Abstract
Objectives
To investigate plasma total ghrelin and obestatin levels and the ghrelin/obestatin ratio prospectively, in hypertensive obese patients.
Methods
Height, weight, and waist and hip circumferences were measured in hypertensive and normotensive obese patients and matched healthy controls; the body mass index and waist to hip ratio were calculated. Fasting glucose and insulin levels were measured and the homeostasis model assessment of insulin resistance (HOMA-IR) was determined. Fasting ghrelin and obestatin concentrations were measured by radioimmunoassay and the ghrelin/obestatin ratio was calculated.
Results
A total of 38 hypertensive obese patients, 40 normotensive obese patients and 38 controls were enrolled. Hypertensive obese patients had lower plasma levels of ghrelin and obestatin than normotensive obese patients or controls. In addition, normotensive obese patients had lower plasma ghrelin and obestatin levels than controls. In hypertensive obese patients, ghrelin and obestatin levels were negatively associated with systolic and diastolic blood pressure, fasting insulin and HOMA-IR. In normotensive obese patients, ghrelin, obestatin and the ghrelin/obestatin ratio were negatively associated with fasting insulin and HOMA-IR. In both patient groups, fasting obestatin and ghrelin concentrations were significantly and positively correlated with each other.
Conclusion
Changes in the levels of ghrelin and obestatin may play a role in the pathophysiology of obesity and hypertension.
Introduction
The increasing prevalence of obesity is a serious health concern in China. 1 Obesity is known to be strongly associated with hypertension and other arteriosclerotic disease, 2 but the pathogenic mechanisms linking hypertension and obesity have not been fully determined.3–5 The possible roles of obestatin and ghrelin in obesity and metabolic syndrome have been studied.6,7 Changes in the concentrations of these hormones, and in the ghrelin/obestatin ratio, may be risk factors for obesity and hypertension. 8
Ghrelin is a peptide hormone secreted primarily from the stomach and duodenum; it is a stimulant of appetite and increases adiposity in rodents. 9 However, many studies have shown that obesity is associated with a decrease in circulating ghrelin. 6 Ghrelin has also been reported to have potent anti-inflammatory actions, including inhibition of proinflammatory cytokine production and mononuclear cell binding in vascular endothelial cells. 10 Ghrelin may therefore have a protective effect on endothelial function and has been shown to lower blood pressure levels. 11 Low plasma ghrelin has been reported to be associated with insulin resistance, hypertension and type 2 diabetes. 12
Obestatin is a 23-amino acid amidated peptide encoded by the ghrelin gene that is also released from the stomach. It has been shown to interact with the orphan receptor G-protein-coupled receptor 39, and to oppose the stimulatory effect of ghrelin on food intake and gastrointestinal function.13,14 Studies in humans have shown that blood obestatin levels are significantly lower in obese subjects and correlate negatively with body mass index, insulin, glucose and the homeostasis model assessment of insulin resistance (HOMA-IR), indicating an important role for obestatin in body weight regulation.15,16 In addition, obestatin has been shown to be positively correlated with ghrelin. 8 This suggests that levels of both obestatin and ghrelin may be altered in obesity and insulin resistance. Obestatin has been reported to decrease vascular cell adhesion molecule-1 expression in endothelial cells when stimulated with tumour necrosis factor-α, and to increase oxidized low-density lipoprotein binding to macrophages. 17 Therefore, it may also have a potential function in the regulation of blood pressure. The aims of the present preliminary study were to measure fasting plasma ghrelin and obestatin concentrations in normotensive and hypertensive obese patients and to investigate their relationship with insulin resistance.
Patients and methods
Patients
Patients with central obesity, defined as a body mass index ≥25 kg/m2 and a waist circumference >90 cm in men and >85 cm in women, attending the outpatient clinic of the Second Hospital, Jilin University, Changchun, Jilin Province, China, between October 2011 and October 2012 were prospectively enrolled in this study. Patients with diabetes, severe hepatic or renal disease, acute cerebrovascular or cardiovascular accidents, gastric ulcer or a history of abdominal surgery were excluded from the study.
Patients with a systolic blood pressure (BP) >140 mmHg or diastolic BP >90 mmHg on at least two visits to the clinic were categorized as hypertensive. Healthy volunteers with a body mass index (BMI) in the range 18.5–23.9 kg/m2 (matched with the patients for sex and age) were recruited from the health-check centre of the Second Hospital, Jilin University, as a control group. None of the controls had hypertension, hyperlipidaemia, diabetes, cardiovascular disease or any other abnormal systemic condition on medical history, physical examination, electrocardiogram, radiography and routine blood chemical analyses.
