Abstract
Objective
To evaluate the immunohistochemical staining pattern of caudal type homeobox 2 (CDX2) protein in germ cell tumours (GCTs) of the testis.
Methods
This study reassessed archival tissue samples collected from patients diagnosed with primary and metastatic testicular GCTs for CDX2 immunoreactivity using standard immunohistochemical techniques. Positive nuclear immunostaining was evaluated with regard to both the staining intensity and the extent of the staining.
Results
Tissue sections from primary and metastatic testicular GCTs (n = 104), germ cell neoplasia in situ (GCNis) (n = 5) and benign testicles (n = 15) were analysed. The GCNis and benign testicular tissues showed no immunoreactivity for CDX2. Strong and diffuse staining of CDX2 was demonstrated only in the mature colonic epithelium of teratomas in both primary and metastatic GCTs. CDX2 positivity in other tumours (one pure yolk sac tumour, one yolk sac component of a mixed GCT and one pure seminoma) was infrequent, and was only weak and focal.
Conclusions
CDX2 immunostaining should be interpreted based on both the staining intensity and the extent of staining so as not to cause misdiagnosis. Teratomas with colonic-type epithelium should be considered in the differential diagnosis if a metastatic tumour with an unknown primary shows prominent CDX2 immunostaining.
Introduction
The caudal type homeobox 2 (CDX2) gene is a homeobox gene that encodes a protein that functions as a nuclear transcriptional factor. 1 It plays a role in regulating the development and differentiation of the embryonic intestinal epithelium by activating intestine-specific proteins.1–3 Among normal adult tissues, CDX2 protein is strongly and diffusely produced in all intestinal epithelial cells, including tissue from the duodenum to the distal rectum, and also in the epithelium of pancreatic small ducts and ductules. 4 Moreover, it has been found in intestinal metaplasia occurring in the stomach, oesophagus, gall bladder, and urinary bladder.5–8 Due to its relative specificity for the gastrointestinal tract, the use of anti-CDX2 antibodies has become part of the immunohistochemistry panel for carcinomas of unknown primary origin; and it is accepted as being highly sensitive for the identification of colorectal adenocarcinomas.9,10 However, various studies have demonstrated CDX2 positivity in a small percentage of ovarian mucinous tumours, 11 endometrial lesions, 12 carcinoids 13 and some adenocarcinomas of other sites such as uterine cervix, 14 prostate, 15 and lung. 16
The large majority of primary testicular tumours originate from germ cells.17,18 These tumours are classified as seminomas, embryonal carcinomas, yolk sac tumours, trophoblastic tumours and teratomas. As they may comprise confusing histological growth patterns, unusual variants, and mimic other tumours, diagnosing germ cell tumours (GCTs) can be challenging. 19 Their correct identification often has important therapeutic and prognostic implications. Particularly when dealing with metastatic tissues, immunohistochemical studies are needed to resolve these dilemmas. The diagnostic utility of CDX2 in GCTs of the testis has not been well studied. This study examined the immunohistochemical staining pattern of CDX2 in GCTs of the testis, germ cell neoplasia in situ (GCNis) and normal seminiferous tubules.
Patients and methods
Patient population
This retrospective study was conducted on archival tissue samples collected from patients diagnosed with testicular GCTs between January 2005 and December 2014 in the Department of Surgical Pathology, Uludag University, Bursa, Turkey. Normal seminiferous tubules were collected from the testicular tissues of patients who underwent surgery for benign testicular disorders. All of the cases diagnosed as testicular GCTs were included in the study. The study was approved by the Institutional Review Board and Scientific Research Committee of Uludag University (no. 2015-14). There was no requirement for informed consent for the study.
