Abstract
Abstracts
Objective
To assess how induction chemotherapy affects the quality-of-life (QoL) in patients with advanced tongue cancer.
Methods
This prospective study included patients who were diagnosed with advanced tongue cancer. Each patient was asked to complete the University of Washington QoL (UW-QoL), version 4, questionnaire preoperatively and at 12 months after surgery. Patients were divided into two groups based on whether or not they received induction chemotherapy.
Results
Of the 192 patients included in the analysis, 145 patients had received induction chemotherapy. There were no significant differences regarding age, sex, tumour stage, node stage, flap reconstruction, tumour resection range and radiotherapy between the two groups. The mean total hospital cost was significantly higher in patients who underwent induction chemotherapy compared with those who did not (68 000 versus 44 000 Yuan Renminbi, respectively). The two groups had similar pre-treatment and post-treatment composite QoL scores and in the 12 individual domains.
Conclusion
Induction chemotherapy had a limited effect on postoperative QoL in patients with advanced tongue cancers, but it cost significantly more to administer.
Introduction
The tongue plays a key role in swallowing and speech, so any surgical resection of the tongue tissues might cause functional deficits. Unfortunately, tongue cancer is the most common malignant tumour in the oral cavity and surgical therapy is the main treatment.1,2 However, the prognosis of advanced stage tongue cancer remains unfavourable.1,2 Therefore, multi-treatment approaches are required in patients with advanced disease in order to achieve better local regional control and improve the disease-specific survival rate.
Induction chemotherapy has been widely used in patients with advanced head and neck cancer, although controversy exists regarding whether it provides a prognostic advantage.3–7 Due to the development of the concept of functional surgery, satisfactory oncological outcome as well as good postoperative quality-of-life (QoL) are equally important. However, to the best of our knowledge, no authors have tried to evaluate how induction chemotherapy affects the QoL in patients with tongue cancer. Therefore, the current study aimed to answer this question.
Patients and methods
Patient population
This prospective study enrolled consecutive patients who were diagnosed with advanced tongue cancer (T3T4M0) according to the Union for International Cancer Control 2010 classification system 8 in the Department of Stomatology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan Province, China between January 2011 and December 2016. All patients had no impairments in communication.
In the Department of Stomatology, these patients were routinely treated with a first cycle of induction chemotherapy (paclitaxel and cisplatin) and then their response to the chemotherapy was evaluated. If the patient had a good response (i.e. complete response or partial response), then a second cycle of induction chemotherapy was performed followed by surgical resection and radiotherapy. If their response to induction chemotherapy was unsatisfactory (i.e. no significant change or disease progression) then they received surgical treatment directly. Some patients refused to receive the induction chemotherapy due to the associated complications. The cohort of patients was divided on the basis of whether the patient received induction chemotherapy. Patients who had just received one cycle of induction chemotherapy were excluded from the study.
All patients were required to complete the University of Washington QoL (UW-QoL), version 4, questionnaire 9 preoperatively and at 12 months after surgery by themselves or with the help of a healthcare professional. Information including age, sex, total hospital cost, and tumour stage were obtained and analysed.
The Institutional Research Committee of Zhengzhou University, Zhengzhou, Henan Province, China approved the study (no. ZZC201034512) and all participants provided written informed consent.
UW-QoL questionnaire
The UW-QoL questionnaire has been shown to be valid and reliable.10–14 It consists of 12 single-question domains each of which has three to six response options, which are scaled in equal stages from 0 (worst possible response) to 100 (best possible response). The domains were pain, appearance, activity, recreation, swallowing, chewing, speech, shoulder, taste, saliva, mood, and anxiety. The individual domains were scored according to the UW-QoL guidelines. 9
Statistical analyses
All statistical analyses were performed using the SPSS® statistical package, version 13.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Student’s t-test and χ2-test were used to evaluate the significance of any differences in the clinical and demographic characteristics between the two groups. The nonparametric Mann–Whitney U-test was used to analyse the UW-QoL scores. A P-value < 0.05 was considered statistically significant.
Results
A total of 263 patients were enrolled at the beginning of the study and completed the questionnaire, but 33 patients had a poor response to the first cycle of induction chemotherapy and were excluded from the study. Of the 230 patients who were included, 29 patients died of the disease within 1 year after surgery and nine patients were lost to follow-up. Therefore, the analyses included 192 patients (117 male and 75 female). The mean ± SD age of the patients was 60.3 ± 8.8 years (range 36–79 years). All patients had a T3/T4 tongue squamous cell carcinoma, and 71 (37.0%) patients had cervical node metastases. Hemiglossectomy and total glossectomy were performed in 96 and 18 patients, respectively. A total of 131 patients received a free-flap reconstruction, which included 81 anterolateral thigh flaps and 50 radial forearm flaps. A total of 35 patients had received modified radical neck dissection. A total of 152 patients received postoperative radiotherapy within 6 weeks after the operation.