None of the participants was receiving any drug treatment at the time of the study. In hypertensive patients taking antihypertensive drugs, medication was discontinued for ≥2 weeks before enrolment. Smokers were asked to refrain from smoking and all participants were asked to refrain from drinking alcohol for ≥24 h before the study.
All participants gave written informed consent and the study protocol was approved by the Ethics Committee of the Second Hospital, Jinlin University, Changchun, Jilin Province, China.
Patient assessment
Height, weight, and waist and hip circumferences were measured in each study participant, and the BMI and waist to hip ratio were calculated. A fasting venous blood sample with a total volume of 20 ml was collected from each study participant: 10 ml of this was used for routine testing of glucose, insulin, low-density lipoprotein-cholesterol and high-density lipoprotein-cholesterol. Insulin resistance was estimated using HOMA-IR, which was calculated as follows: 18 fasting insulin (mIU/l) × fasting glucose (mmol/l)/22.5.
The remaining 10 ml of the blood sample was collected into potassium/ethylenediaminetetra-acetic acid-coated tubes containing 500 KIU aprotinin for the measurement of plasma levels of total ghrelin and obestatin. The samples were then centrifuged at 2000
Statistical analyses
Data were expressed as the mean ± SD. Differences between the groups were analysed using Student’s t-test for continuous data. Pearson’s correlation coefficient was used to analyse correlations between biochemical parameters in each group after checking that the variable’s distribution was normal using the χ2-test. A P-value <0.05 was considered to be statistically significant. All statistical analyses were performed using SPSS® software, version 15.0 (SPSS Inc., Chicago, IL, USA).
Results
A total of 78 obese patients (49 male; 29 female; mean ± SD age, 53.2 ± 10.5 years) and 38 controls (22 male and 16 female; mean ± SD age, 53.32 ± 9.72 years) were enrolled in the study. Of the obese patients, 38 were hypertensive (25 male and 13 female; mean ± SD age 54.97 ± 9.31 years) and 40 were normotensive (24 male and 16 female; mean ± SD age 55.9 ± 10.59 years).
General characteristics and biochemical parameters in hypertensive obese patients, normotensive obese patients and healthy controls in a study investigating ghrelin and obestatin levels.
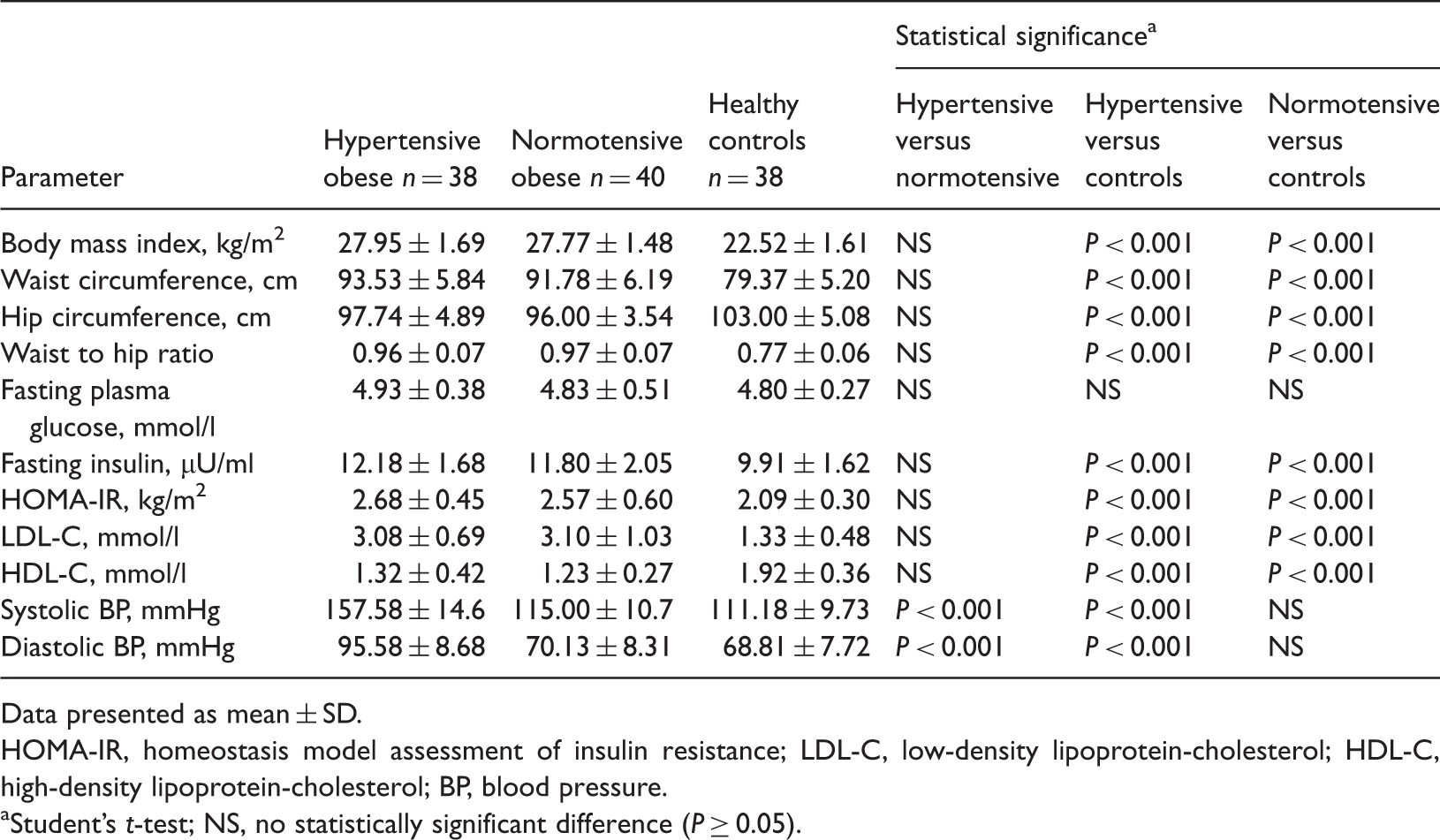
Data presented as mean ± SD.
