Abstract
Objective
To determine whether pre-treatment with high-dose ulinastatin provides enhanced postoperative oxygenation in patients who have undergone aortic valve surgery with moderate hypothermic cardiopulmonary bypass (CPB).
Methods
Patients who underwent aortic valve surgery with moderate hypothermic CPB were retrospectively evaluated. In total, 94 of 146 patients were included. The patients were classified into one of two groups: patients in whom ulinastatin (10,000 U/kg followed by 5,000 U/kg/h) was administered during CPB (Group U, n = 38) and patients in whom ulinastatin was not administered (Group C, n = 56). The PaO2/FiO2 ratio was calculated at the following time points: before CPB (pre-CPB), 2 h after weaning from CPB (post-CPB), and 6 h after arrival to the intensive care unit (ICU-6). The incidence of a low PaO2/FiO2 ratio was also compared among the time points.
Results
Group U showed a significantly higher PaO2/FiO2 ratio (F(4, 89.0) = 657.339) and a lower incidence of lung injury (PaO2/FiO2 ratio < 300) than Group C at the post-CPB and ICU-6 time points.
Conclusion
High-dose ulinastatin improved pulmonary oxygenation after CPB and in the early stages of the ICU stay in patients undergoing aortic valve surgery with CPB.
Keywords
Introduction
Cardiopulmonary bypass (CPB) induces systemic inflammatory responses and postoperative organ injury, which increase perioperative mortality and morbidity.1–4 Several studies have reported that ulinastatin has anti-inflammatory and organ-protective effects in animals and humans.5–9 Ulinastatin attenuates the increase in systemic pro-inflammatory cytokines, enhances release of anti-inflammatory cytokines, and exerts beneficial effects on major organs including the heart, lungs, and kidneys.9–11 Ulinastatin also reduces the duration of postoperative mechanical ventilation in patients who undergo cardiac surgery with CPB.11,12 However, the effects of ulinastatin on CPB-induced lung injury and the postoperative oxygenation profile have not been evaluated.
Because ulinastatin has a short biphasic plasma half-life of 33 min and 2 h, its dosage and duration of infusion must be increased to cover the entire CPB period and thus attenuate CPB-induced organ injury.13,14 We hypothesised that high-dose ulinastatin infusion during the CPB period would attenuate CPB-induced lung injury and improve postoperative pulmonary oxygenation. The present study was performed to test this hypothesis.
Patients and methods
Study population
This single-centre retrospective study was approved by the institutional review board of Konkuk University Medical Centre (KUH 1160051). We reviewed the medical records of patients who had undergone aortic valve repair or replacement under moderate hypothermic CPB from January 2012 to January 2015. Patient consent was not required because of the retrospective nature of the study.
The preoperative exclusion criteria were an age of <19 or >65 years, combined operation, emergency operation, re-do or tri-do operation, left ventricular ejection fraction of <50%, arrhythmia, ischaemic heart disease, pre-existing renal or hepatic dysfunction, and underlying lung disease. The intraoperative exclusion criterion was intraoperative application of an intra-aortic balloon pump. The postoperative exclusion criterion was reoperation to correct intractable postoperative bleeding.
The patients were classified into two groups. Group C comprised patients in whom ulinastatin was not administered. Group U comprised patients in whom 10,000 U/kg of ulinastatin was administered intravenously as a loading dose 30 min before CPB and 5,000 U/kg/h of ulinastatin was infused during the CPB period (Figure 1).

Patient recruitment scheme. Twenty-six patients had more than two exclusion criteria. Group C: patients not administered ulinastatin, Group U: patients administered ulinastatin during cardiopulmonary bypass, op: operation, EF: left ventricular ejection fraction in preoperative transthoracic echocardiography, postop: postoperative.
Anaesthesia and surgery
Non-invasive monitoring was started upon the patient’s arrival in the operating room. Invasive arterial blood pressure monitoring was initiated before induction. Target-controlled infusions of propofol and remifentanil and a bolus and infusion of rocuronium were used. Mechanical ventilation was provided after tracheal intubation: an O2/air mixture (FiO2 of 0.5) of 4 L/min was applied with a tidal volume of 6 mL/kg and positive end-expiratory pressure (PEEP) of 7 to 8 mmHg, and the respiration rate was maintained at an end-tidal CO2 tension of 35 to 40 mmHg. After inducing anaesthesia, central venous and pulmonary arterial catheters were placed and the central venous pressure (CVP), pulmonary arterial pressure (PAP), mixed venous oxygen saturation (SvO2), and cardiac index (CI) were monitored continuously. A two- or three-dimensional trans-oesophageal echocardiographic examination was performed in accordance with the guidelines of the American Society of Echocardiography/Society of Cardiovascular Anaesthesiologists.