Of the 192 patients, 145 received induction chemotherapy and 47 patients did not. There were no significant differences regarding age, sex, tumour stage, node stage, flap reconstruction, tumour resection range, and radiotherapy between the two groups (Table 1). The mean total hospital cost was significantly higher in the patients who underwent induction chemotherapy compared with those who did not (68 000 versus 44 000 Yuan Renminbi, respectively; P < 0.001).
Clinical and demographic characteristics of patients with advanced tongue cancer who did or did not receive induction chemotherapy (IC).

Data presented as mean or n of patients.
*P < 0.001 compared with the IC group; all other comparisons between the two groups were not significant (P ≥ 0.05); continuous data were compared using Student’s t-test and categorical data were compared using χ2-test.
Table 2 presents the pre-treatment composite quality-of-life scores for patients who did and did not receive induction chemotherapy. There was no significant difference in the composite scores or the scores for the 12 individual domains between the two groups.
Comparison of the pre-treatment quality-of-life scores of patients with advanced tongue cancer who did or did not receive induction chemotherapy (IC).
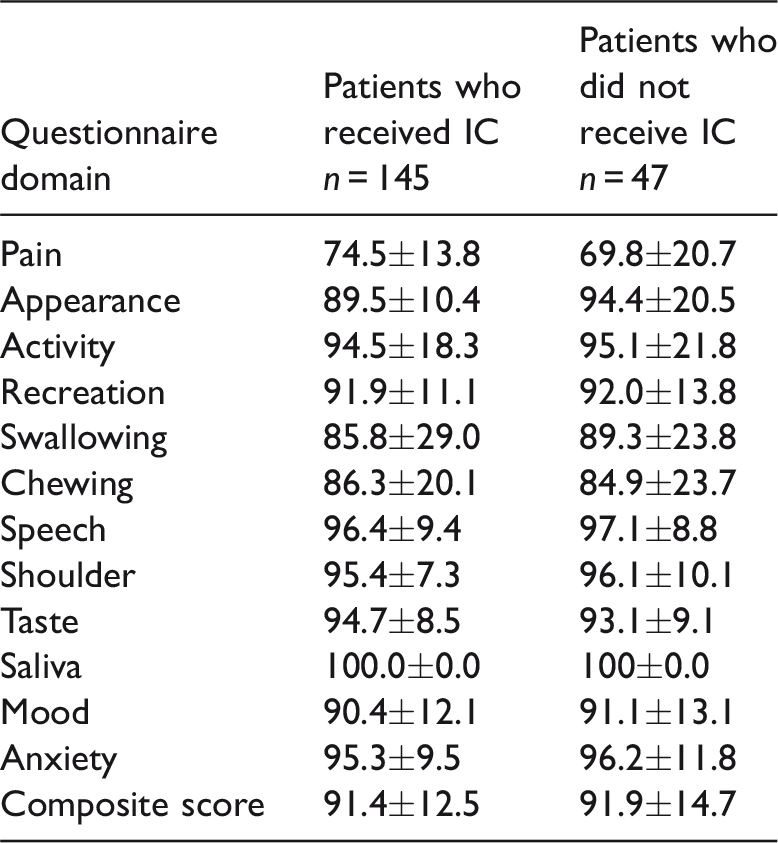
Data presented as mean ± SD.
No significant between-group differences (P ≥ 0.05); Mann–Whitney U-test.
Table 3 presents the post-treatment composite quality-of-life scores for patients who did and did not receive induction chemotherapy. There was no significant difference in the composite scores or the scores for the 12 individual domains between the two groups.
Comparison of the post-treatment quality-of-life scores of patients with advanced tongue cancer who did or did not receive induction chemotherapy (IC).
