Abstract
Objective
The goal of this review was to determine whether calcium silicate (wollastonite) as a bone graft material is a viable alternative to autogenous bone or whether the evidence base for its use is weak.
Methods
In this systematic review, electronic databases (MEDLINE/PubMed and BVS) were searched for relevant articles in indexed journals. Articles published in a 10-year period were identified (n = 48). After initial selection, 17 articles were assessed for eligibility; subsequently, seven articles were excluded and 10 articles were included.
Results
Among the studies included, 20% emphasized the importance of randomization, which adds reliability to the study, minimizing the risk of bias. High variability was observed in the material used, such as additives, amounts, dosage, and chemical alterations, rendering direct comparison among these studies impossible. The experimental periods varied considerably; one of the studies did not include statistical analysis, weakening the evaluation. Nonetheless, the true potential of wollastonite as a graft material conducive to new bone formation was reported in all studies.
Conclusion
The results support the use of wollastonite as a bone graft material. The initial research question was answered despite the significant variability observed among these preclinical studies, which hindered the precision of this analysis.
Introduction
The current gold standard in the treatment of pathological, degenerative, esthetic, or traumatic conditions is autogenous bone. However, there is a need to replace autogenous bone with a new biocompatible natural or synthetic bone substitute for tissue regeneration, to minimize postoperative trauma.1,2
The ideal material should mimic bone in shape, size, texture, and performance, promoting an adequate response in the biological system. 3 Synthetic materials have emerged as a relevant option because there is no risk of disease transmission and because these materials are available in potentially unlimited quantities.
Bone repair materials currently in use are either bioinert, bioresorbable, or biodegradable, depending on the characteristics of the treatment site or the subsequent treatments planned. Bioinert materials remain in the treated site and interact with the medium without inducing rejection by surrounding bone. Biodegradable materials ideally should promote bone formation as they are resorbed, and both the material and its degradation products must be well accepted by the organism. Degradation of bone biomaterials should be gradual and proportional to new bone formation: neither too fast, nor too slow. If too fast, the healing process can leave gaps that may result in voids or fibrosis in the newly formed bone. If degradation is slower than new bone formation, bone repair may be delayed.
Calcium silicate, also known as wollastonite, is capable of inducing
Considering the limitations of wollastonite as a bone graft material, 8 the aim of this systematic review was to seek greater evidence in the scientific literature to support the utilization of this biomaterial, which is still not widely applied in clinical practice. 9
Methods
Protocol and search strategy
The methodology used was based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (http://www.prisma-statement.org) and on the Population, Intervention, Comparison, Outcome (PICO) model to frame the theme and the search strategy (Table 1).10–14
Systematic search strategy (PICO model).

A literature search of the MEDLINE/PubMed and BVS electronic databases was conducted between 23 February 2016 and 23 December 2016; relevant articles published in indexed journals in the previous 10 years were included. Prospective studies were evaluated for possible inclusion.
Focused question and study objective
The focused question in this systematic review was, “Can wollastonite (CaSiO3) be used to effectively aid the bone repair process?”
Screening and selection
Review articles,
Data collection process
The formulations used; characteristics of the bone defects; types of treatment performed; clinical, histological, and radiographic results; and statistical analyses performed in the articles retrieved were systematically recorded.
Risk of bias assessment
The methodological quality of the studies included was evaluated both independently and jointly by two evaluators (AMO, GVOF), using the Cochrane collaboration tool for assessing risk of bias, and the PRISMA and the CONSORT statements.13,15,16 The risk of bias was assessed based on the following quality criteria: randomization, standardization of the study execution, use of test and control groups, standardization of the bone defects, statistical analysis, and results obtained. All of these criteria were established as adequate, inadequate, unclear, or not described. 16
Articles were deemed as presenting low risk of bias when all the criteria were identified and accepted (low likelihood of bias affecting the results), moderate risk of bias when one of the criteria was not found or when there were doubts about the results, and high risk of bias when two or more criteria did not match the parameters selected.
Any discrepancies between the two evaluators were resolved through discussion; when no consensus was reached, a third evaluator was consulted (MSA).
Data analysis
The quality of the studies included in the review was assessed, focusing on the similarities (homogeneity) and differences (heterogeneity) among the studies. GraphPad Prism 7.0c for Mac (GraphPad Software, La Jolla, CA, USA) was used for data analysis.