The surgical procedure was performed under CPB with moderate hypothermia (28°C–29°C). The pump was primed with 1500 to 1700 mL of acetate-buffered balanced crystalloid, 100 mL of 20% mannitol, and 100 mL of 20% albumin. Anticoagulation for CPB was initiated with 3 U/kg of heparin and monitored with an activated coagulation time of >400 s. After cross-clamping the aorta, cold crystalloid cardioplegia solution was administered. CPB was conducted with a non-pulsatile roller pump, polyvinyl chloride tubing, and a membrane oxygenator with hollow polypropylene fibres. The pump flow rate was adjusted to 2.0 to 2.5 L/min/m2 to maintain a mean arterial blood pressure (MBP) of 60 to 70 mmHg. During CPB, continuous positive airway pressure of 5 mmHg was applied. Before weaning from CPB, modified ultrafiltration was applied with a flow rate of 200 mL/min/m2 in all patients. Intermittent antegrade and retrograde cold crystalloid cardioplegia was administered every 30 min, and continuous topical cooling was applied during cardiac standstill. PaCO2 was managed by α-stat during CPB.
CPB was weaned with or without inotropic support when the rectal temperature reached 36°C. Intraoperative fluid management was performed upon trans-oesophageal echocardiography monitoring. Dopamine was infused as a first-line inotropic agent to wean the patients from CPB as needed, and its infusion rate was adjusted to achieve a CI of >2.0 L/min/m2 and MBP of >65 mmHg. The ventilation strategy applied during pre-CPB was restored. Blood in the CPB circuit and reservoir was given back to the patients before weaning from CPB. The threshold for transfusion of packed red blood cells (pRBCs) was a haematocrit of <21% during CPB and a haematocrit of <28% after CPB. Transfusion of fresh frozen plasma, cryoprecipitate, or platelets was performed upon the results of rotational thromboelastometry and routine laboratory coagulation-related tests. Intraoperative bleeding from the surgical site was salvaged and re-infused using a cell saver until closure of the chest wall.
Postoperative care
All patients were transferred to the intensive care unit (ICU) after surgery, where they underwent management according to institutional protocols. Volume–pressure-controlled ventilation was applied with a tidal volume of 6 mL/kg, respiratory rate of 10 to 18/min, PEEP of 5 to 8 mmHg, and FiO2 of 60% Pressure support ventilation with a 10-cmH2O pressure support level and 5-cmH2O PEEP was performed upon recovery of spontaneous respiration.
The extubation criteria were a respiratory rate of 11 to 25 breaths/min, heart rate within 20% of baseline for at least for 1 h, PaO2 of >70 mmHg, PaCO2 of <48 mmHg, and pH of 7.35 to 7.50 on an arterial blood gas analysis.
Data acquisition
The primary objective was an intergroup comparison of the PaO2/FiO2 ratio 2 h after weaning from CPB (post-CPB). We also compared the PaO2/FiO2 ratio and its changes at the following time points: before CPB (pre-CPB), post-CPB, and 6 h after arrival in the ICU (ICU-6). The incidence rates of a low PaO2/FiO2 ratio (<200 and <300) were also compared. Haemodynamic variables including the heart rate, MBP, CVP, PAP, SvO2, and CI were compared at these same time points.
The creatinine kinase-MB (CK-MB) concentration, high-sensitivity troponin-I (Tn-I) concentration, serum creatinine (s-Cr) concentration, glomerular filtration rate (GFR), and C-reactive protein (CRP) concentration were measured preoperatively, immediately after ICU arrival (ICU-0), and 12 h after ICU arrival (ICU-12).
