Abstract
Objective
To investigate the effect of acidity on gastric cancer SGC7901 cells in terms of autophagy and provide a new strategy for therapeutically targeting gastric cancer autophagy in an acidic environment.
Methods
Transmission electron microscopy (TEM) and confocal laser scanning microscopy were used to examine the effect of an acidic environment on autophagosome formation. Light chain 3 (LC3) and p62 levels in SGC7901 cells exposed to acidic conditions were measured using Western blot analysis. To explore changes in autophagy flux, the cells were treated with an inhibitor of autophagy bafilomycin A1. The CCK-8 assay was performed to determine if inhibiting acid-induced autophagy affected cell proliferation.
Results
Increased autophagosome formation was observed by TEM. Punctate LC3 structures were observed in cells cultured under acidic conditions, whereas untreated cells exhibited diffuse and weak staining for punctate LC3 structures. Cytoplasmic LC3-I translocated to the autophagic membrane (LC3-II) levels increased under acidic conditions, whereas p62 levels decreased. The bafilomycin A1-induced inhibition of autophagy caused by the acidic environment inhibited cell proliferation.
Conclusion
The acidic environment upregulates autophagy in SGC7901 cells. In long-term culture, a stable and high level of autophagy is maintained in an acidic environment, which has a protective effect on cells.
Introduction
Gastric cancer is one of the most common malignancies in the world. 1 In 2012, there were approximately 951 600 new cases of gastric cancer and 723 100 deaths from gastric cancer worldwide. 1 However, the biological behaviour of gastric cancer has remained unclear. Although tumour behaviour is mainly determined by parenchymal cells, increasing evidence has indicated that it is also affected by the tumour microenvironment.2–6 Hence, understanding how tumour cells adapt to their microenvironment in order to enhance their drug resistance mechanisms is critical and has the potential to lead to a cure.
Low extracellular pH is a characteristic of the microenvironment of malignant tumour cells. 7 Previous studies have found that the extracellular pH of tumour cells is lower than that of normal tissues, 8 leading to an acidic microenvironment that exerts various biological effects on cancer cell behaviour. Acidity can change gene expression in tumour cells, such as increasing the expression of proteolytic enzymes that promote invasion and migration; and altering vascular endothelial growth factor expression, which promotes tumour angiogenesis.4,7,9 However, the mechanisms underlying how gastric tumour cells adapt to an acidic microenvironment remain unclear. Recent studies on melanoma and breast cancer have shown that an acidic environment can enhance autophagy, but this type of analysis has not been performed on gastric cancer.10,11 We hypothesize that autophagy is upregulated in gastric cancer cells under acidic stress.
Autophagy is a highly conserved mechanism of cell metabolism that degrades misfolded proteins and damaged organelles, and autophagy-degraded proteins and organelles can be reused by cells to promote survival under various stresses. 12 Autophagy in tumour cells plays various roles; however, sustained autophagy may also lead to cell death. 13 Studies have shown that breast, colon and prostate tumours undergo autophagic cell death after antitumour therapy;14,15 and irradiation can also cause tumour autophagy or autophagic cell death. 16 However, autophagy induced by chemotherapeutic agents can also elicit a protective response. 17 Therefore, it is important to determine the specific role of autophagy in tumour cells under acidic stress.
Because autophagy, or type II nonapoptotic cell death, is characterized by the formation of double-membrane vesicles, the first objective of this current study was to investigate the effects of pH on autophagosome formation in SGC7901 gastric cancer cells and to determine punctate light chain 3 (LC3) structure distribution. 18 Using Western blot analysis, the study investigated whether cytoplasmic LC3-I translocated to the autophagic membrane (LC3-II) when autophagosomes were formed. The study also investigated p62, which is a cytosolic protein that transports ubiquitinated proteins to autophagosomes 19 and is degraded during autophagy. When autophagy is suppressed, the level of p62 is elevated; 19 thus, the p62 level can be used as a measure of the level of autophagy. The study also aimed to determine if autophagy induced by an acidic environment plays an important role in cell protection.