Histological examination
Using archival tissue samples from the Department of Pathology, Uludag University, haematoxylin and eosin-stained slides of the cases were reassessed and one representative block was selected for immunohistochemical staining. CDX2 staining was performed on 4 µm-thick sections from formalin-fixed, paraffin-embedded tissues, using the Bond Polymer Refine Detection kit according to the manufacturer’s instructions (Leica Microsystems UK, Milton Keynes, UK). Immunostaining was carried out using a fully automated immunohistochemistry staining system (Leica Bond-Max System; Leica Microsystems (UK) Ltd) according to the manufacturer’s instructions. Ethylenediaminetetra-acetic acid was used for antigen retrieval by the automated system. The sections were incubated with a mouse monoclonal primary antibody against CDX2 (clone AMT28; Leica Microsystems UK). Colonic adenocarcinoma tissue from colectomy specimens from the Department of Pathology, Uludag University, was used as a positive control and a negative control consisted of no primary antibody. The immunoreactivity was evaluated using a light microscope (model BX51TF, Olympus, Tokyo, Japan). Only nuclear staining was considered as positive immunostaining. The staining intensity was scored as follows: 0 for no staining; 1+ for weak staining; 2+ for moderate staining; 3+ for intense staining. Four groups were categorized according to the percentage of nuclear staining (0 for < 1%, 1+ for 1% to 25%, 2+ for > 25% to 50%, 3+ for > 50%). A total of 10 fields (at least 500 cells) were counted at x400 magnification for each specimen.
Results
Distribution of the testicular germ cell tumours evaluated in the study (n = 104).
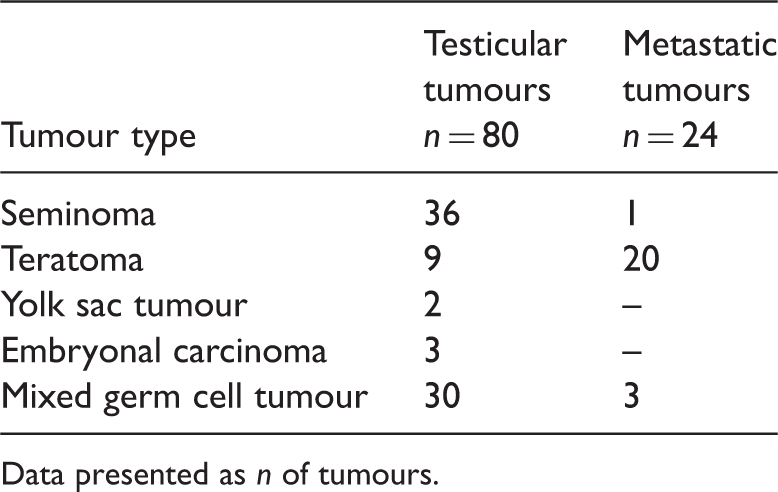
Data presented as n of tumours.
Histological components of testicular mixed germ cell tumours (n = 33).

Data presented as n of tumours.
Mixed germ cell tumours can present with two or more neoplastic components.
Intense (3+) and diffuse (3+) CDX2 nuclear positivity was identified in all teratomas that had mature colonic-type epithelium, which was 11 of 30 cases of testicular GCTs with teratoma (including mixed GCTs with teratoma) and 16 of 22 cases of metastatic GCTs with teratoma (including mixed GCTs with teratoma) (Figure 1). Except for the teratomas, CDX2 immunoreactivity was detected only in one pure yolk sac tumour (Figure 2), one yolk sac component of a mixed GCT and one pure seminoma, with all three tumours showing weak (1+) and focal (1+) staining. No CDX2 immunostaining was observed in the other components of mixed GCTs including embryonal carcinomas and choriocarcinoma. Similarly, GCNis and benign testicular tissues showed no immunoreactivity for CDX2.
Representative photomicrographs showing a section of testicular teratoma including glands with colonic-type epithelium: (a) haematoxylin and eosin staining; (b) showing strong and diffuse immunostaining for caudal type homeobox 2 (CDX2) protein. The colour version of this figure is available at: http://imr.sagepub.com. Scale bar 100 µm. Representative photomicrographs showing a section of testicular yolk sac tumour: (a) haematoxylin and eosin staining; (b) showing weak and focal immunostaining for caudal type homeobox 2 (CDX2) protein. The colour version of this figure is available at: http://imr.sagepub.com. Scale bar 100 µm.

