Abstract
Nonfunctional pancreatic neuroendocrine tumors (NF-pNETs) in patients with multiple endocrine neoplasia type 1 (MEN1), which results from a mutation in the
Introduction
Multiple endocrine neoplasia type 1 (MEN1) is a rare autosomal dominant inherited disease that is caused by mutations in the
Pancreatic neuroendocrine tumors (pNETs) represent the second most common manifestation of MEN1; in one study, 56.1% of patients with MEN1 had pNETs. 2 pNETs arising from pancreatic islet cells in patients with MEN1 can be functional or nonfunctional. 3 All pNETs in patients with MEN1 occur exclusively within the pancreas except for gastrinomas and rare somatostatinomas. 4 Among MEN1-related pNETs, nonfunctional pNETs (NF-pNETs) are the most frequent type of tumors (70%). 5 When compared with functional tumors, NF-pNETs are usually malignant and the most common cause of death. 6 Metastatic disease is more prevalent in the liver than in the surrounding lymph nodes in patients with larger MEN1-related NF-pNETs. 7 MEN1-related NF-pNETs are reportedly more commonly located in the pancreatic head, 8 and fewer are present in the pancreatic tail; 9 they are usually small and multiple and are considered slow-growing tumors. 10 Detection of pancreatic tumors and measurement of their size and location constitute important elements for the management of patients with MEN1. Upon clinical suspicion of pNET, imaging studies such ultrasound (US), computed tomography (CT), and magnetic resonance imaging (MRI) must be performed. If pNETs are confirmed, contrast-enhanced CT should be routinely performed to clarify the anatomy and possible presence of liver metastases before surgery. 11
Although MEN1 is an autosomal dominant inherited disorder, approximately 10% of patients have
NF-pNETs, which are located more commonly in the head of the pancreas and inside the pancreatic parenchyma, are usually small and multiple. We herein describe a Chinese patient with MEN1 who developed a single non-metastatic NF-pNET in the peripancreatic region resulting from a missense mutation in exon 2. The reporting of this rare case will make a significant contribution to the literature.
Clinical report
This study was approved by the ethics committee of Tianjin Medical University General Hospital (approval no. 201304). Verbal informed consent for publication was obtained from the patient.
A 35-year-old Chinese woman was admitted to the Endocrinology Department of the Tianjin Medical University General Hospital (Tianjin, China) in 2010 with a 7-year history of amenorrhea and galactorrhea and a 6-month history of bone pain. The patient had clinical evidence of a prolactinoma with symptoms of amenorrhea and signs of galactorrhea. Her serum prolactin (PRL) concentration was >200 ng/mL, and pituitary MRI showed findings consistent with a pituitary microadenoma. She was clinically diagnosed with a prolactinoma and treated with bromocriptine. Her menstruation resumed and her serum PRL concentration gradually returned to normal. Six months previously, she had developed bone pain, and hypercalcemia had been detected at that time. She had no family history of parathyroid or pituitary disorders. Her medical history included a diagnosis of type 2 diabetes mellitus and chronic hepatitis B, which was treated with adefovir dipivoxil. The physical examination findings were normal except for rib tenderness and “o” type bowed legs.
The patient had evidence of primary hyperparathyroidism with hypercalcemia (3.12 mmol/L; reference range, 2.15–2.55 mmol/L), hypophosphatemia (0.59 mmol/L; reference range, 0.80–1.45 mmol/L), and an inappropriately high parathyroid hormone (PTH) concentration of 227 pmol/L (reference range, 1.1–7.3 pmol/L). A parathyroid CT scan (Figure 1(a)) and contrast-enhanced CT scan (Figure 1(b)) both showed a left inferior parathyroid mass, while 99mTc-MIBI scintigraphy showed increased uptake in this parathyroid area (Figure 1(c)). The patient underwent parathyroidectomy of the left inferior parathyroid gland. During surgery, bilateral neck exploration was conducted and three parathyroid glands were found. An enlarged right upper parathyroid gland was also found, while the left upper parathyroid gland was normal. The serum PTH concentration decreased slightly (107 pmol/L) after resection of the left inferior parathyroid gland. Following resection of the right upper parathyroid gland, it further decreased to 8.1 pmol/L, representing a >50% decrease compared with baseline, with symptomatic hypocalcaemia (1.88 mmol/L). Therefore, the left upper parathyroid gland was preserved. The size of the left inferior and right upper parathyroid masses was 2.0 × 1.0 cm (Figure 1(d)) and 1.2 × 0.6 cm, respectively (Figure 1(e)). Histological examination revealed hyperplasia of the parathyroid glands (Figure 1(f), (g)). The postoperative hypocalcaemia was treated with calcitriol and calcium carbonate until the serum calcium level returned to the reference range after 1 year.