Materials and methods
Cell culture
Human gastric cancer SGC7901 cells were provided by the Central Laboratory of the Affiliated Hospital of Qingdao University, Qingdao, Shandong Province, China; who obtained the cell line from the Shanghai Institutes for Biological Sciences of Chinese Academy of Sciences. The cells were cultured in RPMI-1640 medium (pH 7.4; Hyclone Laboratories, Logan, UT, USA) supplemented with 10% foetal bovine serum (FBS; Biological Industries, Kibbutz Beit-Haemek, Israel), 100 U/ml penicillin and 100 U/ml streptomycin at 37°C under 95% humidity and 5% CO2. Cells were harvested in the logarithmic growth phase for the experiments described below. Regarding pH regulation, 1 M hydrochloric acid was added to freshly prepared complete medium, and the pH was measured after the addition of each drop until the pH reached 6.8 or 6.5. The pH of the medium was measured before each experiment with a pH Meter (LeiCi, Shanghai, China) to prevent pH changes and so that adjustments could be made if changes were detected.
In order to starve the cells, 24 h after the cells were plated, the medium was replaced with Earle's Balanced Salt Solution (EBSS; Solarbio, Beijing, China). After a 15-min incubation, the cells were harvested and the cell lysate was used in Western blotting as a positive control. For the pH experiments, 24 h after the cells were plated, the medium was replaced with freshly prepared medium at a different pH as described above, then incubated for the indicated time described for each experiment.
Transmission electron microscopy
The SGC7901 cells were grown in media of different pH (pH 7.4 and pH 6.5) for 0.5 h. Cells were harvested and fixed in 2% glutaraldehyde (2.5%; Solarbio) for 24 h at 4°C and cells that remained in suspension were discarded. Next, post-fixed cells were incubated with 1% OsO4 (1%; Sigma-Aldrich, Darmstadt, Germany) for 2 h, dehydrated with graded acetone, and embedded in resin embedding medium. Ultrathin sections were cut on a ultramicrotome and examined with a transmission electron microscope (JEM-1200EX; Jeol, Tokyo, Japan).
Immunofluorescent staining of punctate LC3 structures
The SGC7901 cells were plated onto coverslips in 6-well plates and cultured overnight. The medium was changed to pH 7.4 or pH 6.5 medium, and the cells were cultured for 1 h. The medium was removed, and the coverslips were rinsed with 10 mM phosphate-buffered saline (PBS; pH 7.4) three times. Next, 2 ml of 4% paraformaldehyde was added to the plates, which were incubated at 4°C. After 15 min of incubation, the coverslips were rinsed with 2 ml of 10 mM PBS (pH 7.4) three times. The cells were permeabilized with 2 ml of 0.5% Triton X-100 at room temperature for 20 min. Next, the coverslips were rinsed with 10 mM PBS (pH 7.4) for 3 min at room temperature, and 2 ml of 10 mM PBS (pH 7.4) containing 3% FBS was added to the plates, which were incubated at 37°C for 30 min. The coverslips were incubated with rabbit antihuman autophagy marker light chain 3 (LC3) monoclonal antibody solution (diluted 1:200; Cell Signaling Technology, Danvers, MA, USA) at 4°C overnight. The primary antibody solution was removed, and the coverslips were washed with 10 mM PBS (pH 7.4) three times, followed by incubation with goat antirabbit IgG (H+L), F(ab')2 Fragment (Alexa Fluor® 488 Conjugate) (diluted 1:200, Cell Signaling Technology) at room temperature for 1 h. The coverslips were washed with 10 mM PBS (pH 7.4) three times for 5 min each, followed by incubation in 1 µg/ml 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride solution (Solarbio) for 5 min at room temperature, then the coverslips were rinsed with 10 mM PBS (pH 7.4) three times for 5 min each. Next, the coverslips were observed using laser confocal microscopy (×189 magnification; FV1200, Olympus, Tokyo, Japan).
