Abstract
Objective
To evaluate whether polymorphisms in the gamma-aminobutyric acid A receptor α1 subunit (GABRA1) gene influence sleep induction time, bispectral index score (BIS) during sleep induction and the total dose of midazolam required to reach a Ramsay Sedation Assessment Scale (RSAS) score of 4.
Methods
Patients scheduled for elective orthopaedic surgery were enrolled. All patients received initial doses of 0.02 mg/kg intravenous midazolam. If the RSAS score did not reach 4, an additional 1-mg dose of midazolam was administered. Results were compared among groups of patients with five single-nucleotide polymorphisms (SNPs) in GABRA1: rs4263535, rs980791, rs6556562, rs998754 and rs2279020.
Results
A total of 104 patients were evaluated. Polymorphism rs4263535 was associated with the lowest BIS during sedation induction. Multinomial logistic regression analysis demonstrated that polymorphism rs4263535 was significantly associated with the total dose of midazolam required for sedation induction.
Conclusions
Polymorphism rs4263535 in GABRA1 intron 4 was associated with deeper sedation by intravenous midazolam. Patients with the A/A rs4263535 genotype required a smaller dose of midazolam.
Introduction
Midazolam differs from other sedative agents in its more rapid onset and shorter duration of action. 1 Clinical effects are usually observable 2–3 min after intravenous midazolam administration. 1 Comparative studies have shown that midazolam at a dose of 0.05–0.12 mg/kg produces satisfactory sedation and operating conditions as effectively as the administration of 0.06–0.2 mg/kg diazepam in patients undergoing gastroscopy, dental implantation, cataract surgery or spinal anaesthesia.2–10 Compared with diazepam, the onset of action of midazolam is usually more rapid and the degree of amnesia is greater, but the speed of onset of the effect of midazolam without premedication varies between individuals. 11 Although this inter-individual variability is reduced at higher doses of the drug, 4 these differences reduce the predictability of sedation induction using midazolam. The inter-individual variability in sedation induction time is likely influenced by several factors, including dose, rate of administration, premedication status, age, sex and American Society of Anesthesiologists (ASA) physical status score. 11 In addition, sensitivity to benzodiazepines such as midazolam is influenced by complex biochemical interactions that are supported by genetic studies.12–15
Benzodiazepine-sensitive gamma-aminobutyric acid A (GABA-A) receptors are composed of an α-subunit variant (α1, α2, α3, or α5), a β-subunit (β1–3), and a γ-subunit (γ1–3) in triplet configurations including α1β2,3γ2, α2β2,3γ2, α3β2,3γ2, and α5β2,3γ2, and are regarded as the major mediators of benzodiazepine action in the brain. 16 The histidine residue in GABRA1 is characteristic of all major benzodiazepine-sensitive GABA-A receptors.17,18 Sedation, anterograde amnesia and the anticonvulsant effects produced by benzodiazepines are mediated via α1-containing GABA receptors, and anxiolysis and muscle relaxation are mediated by α2-containing GABA receptors. 14 Mutations to arginine residues in the α1, α2, α3, α5, β2, β3, and γ2 subunits can produce a complete loss of potentiation of GABA-induced currents, thereby reducing benzodiazepine sensitivity.12,19–21 Analyses of the pharmacological characteristics of recombinant GABA receptors demonstrated that mutations in the GABA receptor genetic sequences lead to benzodiazepine insensitivity. 12
This present study hypothesized that GABA-A receptor α1 subunit (GABRA1) polymorphisms would significantly influence the pharmacological effects (e.g. onset of sedation) of midazolam. The aim of the present study was to evaluate the association between all known GABRA1 nucleotide polymorphisms (rs4263535, rs980791, rs6556562, rs998754, and rs2279020) and sensitivity to midazolam. The primary experimental endpoint was the analysis of the association between GABRA1 SNPs with the dose of midazolam required to reach a Ramsay Sedation Assessment Scale (RSAS) score of 4. Secondary experimental endpoints were the analyses of the association between each polymorphism with the time-to-reach an RSAS score of 4 (induction time) and the lowest bispectral index score (BIS) measured during sedation induction.
