Abstract
Objectives
To investigate plasma ghrelin and obestatin levels, and ghrelin/obestatin prepropeptide gene polymorphisms, in sequentially enrolled small for gestational age (SGA) infants.
Methods
Neonates were sequentially enrolled into this study and were then subdivided into different groups, according to different study aims and availability of study materials. Consequently, plasma ghrelin and obestatin levels were measured in term SGA, term appropriate for gestational age (AGA), term large for gestational age (LGA), preterm SGA and preterm AGA neonates. Levels of both peptides were also measured in AGA infants of different gestational ages, and in term AGA neonates at different days following birth. Three ghrelin/obestatin prepropeptide gene single nucleotide polymorphisms (SNPs), Arg51Gln, Leu72Met, and Gln90Leu, were measured in neonates.
Results
The study involved a total cohort of 581neonates. Out of 150 neonates (30 term AGA, 30 term SGA, 30 term LGA, 30 preterm AGA, and 30 preterm SGA), plasma obestatin levels were significantly higher in term SGA versus term LGA neonates (0.21 ± 0.02 ng/ml versus 0.17 ± 0.01 ng/ml, respectively). Out of a wider cohort, there were no significant differences in genotypes and allele frequencies of Arg51Gln, Leu72Met, and Gln90Leu SNPs between term SGA and AGA neonates, or between preterm SGA and AGA neonates.
Conclusions
Ghrelin/obestatin prepropeptide polymorphisms were not found to be associated with SGA status in neonates; however, ghrelin and obestatin levels may be involved in growth and development. Further studies are required to understand the relationship between ghrelin, obestatin and prenatal development.
Keywords
Introduction
With continuing improvements in perinatal medicine, the numbers of preterm and low-birth weight infants continue to increase. Large percentages of preterm and low-birthweight infants are classified as small for gestational age (SGA), mainly due to intrauterine growth retardation as a result of fetal, maternal, environmental or other factors.1,2 Infants born SGA have a significantly increased risk of mortality and morbidity in the perinatal period, adversely affected childhood development and increased disease susceptibility in adulthood. Obesity, diabetes, cardiovascular disease and other chronic conditions have been shown to correlate with low birthweight.3,4
Ghrelin, an endogenous ligand of the growth hormone (GH) secretagogue receptor, is mainly produced in the stomach. The human ghrelin/obestatin prepropeptide gene is located on the short arm of chromosome 3 (3p25-26). 5 Ghrelin mRNA is translated into the 117-amino acid preproghrelin, which is cleaved to proghrelin; proghrelin is cleaved to produce a 28-amino acid acyl ghrelin and des-acyl ghrelin. 6 Acyl ghrelin is a potential positive regulator for appetite, lipogenesis and GH release. Ghrelin secretion begins in the fetus, where it correlates with growth and development; ghrelin secretion also correlates with adult nutrition and growth. In addition, ghrelin plays a role in the regulation of food intake and body weight. 7
Obestatin, a polypeptide composed of 23 amino acids, is also produced in the stomach mucosa as a result of post-translational cleavage of residues 76–98 of preproghrelin. Both ghrelin and obestatin require C-terminal amidation modification of the post-translational precursor for bioactivity. 8 Obestatin inhibits food intake, jejunum peristalsis and weight gain, and is thus considered an antagonist of ghrelin.9,10
The ghrelin–obestatin interaction is involved in the regulation of food intake and maintenance of energy balance. 11 These two peptides have been confirmed to be associated with postnatal growth and development, and some adulthood diseases (including heart disease, obesity and diabetes). 12 As the precursor of ghrelin and obestatin, preproghrelin is involved in regulating the expression of these two peptides (which have contrasting biological functions). Single nucleotide polymorphisms (SNPs) of the ghrelin/obestatin prepropeptide gene are known to correlate with growth retardation, obesity, diabetes and bulimia nervosa.13,14
The association of ghrelin and obestatin with birth weight is not well characterized: changes in ghrelin and obestatin levels during the neonatal period, and the roles of these peptides, remain unclear. In addition, the role of polymorphisms in the peptide precursors on peptide expression in SGA pathogenesis is poorly understood. Thus, the purpose of the present study was to address some of these unanswered questions and further our understanding of the molecular mechanisms underlying SGA pathogenesis.
