Abstract
Objective
The performance of cardiac indicators as determinants of stroke outcomes remains debatable, therefore the present study focused on the short-term prognostic value of cardiac indicators in patients who had experienced acute ischaemic stroke.
Methods
Consecutive patients with acute ischaemic stroke were enrolled in a prospective stroke registry. Data on demographics, clinical characteristics and cardiac indicator levels (including troponin I, creatine kinase-MB, myoglobin and brain natriuretic peptide) were prospectively collected. Receiver operating characteristic curves and binary logistic regression models were used to examine the performance of variables in predicting poor functional outcomes (modified Rankin Scale score 2–6).
Results
Out of 337 patients with acute ischaemic stroke, myoglobin had the greatest power for predicting poor outcomes among the cardiac indicators. Forward stepwise multivariate logistic regression analysis revealed that myoglobin could independently predict short-term outcomes. Spearman’s rank correlation tests indicated a positive correlation between myoglobin levels and stroke severity.
Conclusions
Among cardiac indicators, myoglobin may be an independent predictor of short-term outcomes in patients with acute ischaemic stroke.
Introduction
Acute ischaemic stroke is one of the leading causes of disability and death worldwide.1,2 Primarily caused by atherothrombotic changes to the cerebrovascular system, a high proportion of acute ischaemic stroke is found to have a cardioembolic origin, indicating strong links with heart disease. 3 Moreover, sympathetic activation in acute ischaemic stroke is reported to have serious adverse effects on cardiac function 4 and in turn, cardiac complications increase the risk of death. 5 In the 20th century, cardiac complications accounted for almost 20% of mortality in patients with acute ischaemic stroke. 6 Successful prediction of cardiac complications in the early phase of acute ischaemic stroke would, therefore, have a positive impact on its clinical management, and interventions such as cardiac augmentation, may benefit a subset of patients with stroke. 7
Cardiac indicators in acute ischaemic stroke were first documented in the late 1970s. 8 Despite the fact that cardiac indicator levels increase in the initial phase of stroke, their performance as predictors of poor outcomes in patients with acute ischaemic stroke remains unclear. One study demonstrated that troponin I and troponin T (which are considered to be the most accurate biomarkers of myocardial necrosis) may help predict cardiac complications and death in acute ischaemic stroke. 9 Another investigation however, showed that troponin levels did not influence clinical outcomes if other risk factors were taken into account. 10 In contrast to the viewpoint that elevated creatine kinase-MB levels represent biological markers for stroke-related myocardial injury, 8 creatine kinase-MB elevations in stroke patients may be noncardiac in origin. 11 Studies12,13 have found elevated myoglobin levels in patients with all types of acute stroke, however, data regarding the value of the myoglobin level as a prognostic indicator of cardiac complications and the short-term outcomes of acute ischaemic stroke remain limited.
The current prospective, observational study focused on the short-term prognostic significance of admission levels of cardiac indicators (troponin I, creatine kinase-MB, myoglobin and brain natriuretic peptide) in a cohort of consecutive patients with acute ischaemic stroke.
Patients and methods
Study population
Patients with acute ischaemic stroke, aged >18 years, admitted to the Emergency Department, Xinhua Hospital, affiliated to Shanghai Jiaotong University School of Medicine, Shanghai, China, were consecutively recruited between January 2011 and October 2012. Acute ischaemic stroke was further confirmed using a magnetic resonance imaging scanner with 1.5 T (Magnetom Symphony; Siemens Medical Systems, Erlangen, Germany). All aetiologies were determined according to the Trial of ORG 10172 in Acute Stroke Treatment criteria, 14 after an initial aetiological work-up that included transcranial carotid duplex sonography and 12-lead electrocardiography.
Patients meeting the following criteria were excluded: (I) history of recent coronary artery disease, or heart failure within the preceding 3 months; (II) renal insufficiency (creatinine >97 µmol/l); (III) skeletal muscle disease; or (IV) insufficient blood-sample volume for the measurement of cardiac indicator levels.
The study was approved by Shanghai Jiaotong University Xinhua Hospital Ethics Committee and was conducted in accordance with the Declaration of Helsinki. Since the present observational study comprised laboratory indices that are commonly measured for all patients in the Emergency Department of Xinhua Hospital, written informed consent for patient participation in this study was not required by the ethical review board.