Western blotting
The SGC7901 cells were treated with either pH 7.4 medium, pH 6.5 medium or EBSS for 8 h for one set of experiments; and with pH 6.5 medium for 0, 4, 8 and 16 h for a second set of experiments. Cells were lysed with RIPA buffer supplemented with the protease inhibitor phenylmethylsulphonyl fluoride (Solarbio) for 30 min on ice and were vortexed every 10 min. Next, the cell lysates were centrifuged at 14 000 g for 5 min at 4°C in a Neofuge 13R centrifuge (Heal Force, Shanghai, China). The supernatant was collected and the protein concentration of the cell extract was determined with a bicinchoninic acid assay protein kit according to the manufacturer’s instructions (CWBIO, Beijing, China). Cell lysates containing 25–30 μg of protein were separated by 15% sodium dodecyl sulphate–polyacrylamide gel electrophoresis using a Mini-PROTEAN® Tetra Vertical Electrophoresis Cell system (Bio-Rad, Hercules, CA, USA California, USA). The proteins were transferred onto polyvinylidene fluoride membranes (Millipore, Billerica, MA, USA). The membranes were incubated for 1 h at room temperature in Tris-buffered saline Tween-20 buffer (TBST; pH 8.6; 20 mM Tris-HCl, 150 mM NaCl and 0.1 % Tween 20) containing 5% skimmed milk (Solarbio). The membranes were then incubated overnight at 4°C with primary antibodies targeting LC3 (1:3000 dilution; rabbit antihuman), p62 (1:3000 dilution; rabbit antihuman) (both from Cell Signaling Technology) or tubulin (1:5000 dilution; rabbit antihuman; Beyotime, Jiangsu, China) in TBST (pH 8.6) containing 5% bovine serum albumin overnight at 4°C. Tubulin was used as the loading control. The membranes were then rinsed with TBST (pH 8.6) three times for 15 min each at room temperature. Next, the appropriate horseradish peroxidase-conjugated secondary antibody (1:1000 dilution; goat antirabbit; Cell Signaling Technology) was incubated for 1 h at room temperature. The membranes were then rinsed with TBST (pH 8.6) three times for 15 min each at room temperature. Bands on immunoblots were visualized with a Millipore Immobilon enhanced chemiluminescence kit (Millipore) and detected using a Vilber FUSION FX image analyser (Vilber Lourmat, Marne-la-Vallée, France). The levels of LC3 and p62 was measured using ImageJ software (version 1.49).
Inhibition of autophagy
Bafilomycin A1, an inhibitor of autophagy, was used to determine the autophagic flux after exposure to acidic stress. At 24 h after plating, the SGC7901 cells were incubated with pH 7.4 medium or pH 6.5 medium supplemented with or without 100 nM bafilomycin A1 (Santa Cruz Biotechnology, Santa Cruz, CA, USA) for 8 h, using EBSS-treated cells as a positive control for cell starvation. The LC3 and p62 levels were evaluated by Western blot analysis as described above and normalized to the pH 7.4 result.
Cell viability assay
Briefly, 0.2 × 104 cells/well were seeded into 96-well plates and incubated in pH 7.4 medium, pH 6.5 medium or pH 6.8 medium in the presence or absence of 100 nM bafilomycin A1 for 0, 24, 48 and 72 h. Then cell proliferation was measured using the Cell Counting Kit-8 (CCK-8; Solarbio) at 0, 24, 48 and 72 h. Briefly, 10 μl of CCK-8 solution was added to each well and the plates were incubated for 1.5 h at 37°C. Next, the optical density was measured at 450 nm using a Multiskan GO microplate spectrophotometer (Thermo Fisher Scientific, Waltham, MA USA).
Statistical analyses
All statistical analyses were performed using the SPSS® statistical package, version 21.0 (SPSS Inc., Chicago, IL, USA) for Windows®. All of the experiments were performed in triplicate and were repeated three times. The data are expressed as the mean ± SD. Parametric data were analysed using unpaired, two-tailed Student’s t-test. A P-value < 0.05 was considered statistically significant.
Results
Because autophagy, or type II nonapoptotic cell death, is characterized by the formation of double-membrane vesicles, this study used TEM and immunofluorescence to examine the effects of an acidic microenvironment on the formation of autophagic vacuoles in SGC7901 cells. As shown in Figure 1, there were more autophagic vacuoles in SGC7901 cells cultured in pH 6.5 medium compared with those cultured in pH 7.4 medium. Immunofluorescence showed that punctate LC3 structures were increased in cells under acidic stress compared with cells cultured under normal conditions (Figure 2). These results demonstrate that acidic stress increases the number of autophagic vacuoles in SGC7901 cells.

Representative transmission electron microscopy images showing that the number of autophagic vacuoles (arrows) in gastric cancer cells increased under acidic stress. SGC7901 cells were cultured overnight and the culture medium was changed to (a) pH 7.4 or (b) pH 6.5. The cells were collected 0.5 h after the medium change. Scale bar in enlarged images, 500 nm.