Patient and methods
Study population
Neonates at the Children’s Hospital of Fudan University, Shanghai, China were sequentially enrolled into the present study between January and December 2010; these included SGA (birthweight <10th percentile), appropriate for gestational age (AGA; birthweight between 10th and 90th percentile) and large for gestational age (LGA) infants (birthweight >90th percentile). Neonates were classified by gestational age: term (37–41 weeks and 6 days’ completed gestation) and preterm (<37 weeks’ completed gestation). Neonates with congenital deformities, endocrine diseases, severe infections and asphyxia were excluded.
The study was approved by the ethics committee of the Children’s Hospital of Fudan University, Shanghai, China. Written informed consent was obtained from the parents of each child.
Study groups
Neonates were divided into the following five groups: term SGA; term AGA; term LGA; preterm SGA; preterm AGA. AGA neonates were further divided into five groups according to gestational age: ≤30 weeks; 31–33 weeks; 34–36 weeks; 37–39 weeks; 40–41 weeks and 6 days. SGA neonates were divided into two groups according to gestational age: 34–36 weeks (late preterm) and 37–39 weeks.
Term AGA neonates were divided into five groups according to age in days following birth: day 1; day 7; day 14; day 21; day 28. Preterm AGA and preterm SGA neonates were divided into two groups according to age in days following birth: day 1; day 7.
Blood sampling
On the corresponding day as detailed above, peripheral venous blood (0.5 ml) was collected from each infant into a tube containing 2 mg/ml ethylenediaminetetra-acetic acid (EDTA). Plasma was isolated from blood, centrifugated (37℃, 4000
For genetic analyses relating to ghrelin/obestatin, neonates were divided into four groups: term SGA, term AGA, preterm SGA, and preterm AGA. Peripheral venous blood (0.2 ml) was collected from each neonate into an EDTA tube and stored at −80℃ prior to the detection of three ghrelin/obestatin prepropeptide gene SNPs: Arg51Gln (rs34911341), Leu72Met (rs696217), and Gln90Leu (rs4684677).
Laboratory assays
For detection of total plasma ghrelin and obestatin protein levels, plasma samples were thawed at room temperature and analysed using enzyme-linked immunosorbent assay (ELISA) kits (Human Ghrelin EIA Kit, Catalogue No. EIA-GHR-1; Human obestatin EIA Kit, Catalogue No. EIA-OBS [RayBiotech, Norcross, GA, USA]) according to the manufacturer’s instructions. Intra- and interassay coefficients of variation were <10% and <15%, respectively.
Primer and probe sequences used in the present study for single nucleotide polymorphism (SNP) analysis of the ghrelin/obestatin prepropeptide gene in neonates.

rs ID, reference SNP identification number; FAM, 6-carboxyfluorescein (reporter dye); HEX, hexachloro-6-carboxyfluorescein (reporter dye); TAMRA, tetramethylrhodamine (quencher dye).
Each polymerase chain reaction (PCR) contained 20 pmol/µl of each primer and probe in a TaqMan® Universal Master Mix (Applied Biosystems, Foster City, CA, USA), and 1μl genomic DNA in a 20 -µl reaction volume. One negative control sample (to amplify glyceraldehyde-3-phosphate dehydrogenase) was included in each PCR run. Real-time PCR was performed in an ABI 9700 Real-Time® PCR System analyser (Applied Biosystems, Foster City, CA, USA), with the first cycle of 10 min at 95℃ followed by 40 cycles of 15 s at 92℃ and 60 s at 60℃. Following PCR, each sample was automatically analysed by measuring allele-specific final fluorescence in an ABI Prism® 9700HT detection system, using SDS software, version 2.2 for allele discrimination (Applied Biosystems).