Discussion
Germ cell tumours are the most common malignancy of the testis.17,20 These tumours demonstrate a wide range of subtypes with various histopathological patterns.17,20–22 The most common and most significant challenges in testicular pathology are differential diagnoses between seminoma and nonseminomatous GCT, as well as between GCTs and testicular tumours of non-germ cell type, particularly the sex cord-stromal tumours. 22 In addition, diverse morphological (architectural and cytological) features cause diagnostic pitfalls when the tumour is initially diagnosed as a metastatic foci without a known primary tumour. 21 An accurate diagnosis of testicular GCT is required in order for the physician to be able to offer the most appropriate treatment. Therefore, in most cases, supplementary studies are needed to distinguish GCTs from many other morphologically similar tumours.21–25 In this context, immunohistochemical studies that identify a number of biomarkers, including alpha-fetoprotein, CD30, glypican 3, KIT, octamer-binding transcription factor 3/4, sal-like protein 4, placental alkaline phosphatase, sex determining region Y-box (SOX) 2 and SOX17 have been useful in the diagnosis of GCTs. 17 In addition, the molecular analysis of chromosomes 12 p, X, 7, 8, 21, Y, 1 p, 11, 13, and 18, are being investigated as potential diagnostic tools.17,26 These molecular analyses could provide an exact diagnosis, particularly in GCTs derived from GCNis. 26 However, many pathology centres are not equipped to use molecular studies and immunohistochemical studies continue to be easy, affordable and reproducible. Therefore, new immunohistochemical markers are still being investigated to differentiate between these testicular tumours.
The CDX2 gene is a member of the caudal-related homeobox gene family that is expressed during the normal development of the intestinal tract and is important for hindgut development in humans.1–3 As expected, it has been shown to be expressed strongly and extensively in colonic epithelial tumours, and it is considered as an effective marker for the identification of colorectal adenocarcinomas.4,27 Although anti-CDX2 antibodies are included in a panel of antibodies to identify metastatic colonic adenocarcinomas,4,27 subsequent studies have also demonstrated CDX2 immunoreactivity in adenocarcinomas of the oesophagus, stomach, small intestine, ampulla of Vater, mucinous-type ovary, intestinal-type sinonasal cavities, urinary bladder, urachus, prostate and gastrointestinal neuroendocrine neoplasms. 4,11,23,28–30 Staining in these tumours was less frequent and usually patchy/focal with less intense immunoreactivity than that observed in colonic adenocarcinomas. 4 However, some researchers concluded that the anti-CDX2 antibody is not suitable for distinguishing primary tumours from metastatic colorectal adenocarcinoma.11,29,30
There are very limited data on CDX2 immunoreactivity in GCTs, and the studies published so far had small numbers of cases. For example, one study examined 52 malignant GCTs for CDX2 expression. 20 CDX2 positivity was detected in four (100%) out of four cases of teratoma, three (38%) out of eight cases of yolk sac tumour, three (38%) out of eight cases of embryonal carcinomas, two (100%) out of two cases of choriocarcinoma and only one (3%) out of 29 cases of seminoma. 20 As the percentage of positive cases was quite high, the authors concluded that CDX2 expression in a metastatic tumour with an unknown primary should not only point to a colorectal origin, but also testicular GCT. 20 However, in the present study CDX2 positivity was determined in 27 (52%) of 52 tumours with a teratoma component, two (9%) of 22 tumours with a yolk sac tumour component, and one (2%) of 50 tumours with a seminoma component. This present study not only evaluated the tumours located in the testes, but also those at metastatic sites. At both sites, the only strong and diffuse CDX2 positivity was in the mature colonic epithelium of teratomas. The previous study scored positive cases as 2+ and 3+, but the scoring system was not described clearly in the article. 20 This causes confusion when interpreting the extent of the CDX2 positivity. In the present study, because the other positively-stained tumours (two yolk sac tumours and one seminoma) only demonstrated weak and focal staining, only teratomas with colonic epithelium were considered to have positive immunoreactivity for CDX2. In contrast to the previous study, 20 the findings of the present study suggest that CDX2 positive immunoreactivity is unique for colorectal epithelium. The inconsistency between these present findings and those of the previous study might result from the different interpretations of the immunohistochemical staining patterns. 20
