Abstract
Objective
To explore the relationship between cellular apoptosis and hypertrophy of the ligamentum flavum in the lumbar region.
Methods
Thirty patients with lumbar spinal stenosis were evaluated. Hypertrophy of the ligamentum flavum was present in 15 patients and absent in 15. Hematoxylin–eosin staining and transforming growth factor beta (TGF-β) immunohistochemical testing were applied to compare these two groups.
Results
Derangement of fibrous alignment, fibrocartilage changes, and infiltration of inflammatory cells were observed in the patients with hypertrophy of the ligamentum flavum, while fibrous alignment was normal and few inflammatory cells were observed in patients without hypertrophy. Immunohistochemical studies showed positive expression of TGF-β in patients with hypertrophy, while expression was negative in patients without hypertrophy. The integrated optical density was 2.6556708 in the hypertrophy group and 23104671 in the normal controls.
Conclusions
Expression of TGF-β was closely related to hypertrophy of the ligamentum flavum. Appropriate application of the TGF-β expression level can be used to predict progression of hypertrophy of the ligamentum flavum.
Introduction
Lumbar spinal stenosis is one of the main causes of pain and numbness of the back and lower limbs, and hypertrophy of the ligamentum flavum is the main etiology of such pain and numbness in patients with lumbar spinal stenosis.1,2 However, although many studies have been performed to evaluate degenerative changes of the ligamentum flavum in the cervical and thoracic regions, no consensus regarding the mechanism of ligamentum flavum hypertrophy in the lumbar region has been reached.3–5 In the present study, we compared the ligamentum flavum in patients with and without hypertrophic changes by hematoxylin–eosin (HE) staining and immunohistochemical staining for transforming growth factor beta (TGF-β) to identify factors contributing to degenerative changes of the ligamentum flavum in the lumbar region.
Materials and methods
Specimens
All experimental procedures were carried out with the approval of the ethics committee of the Sixth Affiliated Hospital of Xinjiang Medical University. Specimens of the ligamentum flavum were collected from 30 patients (22 male, 13 female; age, 34–76 years) during surgical treatment with the consent of the patients and their families from June 2015 to December 2016. Among those 30 patients, 15 had hypertrophy of the ligamentum flavum in the lumbar region and 15 did not.
Observation of the specimens
Thickening of the ligamentum flavum was measured on the axial T1-weighted image that was perpendicular to the axis of the spinal canal and parallel to the laminae, where the ligamentum flavum was seen along its entire length. Hypertrophy of the ligamentum flavum was diagnosed when the thickness exceeded 4 mm.6,7
The inclusion criterion was hypertrophy of the ligamentum flavum in the lumbar region; 15 patients did not have hypertrophy of the ligamentum flavum. The exclusion criteria were nonspecific low back pain, an infection or tumor in the lumbar spine, and conditions that prevented surgical treatment.
Slices were observed after HE staining and immunohistochemical staining, and the expression level of TGF-β was quantified as the integrated optical density (IOD).
The immunohistochemical studies were performed as follows. After the tissue was embedded in paraffin, 4 -µm slices were cut, incubated in 3% hydrogen peroxide for 10 minutes, and washed in phosphate-buffered saline (PBS) three times for 3 minutes each. Antigen retrieval was performed by microwave oven heating of the tissue. The tissue was immersed in 0.01 ml of citrate buffer, boiled for 10 minutes, cooled at room temperature, and washed in PBS three times for 3 minutes each. The primary antibody was added onto the slice, which was then kept in a humidity chamber at 4℃ overnight. After balancing at room temperature for 5 minutes and washing in PBS three times, biotinylated anti-rabbit IgG and anti-mouse IgG were added. The slice was then kept at 37℃ for 30 minutes, washed in PBS three times, and stained with 3,3′-diaminobenzidine for 10 minutes. It was then washed in tap water, counterstained with hematoxylin, and dehydrated by gradient alcohol dehydration (75%, 80%, 95%, and 100%). The specimens were observed under a light microscope, and photographs were taken for further analysis. Further analysis was then carried out using Image-Pro Plus 6.0 software (Media Cybernetics, Silver Spring, MD, USA).
Five high-magnification visual fields were observed for each slice. In each visual field, 100 cells were evaluated to determine the ratio of positively stained cells. The average value of five visual fields was calculated. The samples were scored according to the percentage of stained cells (0: <10%, 1: 10%–20%, 2: 20%–50%, and 3: >50%) and the color intensity of the cells (0: none, 1: light yellow, 2: brown-yellow, 3: brown). The product of these two scores was used to classify the expression level of TGF-β. Expression of TGF-β was considered negative when the product was <2, positive when the product was 3 to 5, and strongly positive when the product was ≥6. 8
Statistical analysis
The number of fibroblasts positively stained in the immunohistochemical studies and the IOD were compared using independent-sample t-tests between patients with and without ligamentum flavum hypertrophy. Classification of TGF-β immunoreactivity was compared using X2 analysis. All statistical analyses were carried out by SPSS 17.0 software (SPSS Inc., Chicago, IL, USA). The difference was considered significant when P < 0.05.
Results
The average thickness of the hypertrophic ligamentum flavum was 5 mm. They had increased fragility, and were partly calcified and adherent to surrounding tissues. The ligamentum flavum in the normal controls was thinner than 4 mm, had fine surface gloss and flexibility, and had no calcification or ossification. The IOD sum of the specimens was 2.6556708 in the hypertrophy group and 23104671 in the control group. HE staining showed derangement of fibrous alignment, fibrocartilage changes, and infiltration of inflammatory cells in the hypertrophy group, while normal fibrous alignment and few inflammatory cells were present in the control group (Figure 1).