(a) Computed tomography scan and (b) contrast-enhanced computed tomography scan revealed a left inferior parathyroid mass. (c) Parathyroid 99mTc-MIBI scintigraphy revealed intense tracer uptake in a lesion of this parathyroid region. The size of the (d) left inferior and (e) right upper parathyroid glands was 2 × 1 cm and 1.2 × 0.6 cm, respectively. Pathological examination of the left inferior parathyroid gland showed (f) fat cells scattered in the hyperplastic parathyroid tissue (hematoxylin and eosin, 100×) and (g) the hyperplastic parathyroid cells were arranged in a prominent nesting pattern (hematoxylin and eosin, 100×).
MEN1 is clinically diagnosed upon confirmation of a prolactinoma and parathyroid adenomatous hyperplasia. Abnormalities in other endocrine glands or cells are sought for further confirmation. The patient’s laboratory findings are summarized in Table 1; the data indicated that no other endocrine glands or cells were involved. An abdominal CT scan (5-mm slice thickness) revealed a large mass of unknown origin, and its density was the same as that of soft tissue, close to the splenic hilum (Figure 2(a)); the adrenal gland and liver were normal. The abdominal CT scan also revealed splenomegaly. A contrast-enhanced CT scan (5-mm slice thickness) showed that the density of the mass was continuously uneven with significant enhancement. Additionally, multiple spots with unenhanced low-density areas were present (Figure 2(b)). In December 2010, the mass was resected. It was round in shape (5-cm diameter) and connected to the pancreatic tail (Figure 2(c)). A clinical diagnosis of NF-PET was made. Surgery is recommended for NF-PETs of ≥20 mm in diameter,13,14 and >5-mm tumors in the pancreas should be resected. 14 The mass was resected with partial resection of the pancreas tail as well as the spleen, in consideration of the splenomegaly. Intraoperative US revealed no synchronous pancreatic tumor. Microscopic examination confirmed the diagnosis of a well-differentiated pNET (Figure 2(d), (e)) with uncertain behavior (Ki-67, 4%). The tumor was immunohistochemically positive for chromogranin A (Figure 2(f)), synaptophysin, and somatostatin and negative for insulin (Figure 2(g)), cytokeratin 7, adrenocorticotropic hormone, and glucagon. Histological examination confirmed no metastases to the pancreatic tail or vascular system.