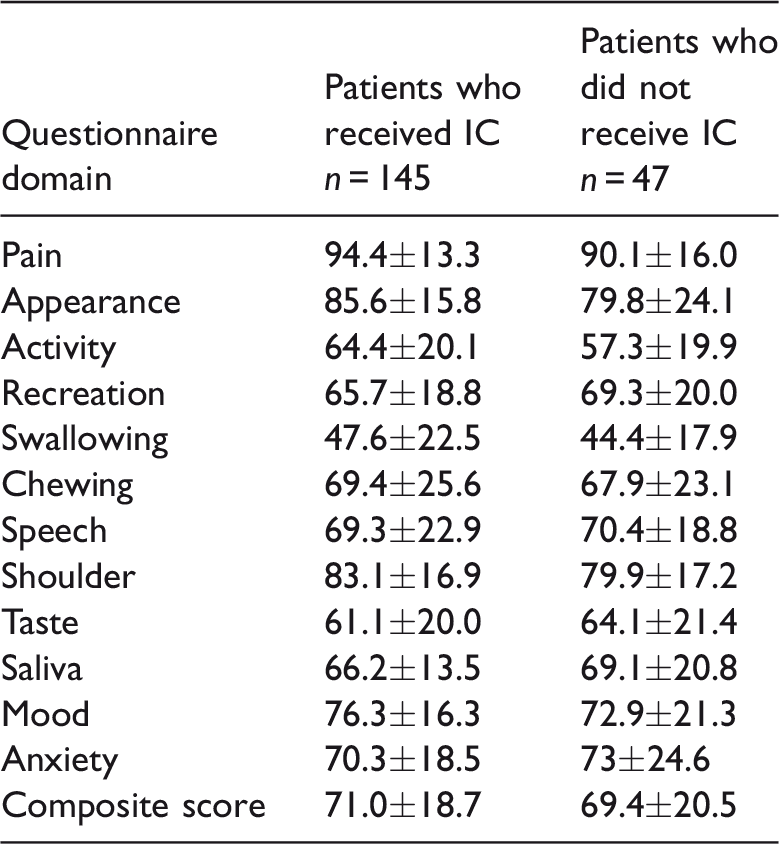
Data presented as mean ± SD.
No significant between-group differences (P ≥ 0.05); Mann–Whitney U-test.
Discussion
The role of induction chemotherapy in terms of providing benefits to patients with head and neck cancers remains unclear.3–7 In a previous study, 3 8031 patients with locally advanced head and neck squamous cell carcinoma (T4 or N2b to N3) undergoing radiotherapy and chemotherapy were divided into concurrent chemotherapy and induction chemotherapy cohorts; and no significant survival advantage was noted in patients receiving induction chemotherapy, but they tended to receive lower radiotherapy doses. Similarly, a randomized phase III trial failed to show any survival advantage by using induction chemotherapy in patients with unresectable head neck cancers. 15 However, in an Italian randomized trial of 421 patients, compared with those treated with locoregional therapy alone without compromising treatment compliance, increased rates of treatment response, overall survival, and progression-free survival were observed in patients with induction chemotherapy. 16 Each of these international trials may be statistically underpowered, highlighting the need for studies with larger sample sizes.
Functional outcomes and oncological results are equally important, because a good postoperative QoL could help cancer survivors regain their normal social life. However, no authors have tried to assess the role of induction chemotherapy in affecting the QoL. A previous study focused on evaluating speech and swallowing ability in 15 patients enrolled in a phase II clinical trial of induction chemotherapy followed by surgical resection. 17 The authors concluded that induction chemotherapy had a negligible effect on speech and swallowing physiology, but might provide symptomatic improvement of pain and swallowing after treatment. 17 Similar results were also reported by a pilot study in patients with head and neck cancer. 18 However, both of these studies had a relatively small sample size and only analysed the immediate influence on outcomes.17,18 Treatment of head and neck cancers usually consists of multiple procedures and long-term functional results are also important.
The current study is the first to report that induction chemotherapy does not appear to help the recovery of the composite QoL score. In contrast, a previous study reported that patients with T4 laryngeal cancer treated with induction chemotherapy followed by chemoradiotherapy were able to return to, and in many patients exceed, pre-treatment performance and QoL. 19 The difference between these findings might be explained by the fact that, unlike the patients in the current study, those patients were treated using organ-sparing approaches without surgical resection. 19 Previous research has reported that patients with more advanced tumour stages were less likely to have worsened swallowing and more likely to have improved swallowing. 20
Restoration of swallowing and speech ability are the main goals of treating tongue cancers. Although the extent of the resection range that was performed in the present study was based on pre-treatment lesion size, it was anticipated that the actual resection range might be smaller than the expected resection range due to tumour shrinkage induced by chemotherapy, which would result in the preservation of more tongue tissue and better post-treatment QoL. However, the present findings showed that the two groups of patients had similar tumour resection ranges. Also, the swallowing, speech and chewing abilities did not differ significantly between the two groups post-treatment. Therefore, limited functional advantages were achieved by induction chemotherapy. No related literature was available for comparison. More studies are needed to address this issue.
The only significant difference between the two groups was that it cost more to treat patients who underwent induction chemotherapy compared with those who did not. One cycle of induction chemotherapy usually costs approximately 10 000 Yuan Renminbi in our hospital, but this finding might be variable owing to different medical insurance policies in different countries.
In conclusion, induction chemotherapy had a limited role in affecting postoperative QoL in patients with advanced tongue cancers, but it costs significantly more to administer.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