Results
After application of the inclusion and exclusion criteria, 48 articles were selected initially.
Specifically, the search using the PICO model, as described in Table 1, yielded 26 articles from MEDLINE/PubMed and 22 articles from BVS published within the past 10 years. After preliminary analysis of the abstracts, 17 articles were selected for full analysis and evaluation, after which seven articles were excluded and 10 articles were selected for detailed analysis (Figure 1).

Flow diagram of the screening and selection process
Only data from
Main data from the studies selected.
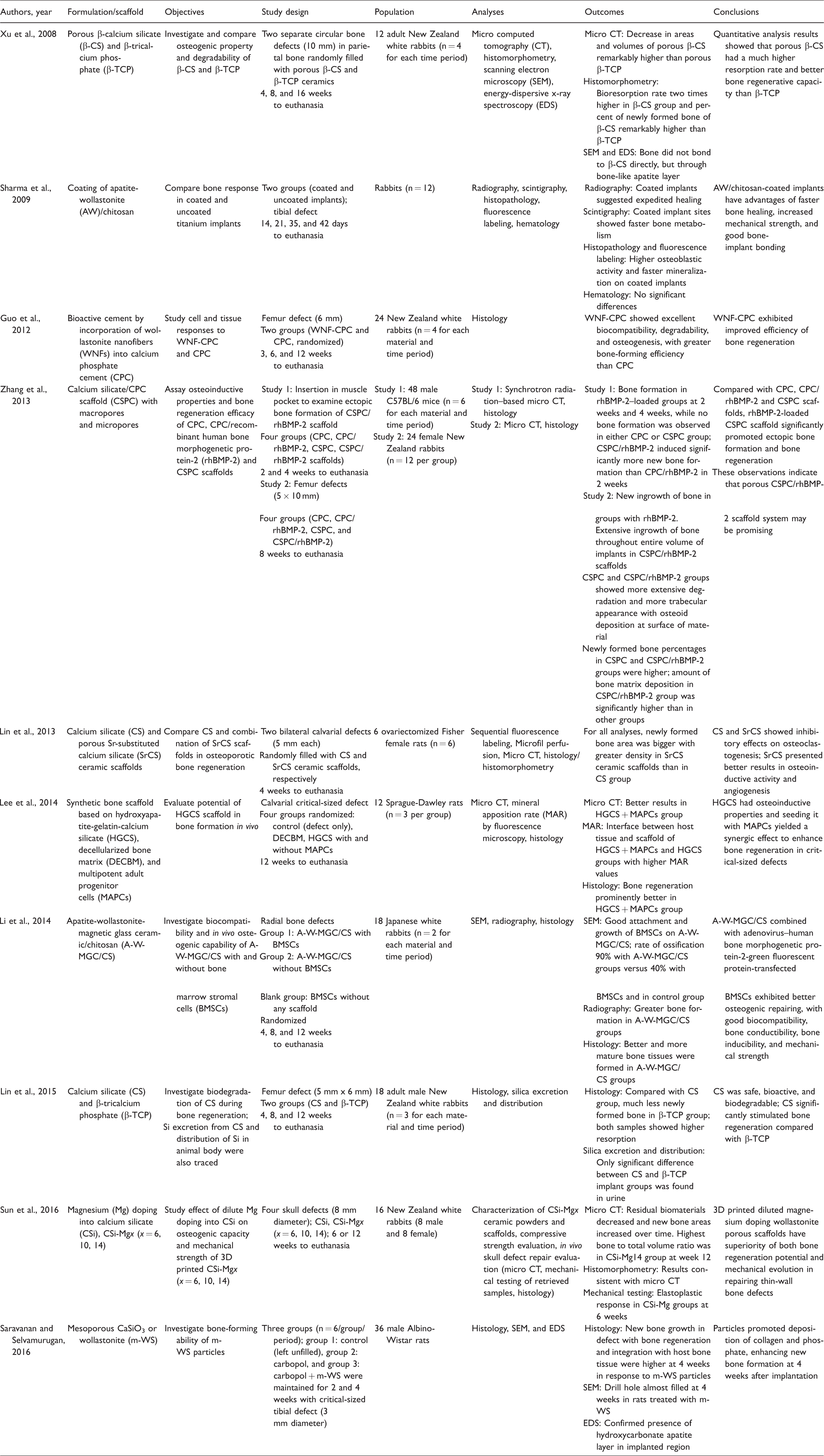
Excluded studies.