Distribution of punctate light chain 3 (LC3) structures at different pH values in SGC7901 gastric cancer cells. The cells were cultured in (a) pH 7.4 or (b) pH 6.5 medium for 1 h. The fluorescent punctate LC3 structures were assessed by fluorescence microscopy (magnification × 189). The number of punctate LC3 structures were determined per cell (c). The data are expressed as the mean ± SD of three independent experiments. *P < 0.05 versus cells cultured in pH 7.4 medium; unpaired, two-tailed Student’s t-test. The colour version of this figure is available at: http://imr.sagepub.com.
Western blot analysis was used to assess the effect of acidic stress on LC3-I translocation to the autophagic membrane (LC3-II) and p62 levels. The change in LC3-II and p62 levels was determined after culture in pH 6.5 medium for 8 h (Figures 3a, 3b and 3c). In these experiments, low pH and starvation by incubation in EBSS induced a significant increase in LC3-II levels and a significant decrease in p62 levels compared with pH 7.4 (P < 0.05 for all comparisons). To investigate whether the autophagy level changed over time, the levels of LC3-II and p62 in SGC7901 cells after 0, 4, 8, and 16 h of culture in pH 6.5 medium were measured using Western blot analysis (Figure 3d). A significant increase in LC3-II levels was observed as early as 4 h after the medium change and the levels remained significantly higher at 4, 8 and 16 h compared with 0 h (P < 0.05 for all comparisons) (Figure 3e). The p62 levels started to decrease significantly at 8 h and remained significantly lower at 16 h compared with 0 h (P < 0.05 for both comparisons), although there was no significant difference between 0 h and 4 h (Figure 3f).
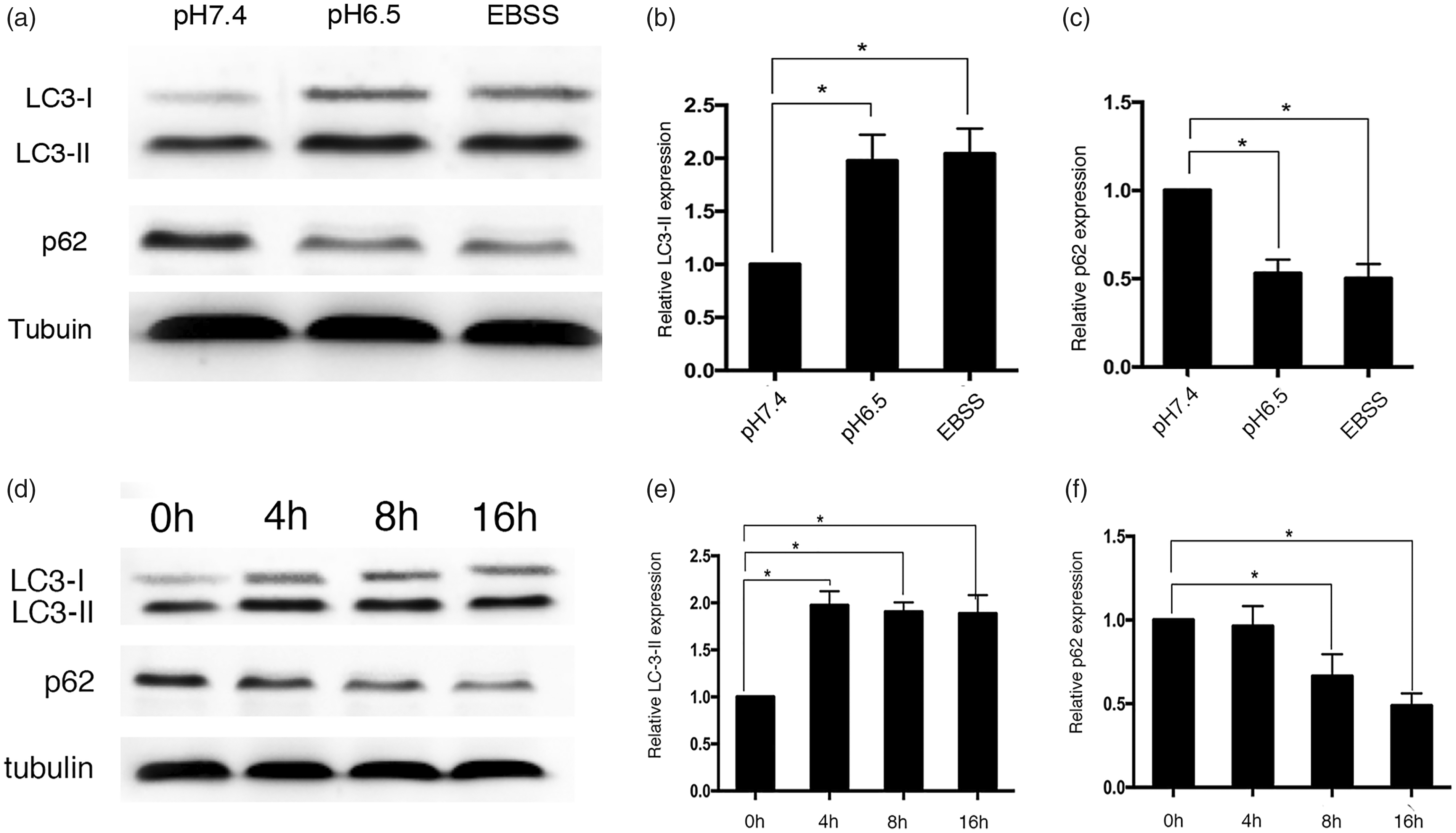
Effect of acidic stress on light chain 3 (LC3-I and LC3-II) and p62 levels in SGC7901 gastric cancer cells. (a–c) SGC7901 cells were exposed to pH 7.4 medium, pH 6.5 medium or Earle's Balanced Salt Solution (EBSS) for 8 h. The LC3 and p62 levels were evaluated by Western blot analysis and normalized to the pH 7.4 result. Tubulin was used as a loading control. (d–f) SGC7901 cells were plated overnight and the culture medium