Statistical analyses
All data were analysed using SPSS®, version 19.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Between-group comparisons were performed using Student’s unpaired
The three SNPs of the ghrelin/obestatin prepropeptite gene were determined on a case–control basis: the case group comprised SGA neonates; the control group comprised AGA neonates. Variables were assessed with Pearson
For all tests a
Results
A total of 581 neonates were sequentially enrolled in the present study; groups of neonates were then subdivided as described below. SNP levels could not be assessed in all neonates due to inadequacy of blood sampling or lack of access to equipment.
Plasma ghrelin and obestatin levels
Clinical characteristics of neonates divided into five groups according to gestational age (term or preterm) and birthweight (small for gestational age [SGA]; appropriate for gestational age [AGA] or large for gestational age [LGA]).

Data presented as
Delivery mode, caesarean section/vaginal.
GA, gestational age; HC, head circumference.
No statistically significant between-group differences (
Ghrelin and obestatin levels in neonates divided into five groups according to gestational age (term or preterm) and birthweight (small for gestational age [SGA]; appropriate for gestational age [AGA] or large for gestational age [LGA]).

Data presented as mean ± SD.
In addition, 100 AGA neonates were divided into five groups according to gestational age: ≤30 weeks, 31–33 weeks, 34–36 weeks, 37–39 weeks and 40–41 weeks and 6 days. Next, 40 SGA neonates were divided into two groups by gestational age: 34–36 weeks and 37–39 weeks, with 20 neonates in each of the seven resulting groups. No significant differences in plasma ghrelin and obestatin levels and ghrelin/obestatin ratios were observed between AGA neonates of different gestational ages (data not shown). Plasma obestatin levels in the ‘late’ preterm (34–36 weeks’) SGA group (0.17 ± 0.02 ng/ml) were significantly lower than those observed in the term SGA group (37–39 weeks; 0.21 ± 0.02 ng/ml;