Patients and methods
Patients
Patients aged 20–40 years, scheduled for elective orthopaedic surgery and classified as having ASA physical status 22 I or II, were enrolled sequentially in the study in the Department of Anaesthesiology and Pain Medicine, College of Medicine, Korea University, Seoul, Republic of Korea, between August 2011 and July 2012. Patients with psychiatric disorders, neurological disorders, hypothermia (<35℃), those using medications that could influence the level of sedation (such as neuromuscular blocking agents, and/or those with a history of drug abuse) were excluded. In addition, any patient who received adrenaline was also excluded because it can influence BIS. 23 The study was approved by the Korea University Hospital Institutional Review Board, Seoul, Republic of Korea (no. ED11137) and the Clinical Research Information Service, Cheongju-si, Chungcheongbuk-do, Republic of Korea (no. KCT0000848). Written informed consent was obtained from each patient for study participation and genetic analysis.
Sedation
After patients arrived at the operating room, electrocardiography, respiratory rate, noninvasive mean blood pressure, oxygen saturation, heart rate and BIS were monitored. Before initiation of sedation, 5 l/min oxygen was supplied using a facemask for 5 min. The anaesthesiologist (S.Z.Y.) asked patients to keep their eyes closed and not to roll during the study. Sedation was induced with midazolam without premedication. An initial bolus dose of 0.02 mg/kg midazolam was administered intravenously over a period of 5 s, and an anaesthesiologist (S.Z.Y.) observed each patient for a period of 3 min to evaluate the onset of sedation. 24 The endpoint for sedation was RSAS 4 (RSAS scores: 6, no response to stimulus; 5, sluggish response to stimulus; 4, brisk response to stimulus; 3, response to commands; 2, cooperative, properly oriented, and tranquil; 1, anxious, restless, or both). 25 RSAS 4 at BIS 60–80, indicating mild-to-moderate sedation, is considered an adequate sedation level for surgery. A brisk response to a light glabellar tap was consistent with the desired level of sedation. If the RSAS score did not reach 4 by 3 min after the initial bolus of midazolam, an additional 1-mg dose of midazolam was administered intravenously every 2 min until an RSAS score of 4 was achieved. The study was terminated if the oxygen saturation dropped below 95% and/or tachycardia, hypertension, or hypotension >20% greater than baseline values developed. The time-to-reach RSAS 4 and the lowest BIS during sedation induction were recorded. In addition, the initial midazolam bolus dose and the total additional amount of midazolam administered were recorded. After the study was completed, patients underwent general anaesthesia for the duration of surgery.
Genomic analysis
Primers used for polymerase chain reaction amplification and pyrosequencing of regions of the human gamma-aminobutyric acid A receptor α1 subunit gene.