Statistical analyses
We used an intention-to-treat strategy; that is, all patients were included in the analysis regardless of whether they had completed the study. Because some patients had missing data on the outcome variables, the missing data were completed using a last observation carried forward analysis. The distribution of continuous variables was first evaluated for normality using the Shapiro–Wilk test and a q-q plot. Normally distributed data are presented as mean ± standard deviation, and the groups were compared using parametric methods. The non-normally distributed data are expressed as median (interquartile range), and these data were analysed via non-parametric methods.
Because the patients’ age, height, CI, SVO2, and PaO2/FiO2 ratio passed the Shapiro–Wilk test, they were analysed parametrically. Weight, CPB duration, and number of transfused blood units did not pass the Shapiro–Wilk test; these data were therefore analysed non-parametrically. Because MBP, CVP, and PAP did not pass the Shapiro–Wilk test, we additionally checked the q-q plot, which did not show marked deviation from linearity. Therefore, the normal assumptions were applied for repeated-measures analysis of variance (RM-ANOVA). Because CI and SvO2 passed Mauchly’s sphericity test, they were analysed by RM-ANOVA with Greenhouse–Geisser correction followed by the paired t-test and Bonferroni correction (α = 0.05/3 and 0.0167). Because Mauchly’s sphericity test indicated that the assumption of sphericity had been violated for MBP [χ2(2) = 59.206, P < 0.001, Mauchly’s W = 0.518], CVP [χ2(2) = 77.383, P < 0.001, Mauchly’s W = 0.423], PAP [χ2(2) = 29.544, P < 0.001, Mauchly’s W = 0.720], and the PaO2/FiO2 ratio [χ2(5) = 16.012, P = 0.007, Mauchly’s W = 0.838], Wilks’ lambda multivariate ANOVA (MANOVA) followed by the paired t-test with Bonferroni correction (α = 0.05/3 and 0.0167 or α = 0.05/4 and 0.0125) was used.
Because s-Cr, GFR, CK-MB, Tn-I, and CRP did not pass the Shapiro–Wilk test and showed marked deviation from linearity, the data were analysed using Friedman’s test followed by the Mann–Whitney test with Bonferroni correction.
Descriptive variables were subjected to χ2 analysis or Fisher’s exact test, as appropriate, and P-values of <0.05 were considered significant. Statistical analysis was performed using SPSS software (ver. 23.0; IBM Corp., Armonk, NY, USA).
Results
The medical records of 146 patients who underwent aortic valve surgery with moderate hypothermic CPB were screened, and 52 patients were excluded. Finally, 94 patients were classified into the 2 study groups (Group C, n = 56 and Group U, n = 38). The demographic data of the 94 patients are shown in Table 1. The preoperative variables were not different between the groups. The surgical procedure performed was either aortic valve leaflet repair (AVP) with/without aortic root remodelling or aortic valve replacement (AVR). There were no inter-group differences in the type of surgery: 45 of 56 patients in Group C and 32 of 38 patients in Group U underwent AVP, and the remaining patients underwent AVR.
Preoperative patient characteristics

Data are presented as mean ± standard deviation, median (25%–75%), or number of patients (%), as appropriate. Group C: patients not administered ulinastatin, Group U: patients administered ulinastatin during cardiopulmonary bypass, CK-MB: serum creatinine kinase subtype MB (reference range: 0.0–6.6 ng/mL), Tn-I: high-sensitivity cardiac troponin I (reference range: 0.0–0.3 ng/mL), s-Cr: serum creatinine (reference range: 0.5–0.9 mg/dL).
The CPB duration, aortic cross-clamping time, and number of intraoperative transfused units were not different between the groups. The volumes of transfused pRBCs and fresh frozen plasma in the ICU were greater in Group U than Group C (Table 2).
Cardiopulmonary bypass duration and units of transfusion

Data are presented as median (25%–75%). Group C: patients not administered ulinastatin, Group U: patients administered ulinastatin during cardiopulmonary bypass, CPB duration: duration of application of cardiopulmonary bypass, CPBmin: minimum value during cardiopulmonary bypass, CPB: cardiopulmonary bypass, post-CPB: after weaning from cardiopulmonary bypass, ICU: intensive care unit, pRBCs: packed red blood cells, FFP: fresh frozen plasma.