Blood measurements
All blood samples were collected within a median 20 h (interquartile range, 15–22 h) of stroke onset and before any treatment was administered, to avoid drug-biomarker interference. Venous blood (3 ml) was drawn into an ethylenediaminetetra-acetic acid-containing tube (BD Vacutainer®, Becton, Dickinson and Company, Plymouth, UK) and centrifuged at 1 500 g for 15 min at room temperature. The plasma was then stored at −80℃ until analysis. Serum troponin I, creatine kinase-MB, myoglobin and brain natriuretic peptide levels were measured using an electrochemiluminescent immunoassay with Access AccuTnI Reagent, Access AccuCK-MB Reagent, Access AccuMyoglobin Reagent, and Access AccuBNP Reagent, respectively (all from Beckman Coulter, Brea, CA, USA). In all of the assays, both the intra- and inter-assay coefficients of variation were ≤10.0%. The normal concentration ranges for the laboratory used in the present study were as follows: troponin I, 0–0.06 ng/ml; creatine kinase-MB, 0–25 ng/ml; myoglobin, 0–70 ng/ml; brain natriuretic peptide, 0–100 ng/ml.
Study outcomes
Demographic and clinical characteristics, including history of symptomatic cardiovascular disease and prevalence of vascular risk factors, were prospectively collected at baseline. Neurological impairment and physical function were evaluated by stroke neurologists (X.J.C., H.R.W.) on admission, and on day 1 and day 7 following admission, using the National Institutes of Health Stroke Scale (NIHSS) score. Short-term clinical progress was defined as the difference between NIHSS score on admission (NIHSS0) and NIHSS score on day 7 (NIHSS7) i.e. Δ-NIHSS = NIHSS0 − NIHSS7.15,16 A positive Δ-NIHSS value indicated clinical improvement; a negative value indicated clinical worsening. Follow-up assessments were conducted with patients who survived the acute phase and were discharged home or to an inpatient rehabilitation facility. Patients’ functional outcomes were assessed using the modified Rankin Scale (mRS) score on day 90 via telephone calls. 17 An excellent outcome was defined as mRS score 0–1 and a poor outcome as mRS score 2–6.
Statistical analyses
Continuous variables were presented as mean ± SD or median (interquartile range), and categorical variables were presented as percentages. The statistical significance of between-group differences were compared using unpaired Student’s t-test or Mann–Whitney U-test for continuous variables, and Pearson’s χ2-test for categorical variables. Receiver operating characteristic curves were created to determine the prediction sensitivity and specificity of the variables. Univariate logistic regression analyses were performed to examine the relationship between unfavourable outcomes and each of the predictors, separately. Forward stepwise multivariate logistic regression analyses were conducted to determine the independent predictors of poor outcomes. Correlations between cardiac indicators and NIHSS score were analysed using Spearman’s rank correlation coefficient. A two-sided P value <0.05 was considered statistically significant. All analyses were performed using IBM SPSS® Statistics software, version 19.0 (SPSS, Chicago, IL, USA).
Results
Baseline characteristics of the study population
Baseline clinical and laboratory characteristics of patients aged >18 years with acute ischaemic stroke, organized according to clinical outcomes on day 90.