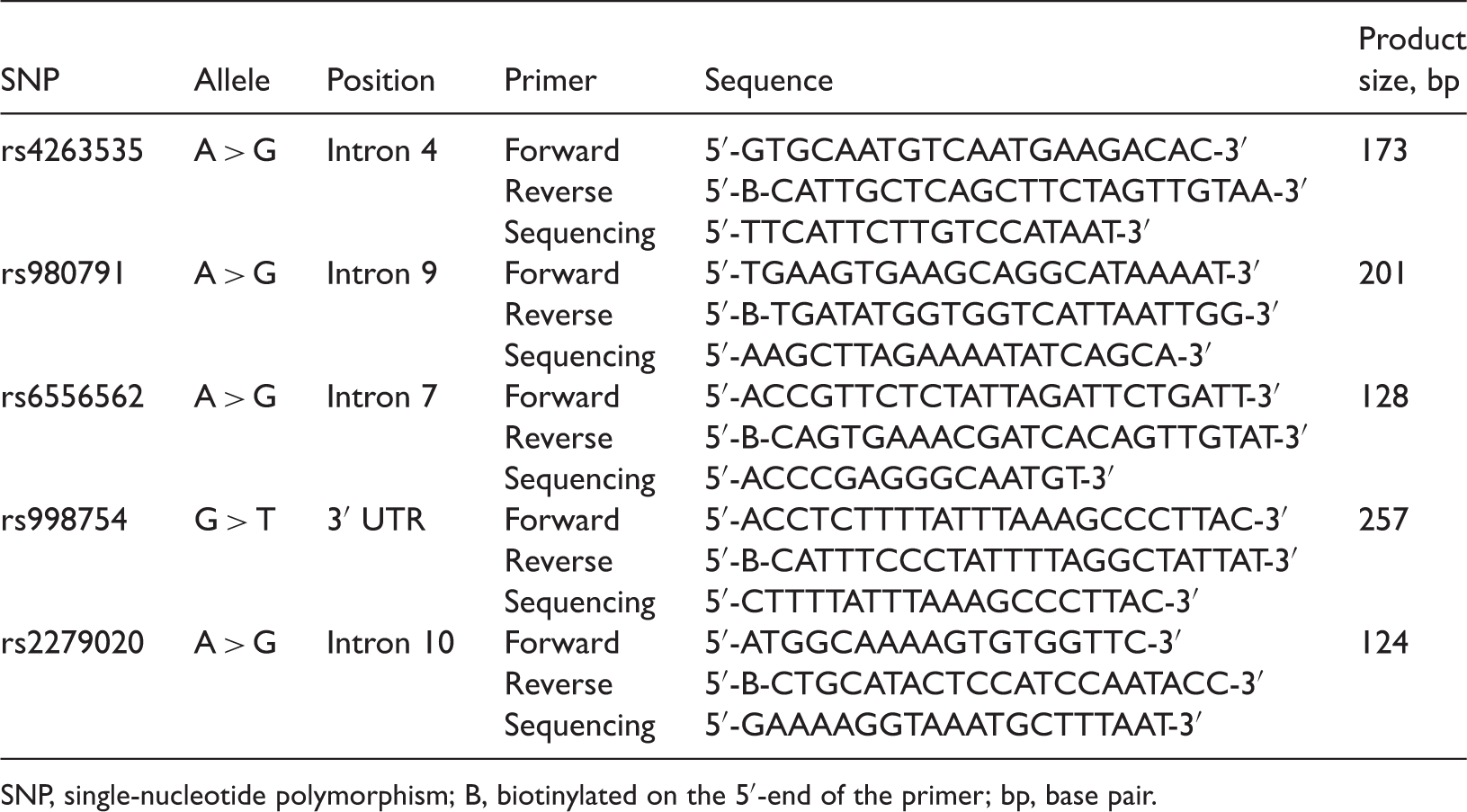
SNP, single-nucleotide polymorphism; B, biotinylated on the 5′-end of the primer; bp, base pair.
For the pyrosequencing, biotinylated PCR template (20 μl) was immobilized by incubation (with shaking at 1400 r.p.m. for 10 min at room temperature) with a mixture of 5 μl streptavidin beads (GE Healthcare Biosciences, Uppsala, Sweden) in 40 μl PyroMark Binding Buffer (Qiagen Korea, Seoul, Republic of Korea). For primer annealing, 40 μl PyroMark Annealing Buffer (Qiagen Korea) containing 0.4 μM sequencing primer was incorporated into each well. The bead–PCR product was transferred onto a probe with a Qiagen Vacuum Prep Workstation (Qiagen Korea), according to the manufacturer’s instructions. The captured beads on probes were incubated in 70% ethanol for 5 s and the solution was flushed through the filters for 5 s. The beads were then treated with a denaturing solution (0.2 M NaOH) that was flushed through the filters for 5 s. A wash buffer (10 mM Tris-acetate, pH 7.6) was used to rinse the beads for 5 s. All liquid was completely drained from the probes, after which the beads were transferred into Pyromark™ Wash Buffer (Qiagen Korea) containing the sequencing primer, which was heated at 85℃ for 2 min then cooled to room temperature (15–25℃ for ≥5 min. Pyrosequencing was performed on a Qiagen PyroMark Q96 ID instrument (Qiagen Korea) according to the manufacturer’s instructions.
Statistical analyses
Correlation coefficients between continuous variables such as body mass index (BMI), total midazolam dose, time to reach RSAS 4, and lowest BIS during sedation induction were obtained using Spearman’s rank correlation analysis. Deviation of the frequency of polymorphisms from the Hardy–Weinberg equilibrium was assessed using the χ2-test.
Median differences in total midazolam dose, time to reach RSAS 4, and lowest BIS during sedation induction between the groups with various GABRA1 polymorphisms were evaluated using the Kruskal–Wallis test. The Mann–Whitney U-test was performed to evaluate differences between the A/A, G/A, and G/G genotypes, and P-values were corrected with the Bonferroni correction according to the number of comparisons.
Multinomial logistic regression was applied to whole-genome single-nucleotide polymorphism (SNP) data to find SNPs associated with the responses of interest. Factors were identified that related to midazolam sensitivity and GABRA1 polymorphisms at rs4263535 using multinomial logistic regression analysis with total midazolam dose, lowest BIS during sedation induction, time-to-reach RSAS 4, age, sex, BMI and ASA physical status as covariates. However, the associations of age, sex, BMI, and ASA physical status with the effects of midazolam in patients with various GABRA1 genotypes were not statistically significant. Therefore, only midazolam dose, lowest BIS during sedation induction, and time to reach RSAS 4 were considered as risk factors using the backward variable selection method.