Patient’s laboratory findings
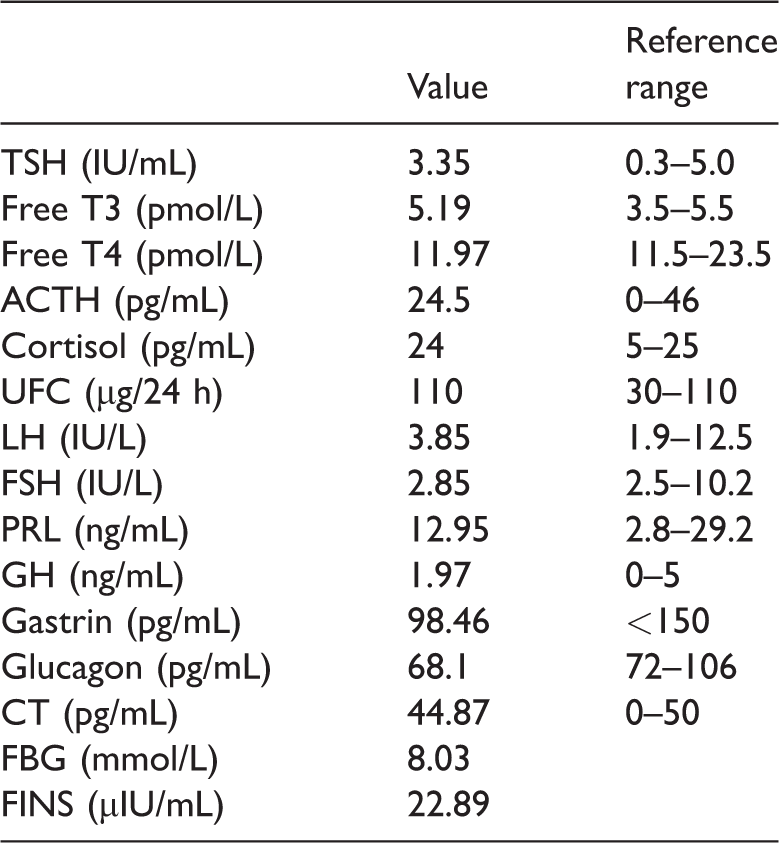
TSH: thyroid-stimulating hormone, Free T3: free triiodothyronine, free T4: free thyroxine, ACTH: adrenocorticotropic hormone, UFC: urine free cortisol, LH: luteinizing hormone, FSH: follicle-stimulating hormone, PRL: prolactin, GH: growth hormone, CT: calcitonin, FBG: fasting blood glucose, FINS: fasting insulin.

(a) An abdominal computed tomography scan (5-mm slice thickness) revealed a mass (arrow) close to the splenic hilum area measuring 5 cm in diameter. (b) A contrast-enhanced computed tomography scan revealed that the density of the mass was continuously uneven with significant enhancement (arrow). Additionally, multiple spots with unenhanced low-density areas were present. (c) The soft tissue tumor was located in the peripancreatic region but was connected to the pancreatic tail. (d) The left part was peritumoral pancreatic tissue, and the right part was tumor tissue with a complete capsule and an obvious dividing line separating it from the peritumoral tissue (hematoxylin and eosin, 100×). (e) The tumor cells were arranged in a prominent nesting pattern, and the cytoplasm of most cells was empty (100×). (f) Insulin antibody was positive in islets of the left peritumoral pancreatic tissue and negative in islets of the right tumor tissue (40×). (g) Chromogranin A was positively diffused in the tumor cells (100×).
DNA samples from the patient and four first-degree relatives (her mother, two half-sisters with the same mother, and her son) were sequenced to achieve a molecular diagnosis. DNA could not be obtained from her father, who had died in a traumatic accident 30 years previously; however, he had shown no clinical features of MEN1. Analysis of the

DNA sequencing results for the 35-year-old woman with multiple endocrine neoplasia type 1. Arrows represent the mutation sites.
Follow-up included annual hormone measurements (calcium, phosphate, PTH, glucose, insulin, glucagon, insulin-like growth factor-1, PRL, adrenocorticotropic hormone, and cortisol) and abdominal CT once a year. After 5 years, a 1.5-cm NF-pNET was detected in the pancreatic neck. Her most recent serum calcium concentration was 2.25 mmol/L, serum phosphate was 1.39 mmol/L, serum PTH was 0.86 pmol/L, and PRL was 29.8 mU/L.
Discussion
MEN1 classically causes adenomas or hyperplasia of the parathyroid glands, pancreas, and anterior pituitary gland. In the present case, all three of these components were involved, and MEN1 was clinically diagnosed. Genetic testing revealed a missense mutation at the coding nucleotide 133 in exon 2. MEN1-related parathyroid adenomas are the most common type of MEN1-related tumors; they typically show the earliest manifestation, presenting about 30 years earlier than sporadic parathyroid adenomas. 15 Prolactinomas are the most common pituitary adenomas and tend to be larger, more aggressive, and less responsive to conservative treatment. 2 The patient in the present case had a prolactinoma, which first presented as a microadenoma and responded well to bromocriptine.