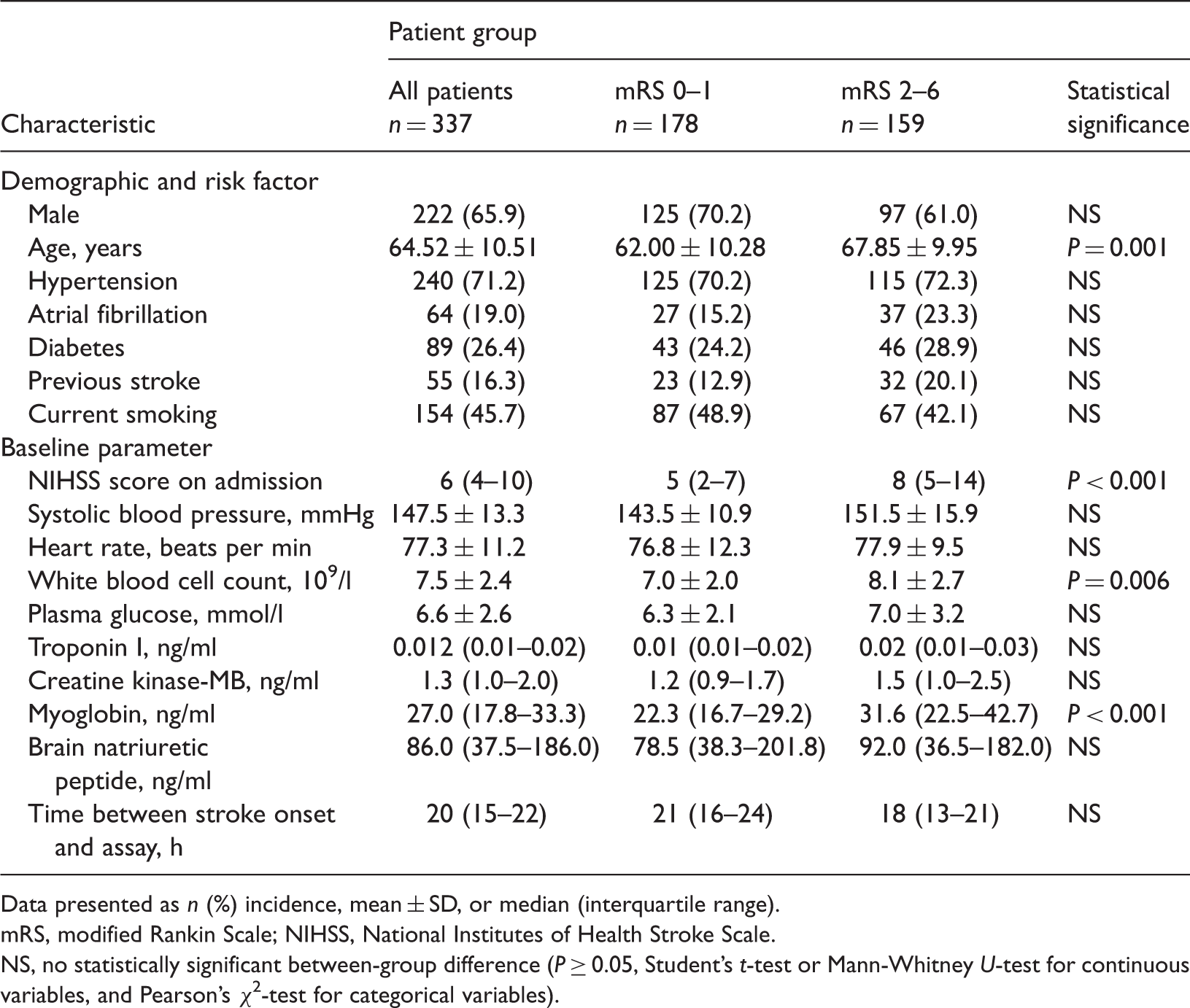
Data presented as n (%) incidence, mean ± SD, or median (interquartile range).
mRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale.
NS, no statistically significant between-group difference (P ≥ 0.05, Student’s t-test or Mann-Whitney U-test for continuous variables, and Pearson’s χ2-test for categorical variables).
The distribution of ischaemic stroke subtypes was as follows: large-artery atherosclerosis, 94 (27.89%); small vessel occlusion, 32 (9.50%); cardioembolism, 56 (16.62%); stroke of undetermined aetiology, 27 (8.01%); and other determined aetiology, 128 (37.98%). There were no statistically significant differences in the distribution of cardiac indicator levels between the different stroke subtypes, and no statistically significant differences in the cause of stroke between patients with different outcomes.
Value of cardiac indicators in predicting short-term outcomes
Patients with poor outcomes demonstrated significantly higher myoglobin levels compared with those with excellent outcomes. Spearman’s rank correlation coefficient indicated a strong positive correlation between myoglobin levels on admission and mRS score on day 90 (r = 0.344; P < 0.001). Receiver operating characteristic curves were therefore constructed, and the area under the curve (AUC) was calculated, to examine the performance of cardiac indicators as predictors of short-term outcomes in patients with acute ischaemic stroke. Among the cardiac indicators, myoglobin had the largest AUC (0.736 ± 0.045; P < 0.001), demonstrating the greatest power to predict poor outcomes (Figure 1). Myoglobin ≥27.15 ng/ml showed a sensitivity of 69.1% and a specificity of 67.1%. The AUC for troponin I (0.595 ± 0.051) was less than that for myoglobin but greater than that for creatine kinase-MB (0.589 ± 0.052). Univariate logistic regression analyses were also performed to examine the correlations of each indicator with short-term outcomes, and the odds ratio (OR) for each variable was calculated. Increased myoglobin levels (OR 1.047, 95% confidence interval [CI] 1.018, 1.077) were associated with poor outcomes (Table 2).