The required sample size for this study was determined based on a pilot study on the effect of the rs4263535 genotype because no data were available on the effect of midazolam in patients with varying GABRA1 genotypes. A power analysis was performed using G*Power software (version 3.1; Dusseldorf University, Dusseldorf, Germany) to determine whether the population means of the total dose of midazolam for the three assessed genotypes were equivalent. 26 The minimum sample size was determined to be 104 based on an α-value of 0.05, a power of 0.80, and an effect size of 0.313 (calculated from the pilot data from 28 patients). Assuming a 10% loss to follow-up observations, 114 patients were calculated to be required.
Statistical significance was defined as P < 0.05 and the statistical analyses were performed with SPSS® software, version 20.0 (SPSS Inc., Chicago, IL, USA) for IBM computers (IBM, Armonk, NY, USA). Calculations of allele frequency and heterozygosity using the Hardy–Weinberg equilibrium test were performed using R software version 2.15.2 (The R Foundation for Statistical Computing, Vienna, Austria).
Results
A total of 104 patients (42 male and 62 female) were enrolled in this study. A flow diagram of the progression through the study is shown in Figure 1. Patients who refused to participate or those undergoing psychiatric management were excluded (n = 10). Characteristics of included patients are presented in Table 2. Correlations between BMI, total dose of midazolam, lowest BIS during sedation induction, and time to reach RSAS 4 are shown in Table 3. Total dose of midazolam was positively correlated with time to reach RSAS 4 (r = 0.781, P < 0.001) and BMI (r = 0.204, P = 0.041), but not with BIS during sedation induction (r = 0.165).
