell carcinoma and leiomyosarcoma) in the hypopharyngeal wall with disease-free margins (pT3N0M0, Stage III). After 10 months of follow-up, the patient died of systemic metastasis.
Discussion
The histogenesis of carcinosarcoma is incompletely understood. Several terms have been used to describe this type of tumor: carcinosarcoma, sarcomatoid carcinoma, polypoid squamous cell carcinoma, carcinoma expleomorphic adenoma, metastasizing mixed tumor, spindle squamous cell carcinoma, squamous cell carcinoma spindle cell variant, spindle cell carcinoma, pseudocarcinosarcoma, pseudosarcoma, squamous cell carcinoma with sarcoma-like stroma, squamous cell carcinoma with pseudosarcoma (Lane tumor), carcinoma with pseudosarcoma, pleomorphic carcinoma, and metaplastic carcinoma.26–28 No systematic review of hypopharyngeal carcinosarcoma has been reported to date. In the present study, we reviewed the English-language literature using PubMed and Web of Science. We searched for the following keywords: (1) polypoidal squamous cell carcinoma or sarcomatoid carcinoma or carcinoma expleomorphic adenoma or metastasizing mixed tumor or spindle squamous cell carcinoma or squamous cell carcinoma spindle cell variant or pseudosarcoma or pseudocarcinosarcoma or squamous cell carcinoma with pseudosarcoma or spindle cell carcinoma or carcinoma with pseudosarcoma or squamous cell carcinoma with sarcoma-like stroma or metaplastic carcinoma or carcinosarcoma or pleomorphic carcinoma and (2) hypopharynx or pyriform sinus or epiglottis or aryepiglottic folds or inlet of larynx or arytenoid cartilage or laryngopharynx or vallecula epiglottica or postcricoid space.
The first case of hypopharyngeal carcinosarcoma, which occurred in the pyriform sinus and was of the infiltrative form, was reported by Kahler in 1908. 29 Harvey and Hamilton presented the second case in 1935, but no detailed report has been published. 30 Since 1949, an additional 17 cases (including the present case) have been described1,13–15,30–37 (Table 1). The patients in these recent cases were predominantly male: 16 were male and only 1 was female. The mean age of the 17 patients was 61.5 years (range, 28–82 years). Seven patients had a history of cigarette smoking, three had a history of alcohol consumption, and two had a history of previous irradiation. Data for the other patients could not be obtained. Of all 17 tumors, 5 (29.4%) were located in the epiglottis, 5 (29.4%) in the pyriform sinus, 4 (23.5%) in the aryepiglottic fold, 3 (17.6%) in the hypopharyngeal wall, 1 (5.9%) in the postcricoid region, 1 (5.9%) in the laryngeal inlet, 1 (5.9%) in the tonsil, 1 (5.9%) in the false cord, and 1 (5.9%) in the vallecula (in some patients, a single tumor affected more than one site). Data for two patients (Cases 15 and 16) could not be obtained. Dysphagia was the most frequently reported symptom (41.2%, 7/17 patients); other symptoms included hoarseness (23.5%, 4/17 patients), dyspnea (11.8%, 2/17 patients), cough (11.8%, 2/17 patients), wheezing (5.9%, 1/17 patients), slurred speech (5.9%, 1/17 patients), a neck mass (5.9%, 1/17 patients), and an abnormal sensation in the throat (sensation of burning, soreness, stridor, foreign body, and discomfort; 41.2%, 7/17 patients). Data for three patients (Cases 8, 15, and 16) could not be obtained. Furthermore, the reported macroscopic features varied. Most tumors were polypoid (23.5%, 4/17 tumors) or pedunculated (17.6%, 3/17 tumors) in appearance. We found only one tumor with surface ulcers (Case 12). Data for four patients (Cases 8, 11, 15, and 16) could not be obtained. Of all patients, only 1 (5.9%) underwent radiotherapy, whereas 15 (88.2%) underwent surgery (6 of whom underwent neck dissection). Of these 15 patients, 1 (6.7%) underwent preoperative radiotherapy, 1 (6.7%) underwent preoperative chemotherapy, and 8 (53.3%) received prescribed postoperative treatments. Among all 17 patients in the study, 5 (29.4%) received radiotherapy alone and 3 (17.6%) received both chemotherapy and radiotherapy. Full data for two patients (Cases 7 and 11) could not be obtained. Eleven patients (64.7%) were alive at follow-up.