Hematoxylin–eosin staining. (a, b) Derangement of fibrous alignment, fibrocartilage changes, and infiltration of inflammatory cells were present in the specimens from patients with hypertrophy of the ligamentum flavum. (c, d) Few inflammatory cells and normal fibrous alignment were shown in the specimens from patients without hypertrophy of the ligamentum flavum.
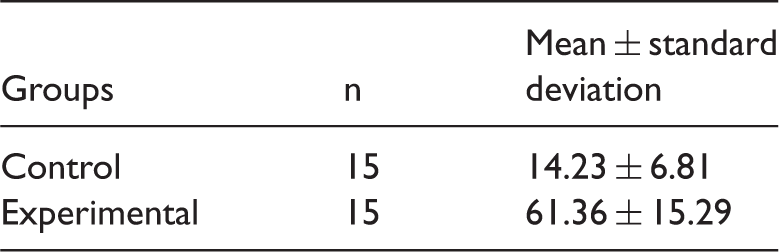
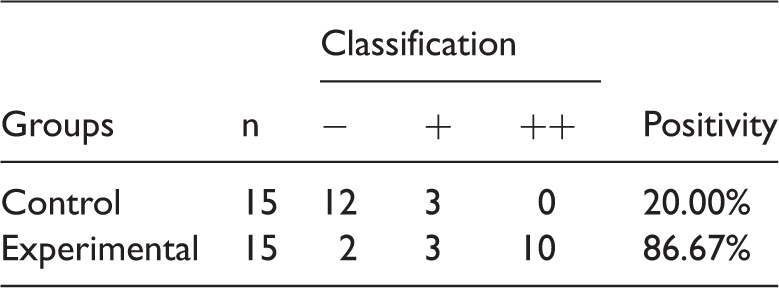
Immunohistochemical studies showed significantly more positively stained fibroblasts in patients with than without hypertrophy (Figure 2, Table 1). Similarly, the classification of TGF-β immunoreactivity was significantly higher in patients with than without hypertrophy (Figure 2, Table 2).
Immunohistochemical studies. (a) Positive expression of transforming growth factor beta was present in patients with ligamentum flavum hypertrophy. (b) Expression was negative in patients without hypertrophy. Number of positively stained fibroblasts. The number of positively stained fibroblasts in the immunohistochemical studies was significantly different between the two groups (independent-sample t-test: t′ = 11.325, P < 0.001). Expression levels of transforming growth factor beta. −: negative, +: positive, ++: strongly positive. The expression levels of transforming growth factor beta were significantly different between the two groups (X2 = 5.921, P = 0.023).
Discussion
Spinal stenosis is a common disease among older patients. It can cause pain and numbness of the back and lower limbs and decreases the locomotive abilities of the patient. 9 The main pathologic changes include osteoproliferation of the lamina and facet joints, protrusion of the vertebral discs, and hypertrophy of the ligamentum flavum, with the last being the main cause of the spinal stenosis.10–12 Repeated mechanical stretching due to instability of the degenerative lumbar spine is reportedly the main reason for injury to the ligamentum flavum, which leads to regional scar formation and hypertrophy of the ligament. 4 Suppression of the cauda equina by the ligamentum flavum is normally the cause of symptoms in patients with hypertrophy of the ligamentum flavum. 13
Park et al. 14 found that the average thickness of the ligamentum flavum was 4.44 mm in patients with hypertrophy and 2.44 mm in patients without hypertrophy. These findings are similar to those in the present study. Sairyo et al. 15 analyzed the ligamentum flavum of 77 patients ranging in age from 10 to 85 years and found that the thickness of the ligamentum flavum increased as the patient’s age increased, and the most dramatic increase occurred in the L3/4 and L4/5 lumbar regions. These findings are also in accordance with those in the present study.
In our case series, HE staining of the ligament specimens revealed inflammatory cells, disarranged fibrous tissues, fibrous cartilage, and ossification of the fibrous tissues in patients with hypertrophy of the ligamentum flavum. These findings were vastly different from those in the normal controls.
Park et al. 14 found that TGF-β1 was more abundant in the ligamentum flavum of patients with than without hypertrophic changes. Nakatani et al. 16 found that the microRNA expression of type I, III, and V collagenous fibers and TGF-β1 were significantly higher in patients with than without ligamentum flavum hypertrophy. These microRNAs were significantly downregulated after adding antibodies to TGF-β1, and they increased after adding exogenous TGF-β1. They stated that this proves the role of TGF-β1 in hypertrophic changes of ligamentum flavum. However, Sairyo et al. 16 found that TGF-β increased only in the early stages of ligamentum flavum hypertrophy and decreased after completion of the scar formation. In the current study, we found that the level of TGF-β was significantly higher in patients with than without ligamentum flavum hypertrophy, which is in accordance with previous studies; it may also indicate that the hypertrophy was still progressing in these patients. Considering that the TGF-β level is related to hypertrophy of the ligamentum flavum, it is possible that higher TGF-β levels can be used to predict more certain and severe hypertrophy of the ligamentum flavum in the future. The relationship between the TGF-β level and the possibility and severity of ligamentum flavum hypertrophy should be studied further in animal models and patient trials.
Conclusions
The expression of TGF-β was closely related to degenerative changes and hypertrophy of the ligamentum flavum. Appropriate application of the TGF-β expression level can be used to predict the progression of hypertrophy of the ligamentum flavum.
Footnotes
Authors’ contributions
Aierken Amudong: study design, patient selection, patient recruitment. Tuerhongjiang Abudourexiti: surgical treatment, data collection. Aikeremujiang Muheremu: statistical analysis, writing of the paper.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was funded by the Natural Science Foundation of Xinjiang Uygur Autonomous Region, China (2016D01C220).
