Abstract
Objective
To determine if there was herd protection conferred to unprotected healthcare workers (HCWs) by N95 respirators worn by colleagues.
Methods
Data were analysed from a prospective cluster randomized clinical trial conducted in Beijing, China between 1 December 2008 and 15 January 2009. A minimum compliance level (MCL) of N95 respirators for prevention of clinical respiratory illness (CRI) was set based on various compliance cut-offs. The CRI rates were compared between compliant (≥MCL) and non-compliant (<MCL) N95 wearers by ward, and between non-compliant wearers and control subjects who did not wear masks.
Results
Data were analysed from 949 HCWs who wore N95 respirators and 125 HCWs who did not wear masks. At 50% MCL there were no significant differences in the CRI rates between compliant and non-compliant N95 wearers by ward. In multivariate analysis, the CRI rate in non-compliant HCWs was significantly lower compared with controls (relative risk 0.26; 95% confidence interval 0.08, 0.82).
Conclusion
This study suggests herd protection from use of N95 respirators by colleagues within a hospital ward.
Introduction
Herd immunity, sometimes referred to as ‘indirect protection’
1
or ‘herd effect’1–3 from vaccination, is commonly defined as “
Personal protective equipment plays an important role in the prevention of respiratory infections in healthcare settings, particularly during the early phase of outbreaks and pandemics when drugs and vaccines are not available.15–17 Respiratory protective equipment such as N95 respirators are recommended by health organizations and countries to reduce the risk of infection among HCWs due to their good filtration efficiency.18–20 Medical masks are also widely used in healthcare settings to prevent the spread of infections from the sick and to protect wearers from splash and spray of blood and body fluids. 19 However, medical masks are not designed for respiratory protection and their efficacy is not proven.19,21
The efficacy of N95 respirators depends on many factors including compliance. Compared with hand hygiene and other non-pharmaceutical interventions, the compliance with respiratory protection is lower among HCWs,22,23 however it is high in Asian countries due to cultural acceptability of wearing masks among HCWs. 24 Compliance among HCWs is related to various individual and organizational factors, including culture, risk perception, presence of adverse events and availability.22,25,26
It is unclear whether the concept of herd protection can be applied to personal protective equipment, or whether the use of N95 respirators by enough HCWs can reduce infection risk in unprotected HCWs within a closed setting such as a ward. Previous research has demonstrated that higher compliance rates reduce the total number of cases of respiratory tract infections. 27 The aim of this study was to examine the herd protection effect of wearing N95 respirators in HCWs within a hospital ward.
Participants and methods
Study design
This study analysed data from a prospective cluster randomized clinical trial that was conducted in Beijing, China between 1 December 2008 and 15 January 15 2009 (Trial Registration: Australian New Zealand Clinical Trials Registry (ANZCTR), ACTRN: ACTRN12609000257268). 21 Participants were hospital HCWs aged ≥18 years from six wards including the emergency departments, fever clinic/infectious disease departments, respiratory clinics, respiratory wards, paediatric departments and other departments from 15 tertiary hospitals. As the unit of randomization was hospitals, all HCWs in a ward wore the same type of masks, and HCWs were randomized into a non-fit tested N95 group, a fit tested N95 group and a medical mask group through a computerized randomization program. A convenience control arm was also included with participants from another nine hospitals. In the control arm, some participants used medical masks, while other participants did not use any masks at all. The study protocol was approved by the Institutional Review Board and Human Research Ethics Committee of the Beijing Ministry for Health (no. 2 (2), Year 2007). 21 Written information about that trial was offered to participants, and verbal informed consent was provided by them.
Given that the difference in infection rates between fit tested and non-fit tested N95 respirator wearers was non-significant and fit test failure was extremely low in the fit-tested arm, 21 both of the N95 arms were pooled into a N95 respirator group. Only no-mask users were grouped into the controls, and these were HCWs who did not use any masks.
Primary endpoint and compliance rate
The primary endpoints of the clinical trial included clinical respiratory illness (CRI), 21 defined as two or more respiratory or one respiratory symptom and a systemic symptom. 28 CRI was analysed as the main outcome variable in this study.
Based on the mean number of working hours and the mean number of hours spent using a medical mask/respirator for each participant over the trial period, a compliance rate variable was created by dividing the mean number of hours of respirator use by the mean number of working hours: compliance rate (%) = mean of N95 respirator wearing hours/mean of working hours × 100.