was changed to pH 6.5. Cells were collected at 0, 4, 8, and 16 h after medium change. The LC3 and p62 levels were evaluated by Western blot analysis and normalized to the value at time 0. Tubulin was used as a loading control. The data are expressed as the mean ± SD of three independent experiments. * P < 0.05; unpaired, two-tailed Student’s t-test.
To analyse the autophagic flux status, SGC7901 cells were cultured at pH 7.4 and pH 6.5 in the presence or absence of the lysosomal inhibitor bafilomycin A1 for 8 h, using EBSS-treated cells as a positive control. SGC7901 cells cultured in pH 7.4 medium, pH 6.5 medium and EBSS were equally sensitive to the effects of bafilomycin A1 (Figure 4), with the levels of LC3-II increasing in the presence of bafilomycin A1 under both pH conditions. Similarly, increased levels of p62 were observed following treatment with bafilomycin A1 in cells under acidic stress. These findings support the notion that autophagy flux increases under acidic stress.

Effect of acidic stress on autophagic flux in SGC7901 gastric cancer cells. Analysis of autophagic flux in SGC7901 cells was evaluated using the levels of light chain 3 (LC3-I and LC3-II) and p62 in cells cultured in pH 7.4 medium or pH 6.5 medium in the presence or absence of bafilomycin A1 (Baf A1; 100 nM) for 8 h, using Earle's Balanced Salt Solution (EBSS)-treated cells as a positive control. (a–c) The LC3 and p62 levels were evaluated by Western blot analysis and normalized to the positive control result. Tubulin was used as a loading control. Tubulin was used as a loading control. The data are expressed as the mean ± SD of three independent experiments. *P < 0.05; unpaired, two-tailed Student’s t-test.
Autophagy can be a prosurvival or a prodeath mechanism under different circumstances. To evaluate the effect of autophagy under an acidic environment on cell proliferation, the CCK-8 assay was performed. Low pH (pH 6.5) reduced cell viability and cell viability was lower in pH 6.5 medium than in pH 6.8 medium (Figure 5). Cell proliferation was additionally decreased after inhibiting autophagy at pH 6.5 and pH 6.8 and the same phenomenon was observed at pH 7.4.