Table 3 shows that there were no differences in the haemodynamic data between the two groups (MBP: [F(3, 89.0) = 0.521: Wilks’ lambda = 0.983, partial eta 2 = 0.017], CVP: [F(3, 89.0) = 0.022: Wilks’ lambda = 0.999, partial eta 2 = 0.001], and PAP: [F(3, 89.0) = 0.403: Wilks’ lambda = 0.987, partial eta 2 = 0.013]).
Haemodynamic parameters

Data are presented as mean ± standard deviation. Group C: patients not administered ulinastatin, Group U: patients administered ulinastatin during cardiopulmonary bypass, pre-CPB: before cardiopulmonary bypass, post-CPB: 2 h after weaning from cardiopulmonary bypass, ICU-6: within 6 h after admission to the intensive care unit, MBP: mean arterial blood pressure, CVP: central venous pressure, PAP: mean pulmonary artery pressure, CI: cardiac index, SvO2: mixed venous oxygen saturation. P-values represent overall inter-group differences in repeated-measures analysis of variance or multivariate analysis of variance, as appropriate.
The PaO2/FiO2 ratio did not differ between the groups at the pre-CPB time point. However, the PaO2/FiO2 ratio was higher in Group U than Group C at post-CPB and ICU-6 (P < 0.05). The MANOVA results showed a difference in the change in the PaO2/FiO2 ratio between two groups [F(4, 89.0) = 657.339, P < 0.001: Wilks’ lambda = 0.033, partial eta 2 = 0.967]. The incidence of a PaO2/FiO2 ratio of <300 was lower in Group U than Group C at the same time point (Table 4).
Changes in PaO2/FiO2 ratio and incidence of low PaO2/FiO2 ratio

Data are presented as mean ± standard deviation or number of patients (%). Group C: patients not administered ulinastatin, Group U: patients administered ulinastatin during cardiopulmonary bypass, pre-CPB: before cardiopulmonary bypass, post-CPB: 2 h after weaning from cardiopulmonary bypass, ICU-6: within 6 h after admission to the intensive care unit. P-values of PaO2/FiO2 ratio represent overall inter-group differences in repeated-measures analysis of variance or multivariate analysis of variance, as appropriate. *P < 0.05 compared with Group C. P-values of incidence of low PaO2/FiO2 ratio represent overall inter-group differences in χ2 analysis or Fisher’s exact test, as appropriate.