Summary of 16 previously described cases of hypopharyngeal carcinosarcoma and the present case

M = male; F = female; Y = yes; N = no; AEF = aryepiglottic fold; PS = pyriform sinus; HPW = hypopharyngeal wall; Tx = treatment; IH = immunohistochemistry; RT = radiotherapy; CT = chemotherapy; Met = metastases; L = left; R = right; LE = local excision; SHPT = subhyoid pharyngotomy; SGLT = supraglottic laryngectomy; BND = bilateral neck dissection; RND = right neck dissection; LND = left neck dissection; TLT = total laryngectomy; PHLHPT = partial hemilaryngohypopharyngectomy; VRS = volume-reduction surgery; THLT = total hypolaryngectomy; IOTEVP = insertion of tracheoesophageal voice prosthesis; HP = hypopharyngeal prosthetics; T = tumor; N = node; M = metastasis; ALO = acute laryngeal obstruction; B = bilateral; CLN = cervical lymph nodes; FU = follow-up; h = hour; d = day; mo = month; A = alive; D = died; AWD = alive with disease; CA = cardiac arrest; PD = progressive disease; MDM = multiple distant metastases; adr = adrenals; kid = kidneys; pc = pericardium; int sept = interauricular septum; Lv/pn inv = lymphovascular and perineural invasion.
*The patient received preoperative radiotherapy/chemotherapy.
#The patient received postoperative radiotherapy/chemotherapy.
/The data were not obtained.
&The patient received previous irradiation.
The surgical procedures included total laryngectomy/hypolaryngectomy, partial laryngectomy/hypolaryngectomy (subhyoid pharyngotomy, supraglottic laryngectomy, and partial hemilaryngohypopharyngectomy), local excision, and volume-reduction surgery. In Case 11, the patient received a tracheoesophageal voice prosthesis. As concluded by Iqbal et al. 1 in 2015, surgery alone or combined with radiotherapy is effective for spindle cell carcinoma of the head and neck. In 2014, Zheng et al. 13 showed that surgery is preferred, while radiotherapy was an effective adjunctive therapy. In 2013, however, Zhang et al. 14 did not recommend radiotherapy because of radioresistance of the mesenchymal component.
Novel targets for targeted therapy, such as TP53, KRAS, EGFR, PIK3CA, and VEGF, have been found in a human uterine carcinosarcoma cell line and patient-derived xenograft models of ovarian carcinosarcoma.20,21 However, treatment with a VEGF inhibitor (BMS-690514) combined with carboplatin/paclitaxel did not inhibit tumor growth or improve animal survival. 21 Thus, better predictive markers of treatment response are needed. GLUT-1, HK-II, HIF-1α, and PI3K are hypoxic markers that play vital roles in the Warburg effect. We previously confirmed that inhibition of GLUT-1 inhibited proliferation and enhanced the radiosensitivity of Hep-2 cells.38,39 GLUT-1 expression is reportedly regulated by the PI3K/Akt signaling pathway and HIF-1α. 40 In one study, the PI3K/AKT pathway was activated in Merkel cell carcinoma and was suggested as a useful new therapeutic target. 41 We have confirmed that co-inhibition of the PI3K/Akt signaling pathway and GLUT-1 expression may improve the radiosensitivity of laryngeal carcinoma. 42 Because HK-II has been detected in various kinds of tumors, targeted inhibition of HK-II may enhance the radiosensitivity of laryngeal carcinoma. 43 In the patient described in the present report, immunohistochemistry showed that GLUT-1, HIF-1α, and PI3K were positive in both carcinomatous and sarcomatous cells, whereas HK-II was positive only in carcinomatous cells. Thus, we propose that targeting GLUT-1, HK-II, HIF-1α, and PI3K may be effective in the treatment of hypopharyngeal carcinosarcoma.
Follow-up information for 16 (93.75%) of the 17 patients described in the literature was available (Case 7 was the exception). The average length of follow-up was 21.9 (range, 0–72) months. Of the 16 cases we reviewed, including our case, 11 (68.8%) patients were alive at the time of follow-up. The 3-year survival rate was 70.0% (Figure 7), which is similar to the 2.5-year survival rate of 65.0% reported by Batsakis. 44 However, the follow-up was not long enough. Only five patients were still alive at 3 years (Cases 3, 4, 5, 12, and 15). Of the remaining 11 patients, 6 were alive (Cases 8, 9, 10, 11, 13, and 14) and 1 (Case 1) was discharged. Thus, accumulation of additional cases is essential to calculate an accurate survival rate and determine appropriate prognostic factors and treatments.

Overall survival probabilities of the 16 patients were estimated using Kaplan–Meier curves.