Study characteristics
Among the studies analyzed, wollastonite was used in animal model studies (parietal, femoral, tibial, and radial bones), usually in association with other biomaterials or growth factors, as well as in adapted formulations. Standardized creation of bone defects was performed in nine studies, and in only one study wollastonite was used as an implant coating. No clinical studies were found.
All studies included histological and/or histomorphometric analyses of the samples obtained, and five studies used micro computed tomography as a tool for analysis.17–20 Additional methods used in the analyses were scanning electron microscopy and energy-dispersive x-ray spectroscopy, 17 radiography,21,22 scintigraphy, 21 fluorescence labeling,18,21 Microfil injection compound perfusion (Flow Tech, Inc., Carver, MA, USA), 18 mineral apposition rate, 19 and scanning electron microscopy.22,23 All analytic methods rendered useful information. Hematological and urinary excretion analyses did not show relevant changes.
Use of wollastonite was associated with better tissue biocompatibility,24–26 faster biomaterial resorption rate,17,21 and improved bone repair,17–19,21,24–26 especially in the adapted formulations.
Quality assessment
Results from the quality assessment of the studies selected for detailed analysis are shown in Table 4. This systematic review followed the CONSORT statement guidelines. 15
Quality assessment of studies analyzed.

Y, yes; N, no; ND, not described; SD, standard deviation; ANOVA, analysis of variance.
Discussion
Wollastonite has been studied mainly in preclinical studies aiming to validate this material for clinical applications. Accordingly, in the present systematic review, only animal model studies were found. Wollastonite does not show evidence of carcinogenicity and has been evaluated as a bone substitute because of its biocompatibility, high mechanical resistance, and excellent bioactivity compared with calcium phosphate bioceramics. 27 Evidence for these qualities has been previously assessed through various tools, such as micro computed tomography, histomorphometric analysis, scanning electronic microscopy, and others. This was corroborated by the articles included in the present review, which also aimed to verify the osteogenic potential of wollastonite particles.17,21,22
New techniques have been developed for the synthesis of wollastonite, including the use of additives and processing at lower temperatures in order to improve its physical, chemical, and biological properties.18,22,24 Analysis of all the procedures employed to improve the performance of this material underscores the fact that great effort has been placed to this end, as demonstrated in the literature. In addition, structural changes and experiments have been performed to test the full potential of this material; favorable results were observed for the association of wollastonite with recombinant human bone morphogenetic protein-2 28 and for magnesium-doped wollastonite, both in terms of bone regeneration potential and for improved mechanical properties. 19 Moreover, the ability of wollastonite to stimulate the bone regeneration process was compared with β-tricalcium phosphate, a well-known and widely used material. 29
De Aza
Among the studies included in this review (n = 10), we verified that only two (20%) emphasized the importance of randomization,18,19 a procedure that adds reliability to the study and minimizes the risk of bias. Nonetheless, analysis of each study showed that in all of them the execution, research model, and type of defect were standardized. Still, great variability was observed with regard to the characterization of the material used, its association with wollastonite and additives, as well as the amounts, dosage, and chemical changes, rendering the direct comparison among these studies impossible. Moreover, the experimental periods varied greatly; one of the studies did not include statistical analysis, weakening the validity of its findings. 23
Greater standardization of the research models, duration of treatment, and materials employed would help to better demonstrate the true potential of wollastonite as a graft material conducive to new bone formation, despite the fact that all the articles reviewed have reported excellent results in this regard.
Clinical studies should be able to confirm the clinical viability of wollastonite, and verify its association with calcium phosphate ceramics in proportions yet to be established, aiming to improve bone repair. Associations with other bone turnover ion cofactors might also be studied, with the same goal.
Conclusion
The preclinical studies included in this systematic review demonstrate that wollastonite (CaSiO3) can be used to effectively aid the bone repair process, thus answering the focused question affirmatively. However, great variability was observed among the studies, hindering the precision of this analysis and highlighting the importance of conducting standardized studies.
Footnotes
Ethical approval and patient consent
Not required (human subjects were not involved).
Authors' contributions
AMO and GVOF developed the systematic review. MSA contributed to the introduction and discussion, in addition to serving as the third reviewer. JMG corrected the manuscript and provided the necessary guidance to ensure the logical sequence of this article.
Availability of data and materials
The datasets generated and/or analyzed in the present study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