Statistical analyses
The minimum compliance level (MCL) of N95 respirators was estimated based on the comparison of the CRI rates of all N95 users between any compliance rate ranging from ≤10% to ≤100% and that of the control arm. The category of ≤10% was excluded from the analysis because there were no participants in the ≤10% cut-off compliance level. The subjects below the MCL were defined as non-compliant HCWs (compliance level < MCL), and those above, as compliant HCWs (compliance level ≥MCL).
The differences in CRI rates between the non-compliant and compliant HCWs by ward were analysed using Pearson’s χ2-test and the relative risk (RR) of CRI was calculated. The CRI rates in non-compliant N95 wearers were compared with the control arm to see if non-compliant HCWs had any herd protection effect, which would result in lower infection rates compared with the control subjects. In the univariate analysis, the RR of CRI in non-compliant HCWs compared with the control arm was calculated by simple logistic regression. Multivariate analysis was conducted to adjust for the potential confounders including sex, handwashing and flu vaccination by multiple logistic regression analysis using the GENMOD Procedure (SAS version 9.4 software; SAS Institute Inc., Cary, NC, USA). A
Results
A total of 1441 HCWs were randomized into a non-fit tested N95 group ( Relative risk of clinical respiratory illness (CRI) in N95 respirator users (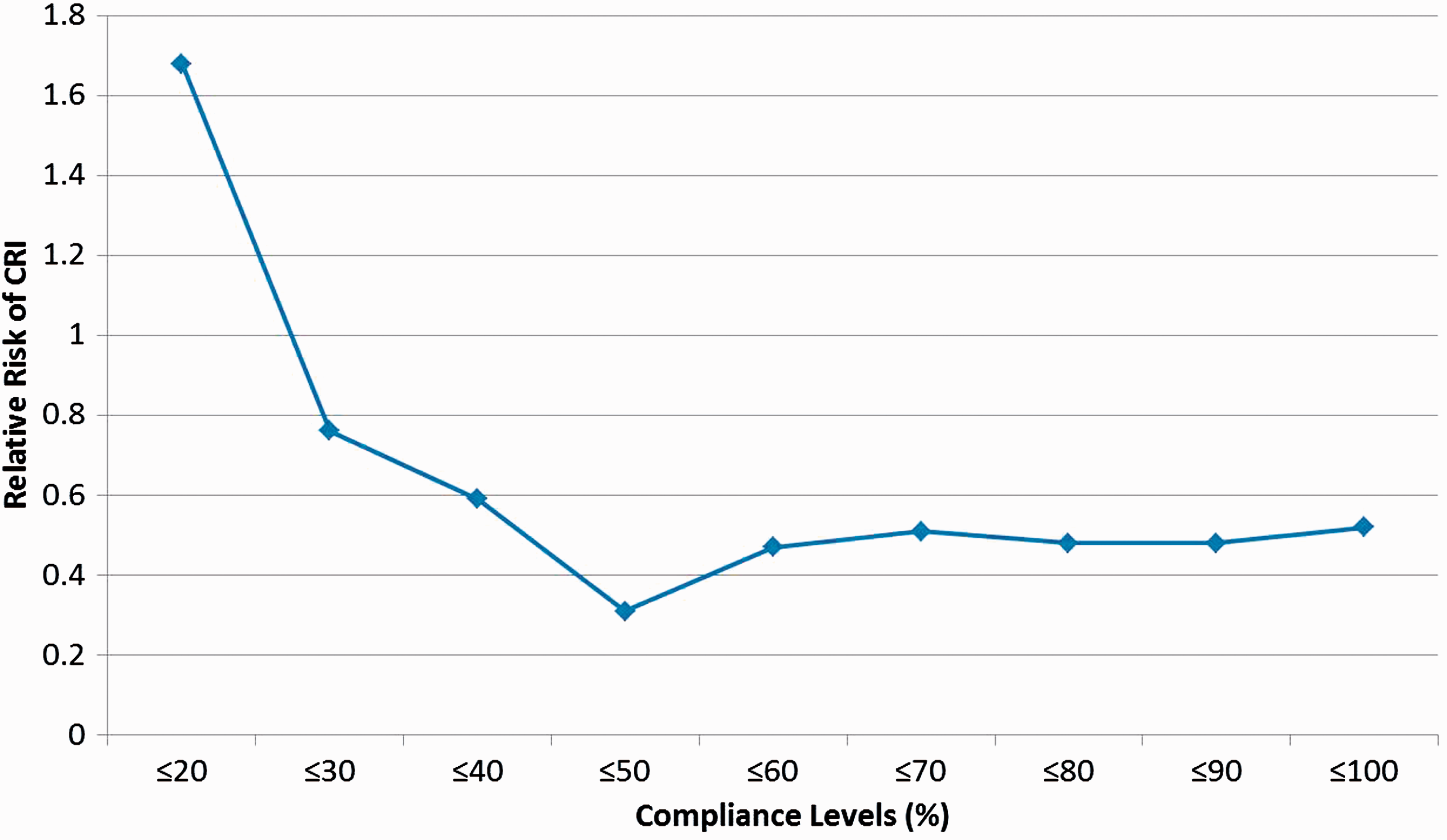
Comparison of rates of clinical respiratory illness (CRI) in N95 respirator users (

Data presented as
Pearson’s χ2-test.