In a series of 40 cases of testicular GCT and eight cases of metastatic GCT that were evaluated for CDX2 immunoreactivity, none of the tumours showed positivity except the yolk sac tumours (12 of 26) and the teratoma components with mature colonic epithelium (seven of 14). 31 Only the extent of the staining was evaluated in that study. 31 While positive yolk sac tumours were mostly interpreted as 1+ (1% to 25%), the report did not state what the score was for the CDX2-positive teratomas. In the present study, there were also yolk sac tumours (two of 22 tumours) showing positive immunostaining, and similar to the previous study, 31 the staining was focal rather than diffuse. In addition, the present study demonstrated that the staining intensity was weak in both of the yolk sac tumours.
In another study, CDX2 positivity of 26 ovarian and testicular yolk sac tumours was evaluated in association with the pattern of tumour growth. 23 All cases with classical patterns showed positivity only in isolated epithelial cells, and staining was patchy in those displaying a somatic glandular pattern. 23 The authors demonstrated diffuse positivity in only two yolk sac tumours with a somatic glandular pattern. 23 Unfortunately, the intensity of staining was not defined in the study. 23
In a study of 124 cases, CDX2 positivity was more frequent in glandular yolk sac tumours (83%) than in adenocarcinomas (63%) arising from GCTs. 32 Despite the fact that the majority of the adenocarcinoma cases (64%) showed no colonic/enteric differentiation morphologically, the rate of CDX2 positivity was quite high, as for the glandular yolk sac tumours. 32 These high rates of CDX2 positivity were striking and may be due to the methodology used to evaluate the staining pattern, as tumours were defined as positive when > 10% of the tumour cells were stained.
Although it was incompatible with the results of the present study, a recent study of 93 testicular GCTs, two primitive neuroectodermal tumours, eight Leydig cell tumours and six Sertoli cell tumours, suggested that CDX2 was a sensitive and relatively specific marker for yolk sac tumours. CDX2 immunostaining was uniformly and strongly positive in the epithelioid cells of all yolk sac tumours (n = 25). Similar to the findings of the present study, glands with intestinal differentiation in teratomas were also CDX2 positive in 11 of 14 cases. 33
The findings of most of these studies give rise to the idea that CDX2 positivity in testicular GCTs could be a potential pitfall when examining metastatic tumours of unknown primary origin.20,23,31,32 Nevertheless, the present study had a higher number of GCT cases and the staining pattern was described in more detail compared with previous studies.20,23,31,32 Hence, these current findings suggest that tumours should only be considered to have positive CDX2 immunoreactivity if the extent of the staining is diffuse and the intensity is strong.
In conclusion, this present study demonstrated strong and diffuse immunostaining for CDX2, as is observed in colorectal adenocarcinomas, only in the mature colonic epithelium found in teratomas in testicular GCTs. CDX2 positivity in other tumour types was infrequent, and presented as weak and focal staining. These current findings suggest that CDX2 immunoreactivity should be interpreted by considering both the staining intensity and the extent of staining so as not to cause misdiagnosis. If a metastatic tumour with an unknown primary tumour shows prominent CDX2 immunoreactivity, aside from colorectal adenocarcinoma, a teratoma with colonic-type epithelium should be considered in the differential diagnosis instead of other GCTs.
Footnotes
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