According to Klijanienko et al., 31 spindle-shaped carcinoma differs from true carcinosarcoma. A diagnosis of carcinosarcoma requires histological evidence of carcinoma and sarcoma together both in the primary and secondary sites. The term carcinosarcoma describes tumors consisting of both carcinomatous and sarcomatous tissues, especially involving malignant skeletal muscle, cartilage, and bone cells. Sarcomatoid squamous cell carcinoma should be differentiated from carcinosarcoma. 31 In addition, thorough immunohistochemical examination is preferred to ensure a correct diagnosis. In sarcomatoid squamous cell carcinoma, CK is positive in both the epithelial and sarcomatoid components. Occasionally, vimentin may also be positive in the sarcomatoid component. Epithelial–mesenchymal transition may be involved in the development of sarcomatoid squamous cell carcinoma 45 because a morphological transition can be seen from epithelial cells to spindle cells. The transitional zone is always seen between the two components. In carcinosarcoma, however, CK is positive in the epithelial component and vimentin is positive in the mesenchymal component. CK is always negative in the mesenchymal component. 10 The transitional zone is rarely observed. Table 2 summarizes the studies in which immunohistochemical examinations were performed.13–15,31,33 Based on the above criteria, only six of the reviewed cases (Cases 8, 9, 11, 12, 13, and 14) can be diagnosed as true carcinosarcoma. Notably, the carcinomatous component of all six cases was positive for CK, and the sarcomatous component was positive for SMA in Case 14, for muscle-specific actin in Case 11, and for vimentin in Cases 8, 9, 12, 13, and 14. In our patient, the squamous cell components were positive for CK, while the spindle-shaped tumor cells were negative for CK but positive for SMA. The Ki-67 index was approximately 25%. Immunoreactivity was positive for CD68 and slightly positive for desmin, whereas it was negative for CD34, MyoD1, myogenin, CD117, and S-100. No transitional zone was observed between the epithelial component and mesenchymal component. The two components were not mixed; they seemed to collide with each other. Because the lesion was a single mass, it was a carcinosarcoma based on the collision tumor hypothesis. The patient was ultimately diagnosed with true carcinosarcoma.
Summary of studies with immunohistochemistry

CK = cytokeratin; MSA = muscle-specific actin; HHF35 = muscle cell actin isotypes; EMA = epithelial membrane antigen; SMA = smooth muscle actin.
We reviewed the median SUVmax or SUVmax of the carcinosarcomas on 18F-FDG PET/CT scans performed in 12 previous studies (Table 3). The various anatomical sites were the uterus (six studies, median SUVmax: 10.13),25,46–50 prostate (one study, SUVmax: 9.70), 23 liver (one study, SUVmax: 17.10), 24 stomach (one study, SUVmax: 11.90), 51 pulmonary system (two studies, median SUVmax: 10.47),52,53 and hypopharynx (one study, SUVmax: 8.06). 15 Furthermore, the median SUVmax of all of these studies was 11.23, similar to the value of 10.30 reported in our study. Therefore, we believe that 18F-FDG PET/CT scans can be chosen to distinguish hypopharyngeal carcinosarcoma from benign tumors. However, the range of the median SUVmax of these previous studies is large (4.10–17.10). Because the morbidity of hypopharyngeal carcinosarcoma is extremely low, it is difficult to differentiate carcinosarcoma from other malignant tumors using only the SUVmax. Further studies are needed to assess the clinical significance of SUVmax for differentiating carcinosarcoma from other malignant tumors.
Summary of median SUVmax or SUVmax of carcinosarcoma on 18F-FDG PET/CT in various anatomical positions

SUVmax = standardized uptake value; 18F-FDG PET/CT = 18F-fluorodeoxyglucose positron emission tomography/computed tomography.
Conclusions
In summary, we have reported a case of a true carcinosarcoma of the hypopharynx. This type of tumor is rare; only 6 cases of true carcinosarcoma verified by immunohistochemistry have been reported among 16 cases reported to date. Smoking, alcohol consumption, and radiation exposure may be predisposing risk factors for hypopharyngeal carcinosarcoma. Surgery is the main treatment for hypopharyngeal carcinosarcoma according to the literature. Targeting of PI3K, HIF-1α, HK-II, and GLUT-1 may be effective in the treatment of hypopharyngeal carcinosarcoma. Sarcomatoid squamous cell carcinoma should be differentiated from carcinosarcoma. A thorough immunohistochemical examination is required to ensure a correct diagnosis. In carcinosarcoma, CK is positive in the epithelial component and vimentin is positive in the mesenchymal component. CK is always negative in the mesenchymal component. 18F-FDG PET/CT may be helpful in the diagnosis of hypopharyngeal carcinosarcoma.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (Nos. 81172562 and 81372903) and the Science and Technology Department of Zhejiang Province, China (No. 2016C33144).