RR, relative risk; CI, confidence interval; NA, not available; NS, no significant between-group difference (
Comparison of rates of clinical respiratory illness (CRI) in non-compliant N95 respirator users (

Data presented as
Undertake handwashing every time or most times after touching a patient.
Influenza vaccination in 2007/2008/2009.
RR, relative risk; CI, confidence interval; NS, no significant between-group difference (
Discussion
Results of the present study suggest that there may be herd protection from respiratory infections by HCWs wearing N95 respirators in hospital wards. In this present study, there was an inverse relationship between the level of compliance with wearing an N95 respirator and the risk of CRI; and the MCL was shown to be 50%. A previous analysis of data from this prospective cluster randomized clinical trial demonstrated that N95 respirators have efficacy in protecting against CRI.
21
However, this present analysis has demonstrated that there was no significant difference between the rates of CRI in compliant and non-compliant N95 users by ward, with each ward being a closed environment for transmission of infection, which suggests that a herd protection effect for the non-compliant users was conferred by the compliant users. This present analysis also showed that non-compliant N95 users had a significantly lower rate of infection than the control subjects who did not use any masks (RR = 0.26; 95% CI 0.08, 0.82;
In contrast to non-communicable diseases, it is the unique contagious nature of infections that results in the potential for herd protection. Transmission of infection within a closed setting is dynamic, with chains of transmission from one infected person to the next being established. 29 As such, interruption of transmission below a critical threshold by any means, whether it is vaccination or personal protective equipment, can reduce the chains of transmission and result in ‘herd protection’. As shown in this present study, even though not 100% of participants of each ward wore N95 respirators above the MCL (50% compliance rate), the reduction of incidence of CRI still occurred in the non-compliant users due to the intervention by N95 respirators. To date, herd protection has been discussed only in the context of immunization programmes. However, this present study suggests that a herd effect as a result of interventions other than vaccinations are possible.
This study had a number of limitations. First, the data from the convenience control arm may have been a source of bias. 21 The control arm was not randomized because mask use was widespread in Chinese healthcare settings and it was unethical to ask HCWs to not wear a mask during the influenza season. Most subjects in the control arm wore medical masks at some stage, but not N95 respirators. Secondly, the sample size within each compliance level for the rates of CRI and for the analyses based on variables such as sex, handwashing and vaccinations was relatively small (mostly <10), which might reduce the power of the study. Thirdly, community exposure cannot be ruled out and participants may have contracted CRI in the community.
In conclusion, this study suggests that herd protection from respiratory infections was provided by N95 respirators. The benefits of respirator use within a closed setting such as a hospital ward may extend beyond the individual who is wearing it. Future studies on the herd effects of other interventions that interrupt transmission of infections, and larger scale studies, are required to confirm the herd effect and implications for hospital infection control.
Footnotes
Acknowledgement
Our sincere thanks go to the staff at the Beijing Centre for Disease Control, Beijing, China, the staff at the hospitals involved in the prospective cluster randomized clinical trial and 3M's help in fit-testing during the trial.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Raina MacIntyre has held an Australian Research Council Linkage Grant with 3M as the industry partner for investigator-driven research. 3M have also contributed supplies of masks and respirators for investigator-driven clinical trials. She has received research grant that laboratory testing was in-kind support from Pfizer, GSK and Bio-CSL. Dr. Abrar Chughtai had testing of filtration of masks by 3M for PhD. Xin Chen declares that she has no conflicts of interest.
Funding
This research received no financial grant from any funding agency in the public, commercial, or not-for-profit sectors.