The length of ventilatory support was not different between Group C and Group U (16 (13–18) vs. 15.5 (11.5–19.5) h, respectively).
CK-MB, Tn-I, s-Cr, GFR, and CRP did not differ between the groups (Table 5).
Changes in cardiac enzymes, indices of renal performance, and CRP
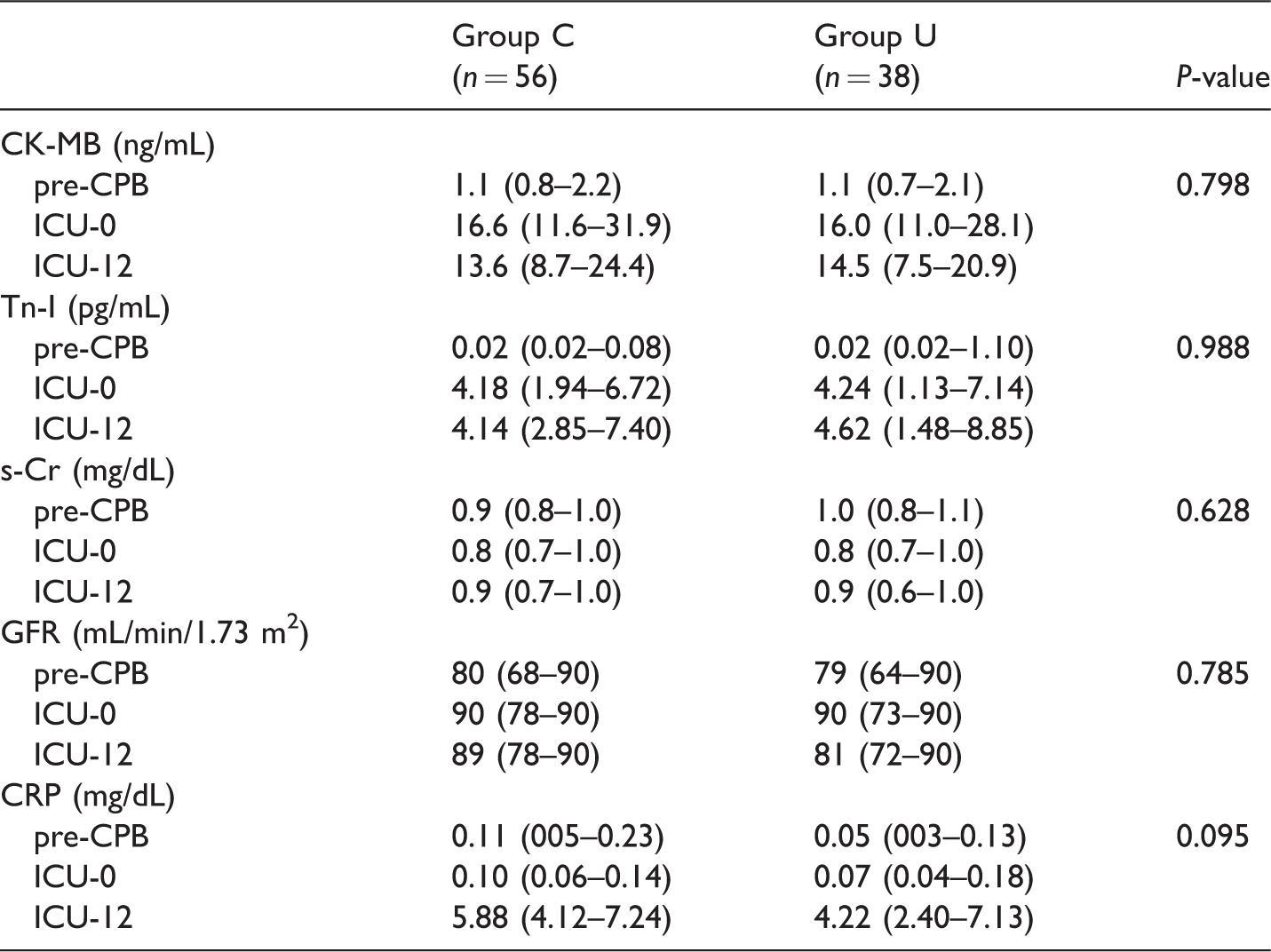
Data are presented as median (25%–75%). Group C: patients not administered ulinastatin-treatment, Group U: patients administered ulinastatin during cardiopulmonary bypass, CK-MB: serum creatinine kinase subtype MB (reference range: 0.0–6.6 ng/mL), Tn-I: high-sensitivity cardiac troponin-I (reference range: 0.0–0.3 ng/mL), s-Cr: serum creatinine (reference range: 0.5–0.9 mg/dL), GFR: glomerular filtration rate (reference value: >90 mL/min/1.73 m2), CRP: high-sensitivity C-reactive protein (reference range: 0.01–0.3 mg/dL), pre-CPB: before applying cardiopulmonary bypass, ICU-0: immediately after admission to the intensive care unit, ICU-12: 12 h after admission to the intensive care unit. P-values represent overall inter-group differences in repeated-measures analysis of variance or multivariate analysis of variance, as appropriate.
Discussion
In the present study, high-dose ulinastatin infusion during CPB provided a higher PaO2/FiO2 ratio after weaning from CPB and in the early stage in the ICU. The higher PaO2/FiO2 ratio in ulinastatin-treated patients may suggest a reduction in postoperative morbidity and mortality, as shown in other studies.15–17 In previous studies, ulinastatin attenuated ischaemia–reperfusion-induced lung injury by inhibition of myeloperoxidase activity in lung tissue and a decrease in the systemic expression of tumour necrosis factor-α in a rat model and attenuated the release of interleukin-6 and -8 immediately after CPB. The degree of changes in the respiratory index and intrapulmonary shunt was correlated with the interleukin-8 level.6,9 However, these inflammatory cytokines were not analysed in the present study because they are not included as routine laboratory tests for patients undergoing cardiac surgery in our institution.