Receiver operating characteristic (ROC) curves for troponin I (TNI), creatine kinase-MB (CKMB), and myoglobin (MYO) measured in 337 patients with acute ischaemic stroke. Among the cardiac indicators, myoglobin had the largest area under the curve of 0.736 ± 0.045 (P < 0.001), demonstrating the greatest ability to predict poor outcomes. Univariate odds ratios (OR) and 95% confidence intervals (CI) of variables for predicting poor outcomes in 337 patients aged >18 years, with acute ischaemic stroke. Odds ratio (OR) indicates the risk of obtaining poor outcomes (mRS 2–6). NIHSS, National Institutes of Health Stroke Scale.

Independent predictive value of myoglobin levels
When the observed baseline variables (Table 2) were included in a forward stepwise multivariate logistic regression model in which poor outcomes were the dependent variable, baseline NIHSS score (OR 1.202, 95% CI 1.113, 1.298) and myoglobin levels (OR 1.046, 95% CI 1.012, 1.081) were found to be the independent predictors of poor outcomes, whereas troponin I, creatine kinase-MB, and brain natriuretic peptide were not. Trends were also observed regarding older age (OR 1.058, 95% CI 1.015, 1.102) and higher white blood cell count (OR 1.216, 95% CI 1.015, 1.457) for predicting a poor prognosis.
Correlations of cardiac indicators with stroke severity and clinical progress
Correlations between cardiac indicators, stroke severity and clinical progress in 337 patients aged >18 years, with acute ischaemic stroke.

NIHSS, National Institutes of Health Stroke Scale; Δ-NIHSS, NIHSS on admission minus NIHSS on day 7; BNP, brain natriuretic peptide.
NS, no statistically significant correlation (P ≥ 0.05).
Discussion
Myoglobin is released rapidly during an acute myocardial infarction, and is a sensitive nonspecific indicator of myocardial injury, providing early sensitivity of 26–58% at presentation, 18 rising to 79–100% with serial assays. 19 Myoglobin has been proposed as the early marker for ‘rule-out’ protocols 20 included in commercially developed marker panels, 21 and advocated by the National Academy of Clinical Biochemistry panel as the most conveniently measured early marker. 22 Myocardial injury is an independent predictive factor for dependency and death following stroke,23,24 therefore, early recognition of myocardial injury using myoglobin may help towards rapid initiation of adequate therapy to improve clinical outcomes in patients with acute ischaemic stroke.
The present study, which examined levels of cardiac indicators in patients with acute ischaemic stroke, produced several novel findings. Myoglobin levels were significantly higher in patients with poor outcomes than in those with excellent outcomes. Myoglobin was also the most powerful and independent predictor of short-term outcomes among the cardiac indicators tested, and a positive correlation was demonstrated between myoglobin levels and stroke severity. In contrast to these results, another study showed no relationship between myoglobin levels and stroke outcomes, 25 however, the relatively small sample size (42 patients) in the earlier study may have affected its conclusions. Other research has shown that indicators such as N-terminal probrain natriuretic peptide, troponin I, and troponin T, were prognostic predictors of acute ischaemic stroke,9,26–28 however, the present study did not detect the prognostic significance of these indicators. The differing results between earlier studies and the present research may be attributed to two reasons. First, myoglobin was not included in some of these earlier studies, and therefore comparison of myoglobin with other indicators in predicting outcomes was not performed. Secondly, the definitions of adverse clinical outcomes differed among these studies. In one study, mRS and Barthel Index scores were treated as combined outcome measures; 27 another study determined in-hospital mortality as an endpoint, 26 while another used the composite of congestive heart failure ≥ class 3, acute myocardial infarction and recurrent stroke or any cause of death as unfavourable outcomes. 28
The mechanistic basis for myoglobin elevations correlating with poor outcomes in patients with acute ischaemic stroke remains unclear, although there are a variety of plausible explanations. Central activation of the sympathetic nervous system in acute ischaemic stroke is known to have serious adverse effects on cardiac function and myocardial structure,4,29 and to increase the release of myoglobin from injured myocardium, which may explain the propensity for adverse outcomes in patients with acute ischaemic stroke and increased myoglobin levels. In addition, myoglobin elevations may be a marker of unstable plaques in the coronary circulation, indicating a higher probability of an adverse prognosis due to recurrent myocardial ischemia following stroke.