Ghrelin and obestatin levels in appropriate for gestational age term neonates divided into five groups according to age in days following birth.
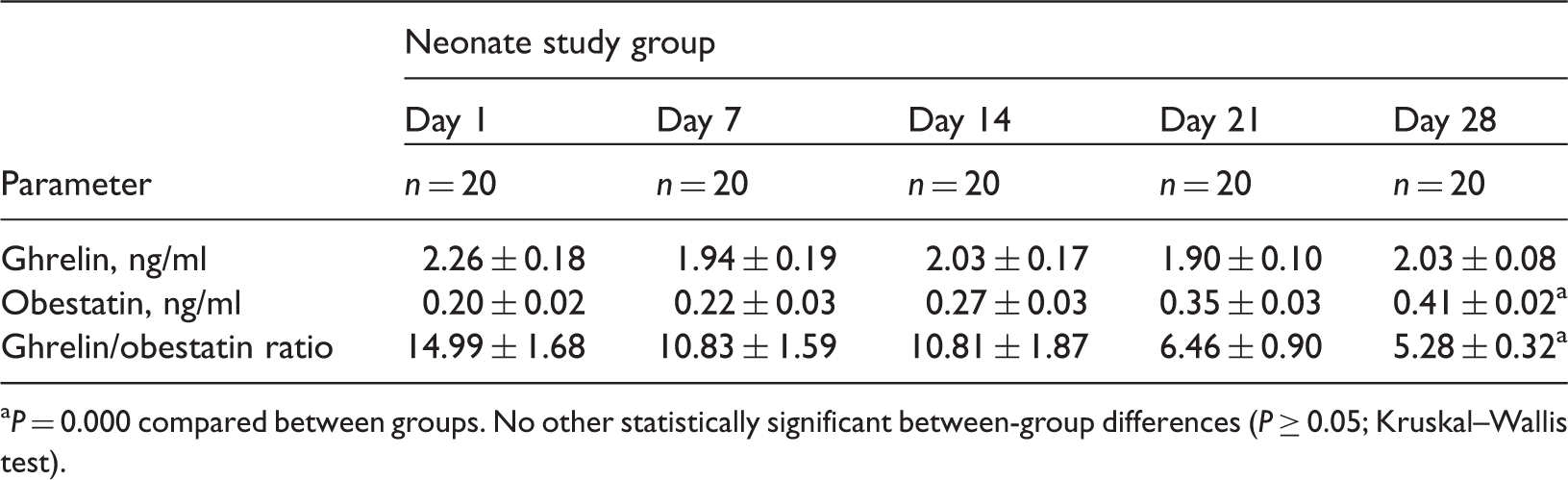
Ghrelin/obestatin prepropeptide gene polymorphisms
Clinical characteristics of 496 neonates divided into four groups according to gestational age (term or preterm) and birthweight (small for gestational age [SGA] or appropriate for gestational age [AGA]); neonates were analysed for single nucleotide polymorphisms within the ghrelin/obestatin prepropeptide gene.

Data presented as
GA, gestational age.
No statistically significant between-group differences (
Genotype and allele frequencies of ghrelin/obestatin prepropeptide gene single nucleotide polymorphisms (SNPs) in neonates divided into two groups: small for gestational age (SGA) and appropriate for gestational age (AGA).

Data presented as
Genotype and allele frequencies of ghrelin/obestatin prepropeptide gene single nucleotide polymorphisms (SNPs) in neonates divided into two groups: term small for gestational age (SGA) and term appropriate for gestational age (AGA).

Data presented as
Genotype and allele frequencies of ghrelin/obestatin prepropeptide gene single nucleotide polymorphisms (SNPs) in neonates divided into two groups: preterm small for gestational age (SGA) and preterm appropriate for gestational age (AGA).
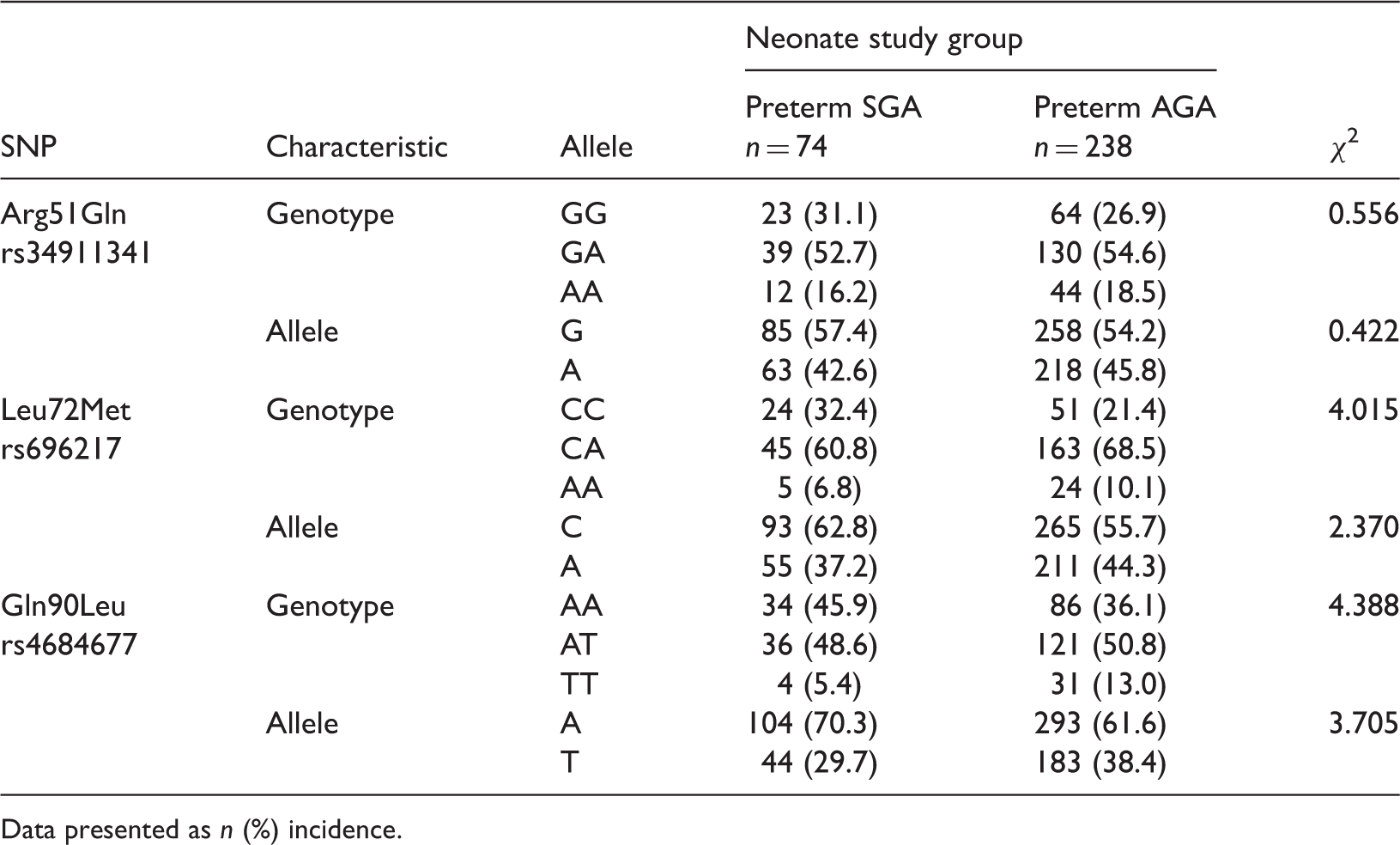
Data presented as
Discussion
Ghrelin was discovered in 1999 and is of research interest due to its wide-ranging functions. Ghrelin can regulate blood glucose levels by stimulating GH secretion, promoting insulin resistance and stimulating glycogenesis. 15 The dose–effect relationship between ghrelin and GH secretion suggests that ghrelin may play an important role in growth and development during the fetal and postnatal period. 