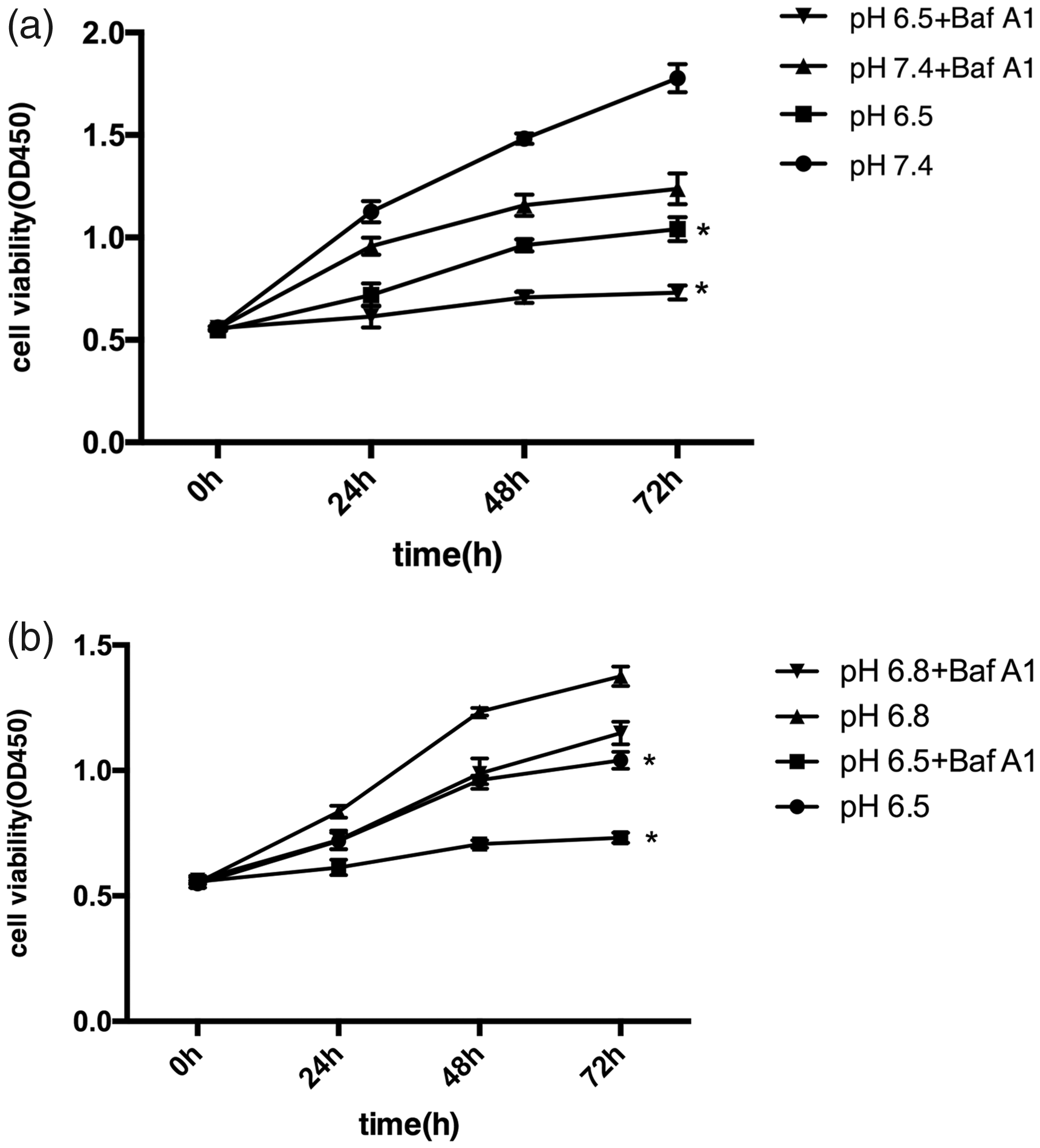
Effect of inhibition of autophagy in an acidic microenvironment on the proliferation of SGC7901 cells which were grown in pH 7.4 medium (a), pH 6.5 medium (a) or pH 6.8 medium (b), in the presence or absence of bafilomycin A1 (Baf A1; 100 nM) for 0, 24, 48 and 72 h. *P < 0.05; unpaired, two-tailed Student’s t-test. The data are expressed as the mean ± SD of three independent experiments.