HOMA-IR, homeostasis model assessment of insulin resistance; LDL-C, low-density lipoprotein-cholesterol; HDL-C, high-density lipoprotein-cholesterol; BP, blood pressure.
Student’s t-test; NS, no statistically significant difference (P ≥ 0.05).
Fasting plasma ghrelin and obestatin levels and the ghrelin/obestatin ratio in hypertensive obese patients, normotensive obese patients and healthy controls.

Data presented as mean ± SD.
Student’s t-test; NS, no statistically significant difference (P ≥ 0.05).
Correlations between various biochemical parameters and fasting plasma ghrelin and obestatin levels, and the ghrelin/obestatin ratio, in hypertensive obese patients, normotensive obese patients and healthy controls.

HOMA-IR, homeostasis model assessment of insulin resistance; BP, blood pressure.
Pearson’s correlation coefficient; NS, not statistically significant (P ≥ 0.05).
Discussion
The results of the present study showed that plasma levels of ghrelin and obestatin in patients with hypertension and obesity were significantly lower than in patients with simple obesity, and that ghrelin and obestatin concentrations in obese patients were lower than in controls. These findings suggest that decreased ghrelin and obestatin concentrations may be associated with both hypertension and obesity.
These findings are in agreement with those of previous studies showing reduced circulating ghrelin in obese patients. Ghrelin has been reported to be negatively associated with systolic and diastolic BP, fasting insulin and HOMA-IR. 13 In obese patients, ghrelin production has been found to be downregulated by obesity-associated insulin resistance. 19 Some studies have also reported that reduced plasma ghrelin occurs in association with other insulin-resistant states such as hypertension, type 2 diabetes and polycystic ovary syndrome.12,13,19,20 A negative correlation between fasting ghrelin and insulin has also been demonstrated. 21 The findings in the present study that ghrelin was negatively associated with systolic and diastolic BP, fasting insulin and HOMA-IR in hypertensive obese patients are consistent with these results. It has been reported that ghrelin reverses endothelial dysfunction in patients with metabolic syndrome by increasing nitric oxide bioactivity, 22 suggesting that decreased circulating levels of ghrelin might play a role in the pathobiology of atherosclerosis and hypertension. 23
The role of obestatin in BP regulation and insulin sensitivity is unclear, but systolic BP has been shown to be an independent predictor of the ghrelin/obestatin ratio. 24 In addition, fasting plasma concentrations of obestatin are reduced in insulin resistance and are positively associated with whole-body insulin sensitivity in nondiabetic humans. 15 In the present study, obese patients had lower fasting plasma obestatin concentrations than controls. Furthermore, there was a clear relationship between obestatin and both BP and HOMA-IR, suggesting that obestatin might play a role in BP regulation.
In the present study, the ghrelin/obestatin ratio was significantly and negatively correlated with fasting insulin and HOMA-IR in normotensive obese patients, indicating that this ratio may be a risk factor for obesity. Plasma obestatin and ghrelin levels were closely correlated with each other, with ghrelin levels being highly predictive of obestatin levels. This link is reasonable, given that both hormones derive from the same precursor.
The present study has some limitations. The patient groups were small and therefore some results may not reach statistical significance. Further detailed studies based on a larger population are needed for a more comprehensive evaluation.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
We thank Dr Li Shumei for help in the performance of this study.