15 Ghrelin is also involved in the regulation of energy homeostasis through its effect on the hypothalamus. 12 Ghrelin levels have been shown to correlate closely with insulin resistance, hypertension and type II diabetes. 16 Furthermore, ghrelin is a hormone that can promote appetite and lipogenesis. Expression of ghrelin mRNA and protein in rat gastric tissues and serum, respectively, has been shown to increase after fasting for 48 h. Postfeeding serum ghrelin levels returned to the original level, which indicated that fasting may promote ghrelin synthesis and secretion. 17 Ghrelin exerts a long-term effect on body weight through regulation of diet and energy balance. Studies have revealed that elevated ghrelin levels can increase body weight and, in turn, lead to obesity.11–13 Ghrelin and obestatin are brain–gut peptides produced by the stomach mucosa; both are derived from the common preproghrelin following post-translational splicing and modification. Ghrelin is formed by acetylation of preproghrelin, whereas obestatin is formed by C-terminal amidation of preproghrelin. 18 Ghrelin increases food intake and body weight, whereas obestatin acts as a ghrelin antagonist, suppressing ghrelin’s positive effects on appetite and jejunum contraction. Ghrelin and obestatin maintain homeostasis through a complex network of hormones and neural signals. 19
Ghrelin and obestatin are involved in the secretion of GH, energy homeostasis and metabolism, and regulation of growth and development.18,20 In the present study, changes in ghrelin and obestatin levels during the neonatal period were assessed using several grouping methods. There were no significant differences in plasma ghrelin levels between the different groups in the present study, which may have been due to the total ghrelin levels detected, without assessing the levels of octanoylated ghrelin (which is the active form of ghrelin): this may have provided better insights into the role of ghrelin during fetal development.