The results of the present study did not show improved clinical outcomes, such as shortening of the mechanical ventilation time, intubation time, or ICU stay. Several studies have revealed shortened durations of mechanical ventilation, intubation, and ICU stay in patients treated with ulinastatin. Zhang et al. 11 concluded that ulinastatin treatment might shorten the duration of mechanical ventilation as well as the duration of intubation, but the ICU stay was not shortened. He et al. 12 also found that the use of ulinastatin reduced the duration of mechanical ventilation, but the length of ICU stay was not different. Song et al. 18 found that patients treated with ulinastatin had shorter ICU stays than the control group. Xu et al. 10 also concluded that high-dose ulinastatin shortened the ICU stay after application of CPB with deep hypothermic circulatory arrest.
We used high-dose ulinastatin during the entire CPB period (total dose of 20,000 U/kg with a 2-h CPB period and 25,000 U/kg with a 3-h CPB period). Previous studies also recommended a higher dose of ulinastatin (5,000–20,000 U/kg) to overcome the dose-dependent heterogeneity in terms of exerting organ-protective effects.10,11
Despite the improved oxygenation profile, which is probably due to ulinastatin’s attenuation of CPB-induced acute lung injury, we found no positive improvement in ultimate outcomes such as shortening of the duration of intubation and ventilator support. These findings are not in accordance with those of Esteve et al., 19 who reported that the PaO2/FiO2 ratio 3 h after ICU admission may be useful to predict the outcome of cardiac surgical patients during their hospital stay. We may consider two major factors. First, because the duration of action of ulinastatin is much shorter than the length of ventilatory support, ulinastatin cannot attenuate the inflammatory stress that occurs after CPB. Second, transfusion-related acute lung injury could aggravate postoperative pulmonary function. The volume of transfused pRBCs in the ICU was greater in Group U than in Group C in this study (5 vs. 2 U, respectively). This discrepancy suggests ulinastatin's additional efficacy for attenuating transfusion-related acute lung injury in cardiac surgery. 18
In this study, we identified no clinical outcomes suggesting the beneficial effects of ulinastatin in attenuating CPB-induced myocardial or kidney injury. The absence of inter-group differences in myocardiac biomarkers, such as Tn-I and CK-MB, contradict the findings of a previous report showing significant reductions in the levels of cardiac markers and blood pressure in patients undergoing ulinastatin treatment, suggesting ulinastatin’s protective effects against myocardial injury. 11 The routine use of ultrafiltration at the end of CPB to remove free water and improve post-CPB pulmonary function might affect the level of myocardial biomarkers.20,21 The s-Cr concentration and GFR showed no inter-group differences in our study. A few studies have shown that ulinastatin may reduce the risk of acute kidney injury after CPB, but this finding is controversial because of the absence of a difference in the s-Cr or cystatin C concentration after aortic valve surgery with CPB.22,23
This study has some limitations. First, because this study involved a retrospective analysis of patients’ medical records, no analyses of inflammatory cytokines were included; however, such analyses might have supported ulinastatin’s efficacy in attenuating anti-inflammatory cascades and organ protection. 24 Instead, CRP was analysed, indicating the degree of the anti-inflammatory effects of neutrophil elastase inhibitors,25,26 and showed no significant inter-group differences in our study. Second, we analysed the PaO2/FiO2 ratio, which is inferior to the alveolar-arterial O2 tension difference, P(A-a)O2, or ratio of P(A-a)O2 to PaO2 in the early diagnosis of acute lung injury. 8 Third, our study’s primary outcome was confined to the immediate post-CPB period; other clinical outcomes beyond the immediate postoperative period were not analysed. Finally, because the study was performed in a retrospective manner, it was not possible to standardise or minimise possible biases that could have affected the outcome, and the data of only 94 patients were included in the final analyses. However, the type of surgery was confined to aortic valvular surgery, so the patients had uniform clinical courses.
In conclusion, ulinastatin treatment during the CPB period provided a better oxygenation profile after CPB and in the early stages in the ICU in patients undergoing aortic valve surgery with moderate hypothermic CPB.
Footnotes
Acknowledgement
Authors’ contributions
Research conception and design: KYR, TYK. Data analysis and interpretation: KYR, TYS, JDK, NM. Statistical analysis: HK. Drafting of the manuscript: KYR, HK. Critical revision of the manuscript: TYK. Approval of final manuscript: all authors.
Disclosure
The preliminary study described in this manuscript was presented as an abstract at the 42nd Annual Meeting of the Japanese Society of Intensive Care Medicine.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper was supported by Konkuk University. This study was supported by Konkuk University Medical Centre.