Two phenomena occurred during the present study that suggest myoglobin elevations may be not derived from the myocardium. First, myoglobin levels were significantly higher in patients with poor outcomes than in those with excellent outcomes (median, 31.6 and 22.3 ng/ml, respectively), and both were within the normal range (0–70 ng/ml). Indeed, the degrees of myoglobin elevation in the present study did not meet the criterion of diagnosing stroke-related myocardial injury. Secondly, there were no significant elevations of troponin I and creatine kinase-MB, which are considered highly specific for myocardial injury. Other research has suggested that myoglobin elevations observed in patients with acute ischaemic stroke are likely to derive from damaged skeletal muscle. In patients with acute ischaemic stroke and concomitant acute respiratory failure requiring tracheostomy due to seriously impaired consciousness, biopsies of the sternothyroid muscle obtained during tracheostomy showed hypoxaemic changes (such as degenerating and regenerating fibres, ragged red fibres, and increases in acid phosphatase activity. 12 Negative myoglobin immunostaining in hemiplegic muscle and serum myoglobin elevations were seen in these patients, suggesting that serum myoglobin was likely to originate from skeletal muscles. Patients with higher negative myoglobin immunostaining had poor functional recovery of hemiplegia following stroke onset, implying that these muscular alterations may hamper functional recovery, and the extent of skeletal muscles lacking myoglobin may provide valuable information for predicting the functional prognosis of patients with acute ischaemic stroke. The true origin and underling mechanisms of myoglobin elevations in patients with acute ischaemic stroke and unfavourable outcomes may become the focus of the future studies.
Several limitations of the present study deserve comment. First, the study relied on a single baseline blood sample and could not account for the time course of the myoglobin variations. Secondly, the study lacked data regarding the possible influence of brain lesion location on the occurrence of myocardial damage. In particular, the involvement of the insular cortex is considered to be associated with a more frequent occurrence of cardiac complications following acute ischaemic stroke, probably via an increased sympathetic tone mediated by the cerebral autonomic centres. 30 Thirdly, as the present study only included patients with acute ischaemic stroke and did not include age- and sex-matched nonhospitalized healthy or at-risk control subjects, it could not be ascertained whether cardiac indicators were markedly elevated in patients with acute ischaemic stroke compared with healthy or at-risk controls. Fourth, the applicability of data collected at other centres was not assessed, thus the results derived from the present derivation cohort study could not be externally validated. Fifth, the use of a telephone survey to assess 90-day outcome may reduce the reliability of analytic results and increase the errors from outside interference. Finally, patients included in the present study were recorded as not having coronary artery disease or heart failure, based on their medical history. Since 38% of patients with acute ischaemic stroke are reported to have asymptomatic coronary artery disease, 31 some of the included patients may have had undiagnosed cardiac disease at the time of study entry. Given that the sample size of the study was so small, further large-scale studies are required to confirm these initial findings.
In conclusion, the present study suggests that myoglobin may be an independent predictor of short-term outcomes compared with other cardiac indicators, including troponin I and creatine kinase-MB. In addition, a positive correlation was observed between myoglobin levels and stroke severity. Clinicians could consider adding a myoglobin test to the admission work-up, to stratify patients with different prognoses or stroke severity in the early phase of acute ischaemic stroke, then target therapeutic interventions accordingly.
Footnotes
Acknowledgements
The authors thank all of the study collaborators, residents, neurologists, and nurses of our emergency department-based stroke unit and are especially grateful to all the study patients.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported by a grant from the Shanghai Health Bureau Scientific Research Foundation (Grant No. GWDTR201219).