Obestatin expression in the neonatal period is not well characterized. The present study revealed a significant difference in obestatin levels between SGA and LGA infants, suggesting that obestatin is involved in fetal development. A significant difference in obestatin levels between late preterm SGA infants and term SGA infants was also found in the study. These data indicate that obestatin levels might increase gradually in the neonatal period, which raises the possibility that obestatin levels would continue to increase and play a role in the growth and development after the neonatal period. No antagonistic relationship between ghrelin and obestatin was observed in the present study, which may be due to low expression levels of ghrelin and obestatin during the neonatal periods analysed, or because these peptides are not involved in food intake and energy balance during the neonatal period. Further studies are required to address these questions.
The ghrelin/obestatin prepropeptide gene is located on chromosome 3 and encodes the ghrelin and obestatin peptides. Ghrelin/obestatin prepropeptide gene SNPs are associated with obesity, diabetes, bulimia nervosa, polycystic ovarian syndrome and breast cancer.21,22 A study concerning the –604 C > T promoter region SNP and the Leu72Met polymorphism of the ghrelin/obestatin prepropeptide gene showed that the Met72 mutation and -604 T allele play protective roles in insulin resistance. 23 The study concluded that these SNPs or some unknown functional variations may increase ghrelin levels and improve insulin sensitivity. The 3056 T > C SNP polymorphism in intron 2 has been found to correlate with basal ghrelin levels (high levels of acylated ghrelin), as well as with diet-related physiological and psychological variables and obesity. 13 Three different large-sample studies have indicated that Arg51Gln mutations are associated with low levels of plasma ghrelin, but not with obesity. 24 Additionally, Met72 was found to inhibit fat accumulation, and was correlated with metabolic diseases. 24 Another study of polymorphisms in the ghrelin/obestatin prepropeptide gene 25 found no significant differences in the genotype and allele frequencies of Arg51Gln, Leu72Met, and Gln90Leu between children with short stature and age- and sex-matched healthy controls, as well as between patients with growth hormone deficiency (GHD) and those with idiopathic short stature. Plasma ghrelin levels in the GHD group, however, were significantly lower than in the control group, suggesting a role of ghrelin in GH secretion and growth regulation. 25 Low ghrelin levels correlated with GH deficiency, however, ghrelin/obestatin gene polymorphisms did not correlate with GH deficiency. 25 In contrast, another study did not support the hypothesis that the ghrelin gene polymorphisms have a relevant impact on the secretion of total and acylated ghrelin. 26
In the present study, three ghrelin/obestatin prepropeptide gene SNPs – Arg51Gln, Leu72Met, and Gln90Leu – were detected in neonates. There were no significant differences in the allele and genotype frequencies between the SGA and AGA groups. To assess the correlation between these three SNPs and the different aetiologies of term SGA and preterm SGA infants, the frequencies of the SNPs were compared between term SGA and term AGA neonates, as well as between preterm SGA and preterm AGA neonates. There were no significant differences in the frequencies of homozygous and heterozygous genotypes, or alleles of the three SNPs between the SGA and AGA groups. However, we acknowledge that the small sample size may have been inadequate to effectively evaluate the relationship between ghrelin/obestatin prepropeptide gene polymorphisms and SGA.
In summary, ghrelin and obestatin are of considerable research interest because of their broad biological functions and possible roles in growth and development. The present study suggests that there may be a relationship between obestatin and fetal or neonatal development. Expression of ghrelin and obestatin during the neonatal period requires further study with larger sample sizes and longer follow-up periods, to confirm the relationship between SGA and ghrelin and obestatin levels or ghrelin/obestatin prepropeptide gene polymorphisms.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