Flow diagram of the progression of patients undergoing routine elective orthopaedic surgical procedures through the study. Characteristics of patients (n = 104) undergoing routine elective orthopaedic surgical procedures who were enrolled in the present study. Values presented as mean ± SD, median (range) or n (%). Between-group differences in continuous variables were analysed using Student’s t-test or Mann–Whitney U-test. Differences in categorical variables were analysed using χ2-test or Fisher’s exact test as appropriate. ASA, American Society of Anesthesiologists physical status; BMI, body mass index; BIS, bispectral index; RSAS, Ramsay Sedation Assessment Scale; NS, no statistically significant difference (P ≥ 0.05). Results of the Spearman’s rank correlation coefficient analysis to test the association between two ranked patient characteristics. P < 0.05 when data were analysed using Spearman’s rank correlation coefficient. BIS, bispectral index; RSAS, Ramsay Sedation Assessment Scale; BMI, body mass index.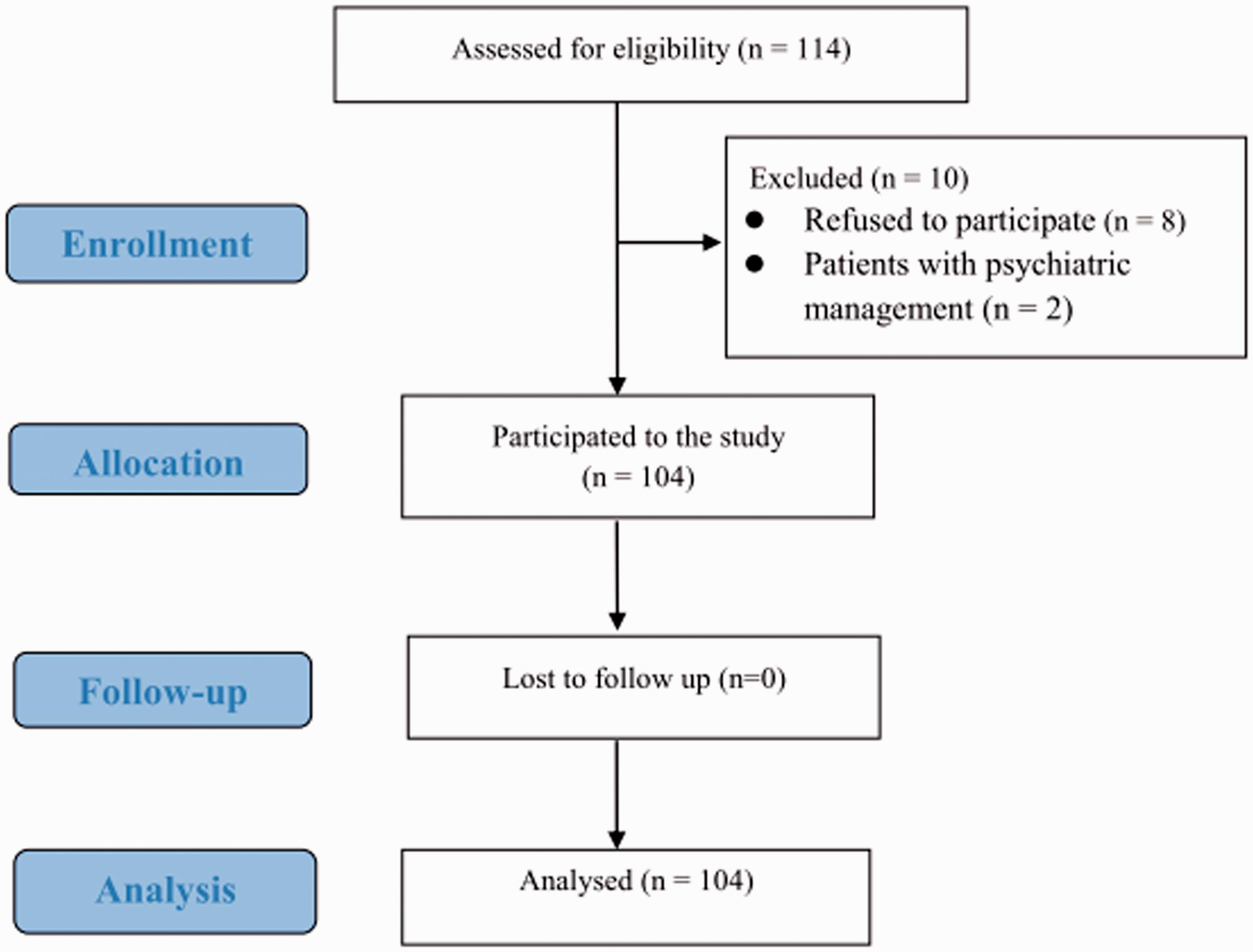


Genotypes with allele frequencies and heterozygosity of the human gamma-aminobutyric acid A receptor α1 subunit gene polymorphisms.

Values were presented as n (%).
SNP, single-nucleotide polymorphism.
All P-values for the Hardy–Weinberg Equilibrium were not significant (P ≥ 0.05).
Evaluation of the equivalence of the population medians of total midazolam dose, lowest bispectral index (BIS) during sedation induction, and time to reach Ramsay Sedation Assessment Scale (RSAS) 4 across the three evaluated single-nucleotide polymorphism (SNP) genotypes of the human gamma-aminobutyric acid A receptor α1 subunit gene using the Kruskal–Wallis test.
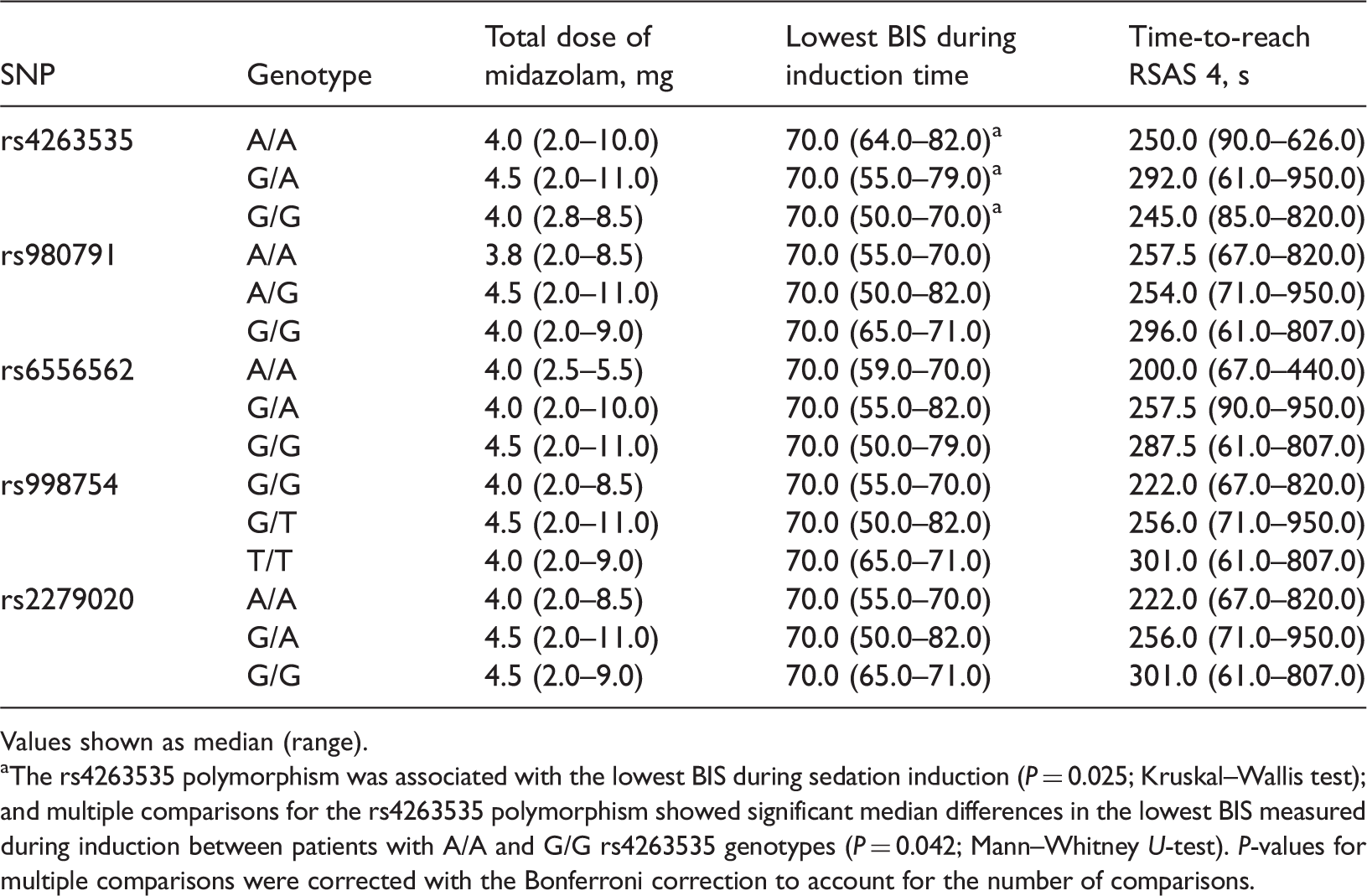
Values shown as median (range).
The rs4263535 polymorphism was associated with the lowest BIS during sedation induction (P = 0.025; Kruskal–Wallis test); and multiple comparisons for the rs4263535 polymorphism showed significant median differences in the lowest BIS measured during induction between patients with A/A and G/G rs4263535 genotypes (P = 0.042; Mann–Whitney U-test). P-values for multiple comparisons were corrected with the Bonferroni correction to account for the number of comparisons.