Discussion
This present study investigated whether autophagy has a protective effect under acidic stress in human gastric cancer SGC7901 cells. After exposure to a pH 6.5 environment, autophagic vacuoles and fluorescent punctate LC3 structures significantly increased. LC3-II levels increased after culture in pH 6.5 medium and was further increased after bafilomycin A1 treatment. The p62 level decreased under acidic stress and increased after bafilomycin A1 treatment. These data suggest that acidic stress increases LC3-II levels and decreases p62 levels in SGC7901 cells. Collectively, our results demonstrate that human gastric cancer SGC7901 cells upregulate autophagy as a protective mechanism under acidic stress.
Upregulated autophagosome formation under acidic conditions was demonstrated in two ways. First, as shown in Figure 1, autophagosomes were more abundant in cells grown in pH 6.5 medium than in cells cultured in pH 7.5 medium. Secondly, as shown in Figure 2, LC3 fluorescence had a punctate distribution. The results from Western blotting indicated that LC3-II levels were elevated and p62 levels were decreased under acidic stress, providing evidence of increased autophagy. Increased p62 and additional LC3 accumulating in cells treated with bafilomycin A1 also supports the conclusion that autophagy flux is upregulated under acidic stress. Notably, no significant changes in p62 levels were observed after 4 h of culture, possibly indicating that p62 had shuttled ubiquitinated proteins to autophagosomes but had not yet begun to degrade during the initial stages of autophagy. These data suggest that acidic stress increases LC3-II levels and decreases p62 levels in SGC7901 cells. The CCK-8 assay showed that blocking autophagy at pH 6.5 with bafilomycin A1 further inhibits the proliferation of SGC7901 cells, suggesting that this type of autophagy is protective. The current data also suggest that autophagy functions as a protective mechanism at pH 7.5, pH 6.8 and pH 6.5.
Several studies in related fields have clearly demonstrated that autophagy plays an important role in allowing breast cancer cell lines and lung cancer cell lines to adapt to acidic environments.11,20 Likewise, this present study demonstrated the autophagy flux in four experiments and the results confirmed that the autophagy flux was higher in the pH 6.5 group than in the pH 7.4 group. These findings suggest that autophagy flux increases under acidic stress. The current finding that autophagy has a protective effect under an acidic environment also agrees with the findings of a study on melanoma. 10
The relationship between autophagy and tumour malignancy is still not fully understood. Autophagy can play different roles depending on the microenvironment, stage, tumour type and other factors. For example, in the early stages of tumorigenesis, autophagy mainly inhibits the occurrence of malignant tumours.12,21 In already formed tumours, autophagy can be protective or deleterious.17,22,23 Studies have shown that autophagy is increased mainly under metabolic stress conditions to reduce cell damage and maintain the integrity of cells. 24 Therefore, if autophagy is used as a target for tumour therapy, it will be necessary to determine the influence of autophagy on the biological behaviour of tumour cells under these conditions. This current study determined that autophagy under acidic stress is a protective mechanism for the gastric cancer cell line SGC7901. It is likely that this phenomenon exists in other gastric cancer cell lines.
This current study had several limitations. First, only the gastric cancer cell line SGC7901 was used; thus, it is unknown whether these results are applicable to other gastric cancer cell lines. Further studies with additional cell lines are needed. Secondly, this study focused on in vitro cell experiments, so the results might not reflect these processes in an intact organism. Studies are currently underway to confirm these findings in mice. Thirdly, although the CCK-8 assay was used to confirm that autophagy under acidic environments is protective, changes to the cell cycle distribution remain to be determined. Flow cytometry assays will be used to investigate cell cycle pattern changes.
The next step is to investigate autophagic changes in other gastric cancer cell lines such as BGC-823, MKN-45, MGC-803, HGC-27 and AGS under acidic stress. Additional studies are needed to identify the signalling pathways involved in acidic stress-related autophagic upregulation. These current findings have provided questions that should be explored in vivo. Research is already underway to investigate the changes in apoptosis, cell cycle, migration and invasion under stress with and without autophagic inhibition in gastric cancer cell lines.
In conclusion, autophagy may be an important survival mechanism for SGC7901 cells under acidic stress. As shown in this current study, blocking autophagy inhibited proliferation in the human gastric cancer cell line SGC7901. A better understanding of the precise function of autophagy in the gastric cancer cell line SGC7901 is needed.
Footnotes
Declaration of conflicting interests
All authors declare that there are no conflicts of interest.
Funding
This study was supported by the National Natural Science Foundation of China (no. 81472338).