Functional pNETs such as insulinomas, glucagonomas, and somatostatinomas are easily identified because of their propensity to secrete various hormones and cause hormone-related clinical signs and symptoms. The prevalence of NF-pNETs is largely underestimated if associated functional pNETs are absent, and they are either incidental findings or associated with an expanding mass. In recent years, the diagnosis of NF-pNETs in patients with MEN1 has substantially increased because of the widespread use of high-quality imaging techniques. Endoscopic US is a more recent imaging technique that is highly sensitive in detecting pancreatic endocrine tumors. 13 However, endoscopic US is considered an invasive method because it requires general anaesthesia, 16 and this widely restricts its use. Compared with US, which is the least sensitive in detecting such pNETs, CT and MRI have been shown to be effective in identifying primary tumors, with a sensitivity ranging from 55% to 78%. 17 NF-pNETs may now be the most common pNET in patients with MEN1. 5 Although our patient had no physical or biochemical indicators of pNET, we conducted a CT examination to rule out an NF-pNET and discovered an abdominal mass. However, despite the performance of a contrast-enhanced CT scan, the tissue origin could not be determined preoperatively because the tumor was not located inside the pancreatic parenchyma. The tumor was found to be connected to the pancreatic tail, and histological examination of the tissue confirmed the pancreas as its origin. Approximately 47.0% to 55.0% of MEN1-associated NF-pNETs are reportedly located in the pancreatic head, 8 while only 14.8% are located in the pancreatic tail. 9 Although the mass in our case was located outside the pancreas, it was adjacent to the pancreas tail with soft tissue density (Figure 2(c)). We speculated that the NF-pNET may have originated from the islet cells close to the pancreatic surface and broken away from the pancreatic tail because of the tensile force associated with its growth. MEN1-related NF-pNETs are usually multiple and small. 10 However, NF-PETs can also be macroadenomas (>1 cm). 18 In a recent study, the mean size of the NF-pNETs in patients with MEN1 was 3.1 cm. 5 For most patients with MEN1, NF-pNETs follow a benign course;10,19 however, one study showed that NF-pNETs are frequently malignant. 6 Metastasis is the main cause of death, 7 and the metastasis of MEN1-related NF-pNETs varies according to their size; 4% of patients with NF-PETs of ≤1.0 cm in diameter had liver metastases, 10% with NF-PETs of 1.1 to 2.0 cm, 18% with NF-PETs of 2.1 to 3.0 cm, and 43% with NF-PETs of >3.0 cm. 5 In contrast to metastasis, the occurrence of which depends on the tumor diameter, most patients developed new pNETs in the pancreatic remnant. 5 In the present case, the NF-pNET was single and large and had no synchronous liver metastasis at diagnosis; however, the patient had a new pNET in the pancreatic remnant.
MEN1 syndrome may occur in either familial or sporadic forms. The sporadic form of MEN1, in which only one affected person is identified in a previously unaffected family, is observed much less frequently (10% of cases) than the familial form (90% of cases).
11
Our patient had sporadic MEN1, and genetic testing revealed a heterozygous mutation of a G-to-A transition at coding nucleotide 133 in exon 2 of the
In summary, this report describes a case in which a patient with sporadic MEN1 had a single asymptomatic non-metastatic pNET located outside the pancreatic parenchyma that could have easily been misdiagnosed as accessory spleen. We wish to highlight the following two key points. First, NF-pNETs can be located outside the pancreatic parenchyma. Second, MEN1-related NF-pNETs can be single and may produce a new pNET in the pancreatic remnant. Although the present case involved a well-differentiated tumor, regular follow-up is necessary.
Footnotes
Declaration of conflicting interest
The authors declare there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