Multinomial logistic regression analysis of the association between the human gamma-aminobutyric acid A receptor α1 subunit gene polymorphism rs4263535 genotypes with other factors.
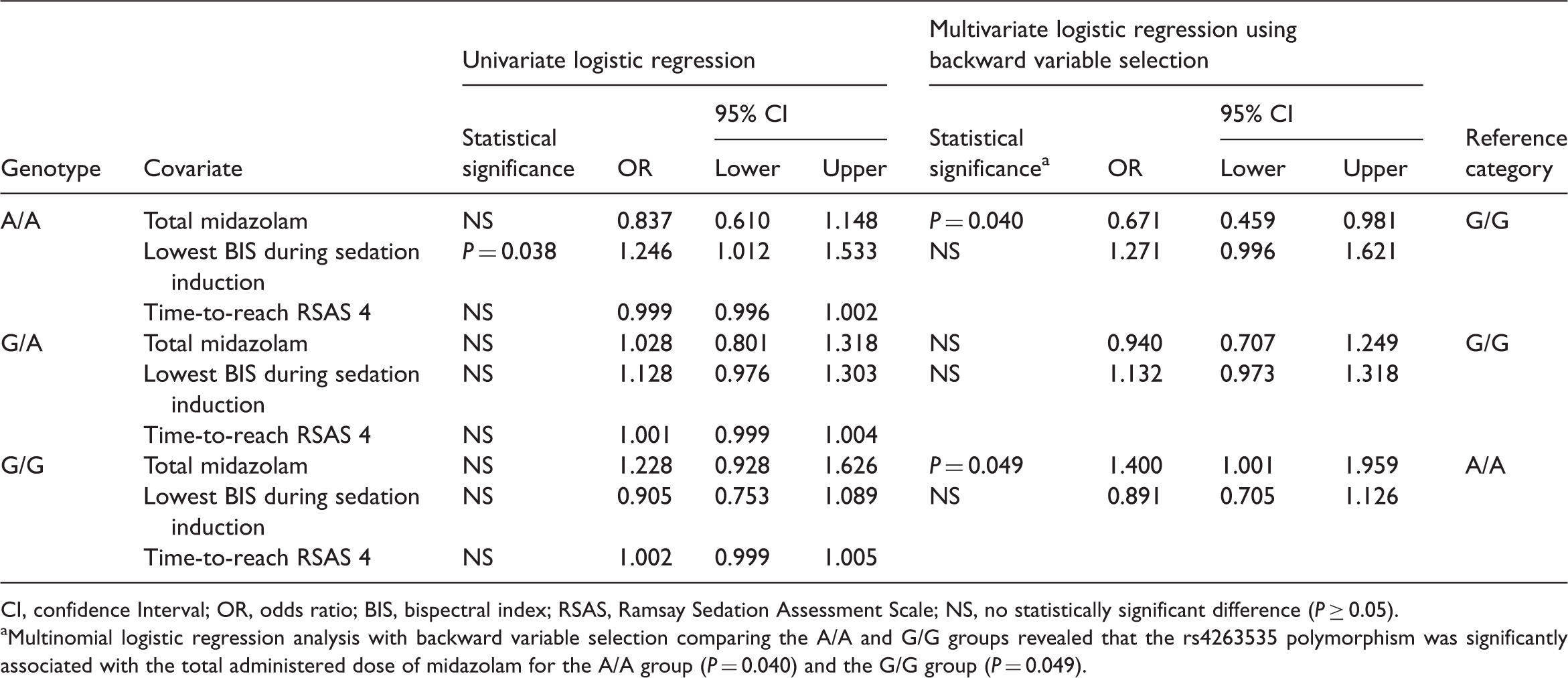
CI, confidence Interval; OR, odds ratio; BIS, bispectral index; RSAS, Ramsay Sedation Assessment Scale; NS, no statistically significant difference (P ≥ 0.05).
Multinomial logistic regression analysis with backward variable selection comparing the A/A and G/G groups revealed that the rs4263535 polymorphism was significantly associated with the total administered dose of midazolam for the A/A group (P = 0.040) and the G/G group (P = 0.049).
Discussion
This study is the first to evaluate the influence of genetic variation on the induction time for midazolam sedation. Current results show that the polymorphism rs4263535 in intron 4 of the human GABRA1 gene is associated with deeper sedation by intravenous midazolam, compared with other polymorphisms in this gene. Furthermore, this current study showed that patients with the A/A rs4263535 GABRA1 genotype required smaller doses of midazolam to achieve sedation, and showed a lower BIS value during sedation induction, compared with patients with the G/G rs4263535 GABRA1 genotype.
Benzodiazepines such as midazolam have hypnotic, sedative, anxiolytic, amnesic, anticonvulsant and centrally produced muscle relaxant properties. 2 The mechanism of action of benzodiazepines is reasonably well understood and does not involve release, synthesis, or altered metabolism of GABA.1–3 Instead, benzodiazepines potentiate the inhibitory action of GABA by increasing the flow of chloride ions through ion channels, which reduces the ability of the affected cell to generate an action potential. 7 The hypnotic effect of midazolam is known to be associated with GABA accumulation and benzodiazepine receptor occupation. 27 Midazolam interferes with GABA reuptake, which results in GABA accumulation. 4 In addition, the occupation of GABA receptors with benzodiazepines such as midazolam increases membrane hyperpolarization and neuronal inhibition.12,28 Midazolam also causes phasic inhibition by prolonging inhibitory post-synaptic currents, and its action at extra-synaptic GABA-A receptors produces tonic inhibition, which is a continuous increased inhibitory current.5,29 These midazolam-induced changes lead to neuronal hyperpolarization that reduces neuronal excitability. 29 In addition, midazolam reduces thalamic and cerebral cortical blood flow, as well as metabolic activity, in those regions. 12 These well-established effects of midazolam in the brain produce altered neuronal activity that changes the level of consciousness of patients administered this drug. 29
Research has suggested that mutations and polymorphisms in GABRA1 are associated with alcohol-related behavioural alterations including excitement, sleep, withdrawal symptoms and dependence. 30 A polymorphism in GABRA1 intron 9 (rs980791) has been associated with alcohol dependence (P = 0.01) and susceptibility to blackout (P < 0.01). 31 A polymorphism in GABRA1 intron 4 (rs4263535) has been associated with the age of first drunkenness (P < 0.01), and a polymorphism in GABRA1 intron 10 (rs2279020) has been associated with blackout (P = 0.02) and alcohol dependence (P = 0.03). 31 Essential tremors, 32 juvenile myoclonic epilepsy, 33 autism 34 and schizophrenia 35 have also been associated with GABRA1 mutations. In the same manner, GABRA1 polymorphism rs4263535 seems to be associated with determining the pharmacodynamics and pharmacokinetics of benzodiazepine in this study.
As shown in Table 5, the A/A rs4263535 GABRA1 genotype was associated with the lowest BIS measured during sedation induction in this study. However, the total dose of midazolam required to reach RSAS 4 did not vary significantly among the rs4263535 genotypes, indicating that the level of sedation, as estimated by BIS, in all patients after an identical dose of midazolam may not be an equivalent effect. Patients reached the same RSAS after midazolam administration, but the level of consciousness as measured by the BIS was likely to be higher in those with the A/A rs4263535 GABRA1 genotype, suggesting that such patients were more resistant to the effects of midazolam.
The level of consciousness of a patient is generally assessed by subjective clinical scales such as the observer assessment of alertness/sedation score or the RSAS score,36,37 which broadly categorize sedation levels as mild, moderate, or deep. More recently developed BIS methodology has provided an additional means of assessing the level of consciousness of a patient: the BIS is regarded as a valuable addition to the clinician’s repertoire of methods for monitoring the level of consciousness in patients undergoing midazolam sedation.7–10,15 BIS monitoring provides an objective real-time measurement of the depth of anaesthesia via a weighted sum of electroencephalographic subparameters that are transformed into numeric values ranging from 0 (no electrical activity in the brain) to 100 (fully conscious). As the state of consciousness evolves from awake to sedated, or exhibits burst suppression, the BIS decreases. As an anaesthesia guideline, a BIS of 80–90 is considered to be indicative of conscious sedation, a BIS of 60–80 is indicative of moderate or deep sedation that produces anxiolysis and/or analgesia, 38 and a BIS of 40–60 is required for maintenance of general anaesthesia. 39 BIS values observed during the present study were consistent with increased brain activity indicative of higher levels of consciousness in patients with the A/A genotype for polymorphism rs4263535, compared with patients with the G/G genotype for this polymorphism.
This present study had some limitations. Various factors affect benzodiazepine pharmacodynamics, and the effect or function of a drug is related to serum levels of the drug at its site of action, which can be estimated by pharmacokinetic methodology. Using plasma concentration data and pharmacokinetics simulations, it has been estimated that a benzodiazepine receptor occupancy of <20% may be sufficient to produce an anxiolytic effect, while sedation is observed with 30–50% receptor occupancy, and unconsciousness requires ≥60% occupancy. 40 Therefore, lipophilicity, receptor affinity, density, occupancy and receptor reserves all affect the pharmacodynamics of benzodiazepines. In addition, metabolic stability, heterogeneity of other receptors, distribution of receptors in the brain and receptor pathology can also influence the effects of medications. In addition, any other genetic variations that affect the pharmacokinetics of midazolam need to be considered. The pharmacokinetics of midazolam might affect the amount of midazolam that reaches the receptor, therefore it might also have an effect on the intensity of sedation. Generally, high-frequency electroencephalography (EEG) activity decreases as consciousness level decreases in drug-free patients. However, sedative benzodiazepines increase beta activity.41,42 In this present study, it was not possible to analyse the beta ratio, signal quality index and an electromyography level in patients because BIS data were collected manually. In future, EEG analysis will be used to more precisely evaluate the effect of GABRA1 polymorphisms on the pharmacodynamics of midazolam. In addition, this present study may have been underpowered because the minimum sample size was determined to be 114, but only 104 patients were analysed.
In conclusion, results of the present study show that the rs4263535 polymorphism in intron 4 of the human GABRA1 gene was associated with deeper sedation by intravenous midazolam, and patients with the A/A rs4263535 genotype required smaller doses of midazolam. Thus, patients with the A/A genotype might require a smaller dose of midazolam to achieve RSAS 4 during the sedation induction period, compared with other patients. This finding should contribute to the ability of clinicians to induce optimal anaesthesia with midazolam by allowing them to customize dosing regimens based on patient genotypes.
Footnotes
Acknowledgements
This report contains a portion of the doctoral dissertation of Shin Yuong Lee.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI13C2181). No funding other than personal was used in conducting the audit as well as writing the manuscript.
