Abstract
Objectives
To determine if skin flap failure rates could be improved with the use of a dissolved oxygen wound dressing in a porcine model.
Methods
Full-thickness skin flaps (4 × 16 cm) were raised on pigs. Flaps were randomly assigned after surgery to experimental treatment with a dissolved oxygen dressing (treatment group) or a hydrogel dressing (control group). Flaps were evaluated daily for 14 days. Skin flaps that failed any one of four key clinical outcomes were considered failures. Histological parameters (including skin and subcutaneous necrosis, inflammation, ischemia, fibrosis, and bacterial load) were compared by a blinded histopathologist.
Results
Sixteen full-thickness skin flaps were raised on four pigs. All animals survived surgery and all incisions were evaluable. Clinical flap failure was observed in six (75%) control-treated wounds and in two (25%) dissolved oxygen-treated wounds. Histological evaluation demonstrated no significant differences in the proximal 75% of the flaps. There were significant differences in a number of histological parameters in the distal 25% in favor of the dissolved oxygen dressing.
Conclusions
Flaps treated with a dissolved oxygen dressing had fewer clinical failures and improved histological profiles compared with control-treated flaps, suggesting that increasing local oxygen supply may improve the local wound healing environment.
Introduction
Skin flap survival depends on the vascular supply at the base of the flap. It is well established that excessive length-to-width ratios result in distal flap failure from persistent ischemia. 1 Prevalent ischemia can impair incision healing and contribute to the development of surgical site infection. 2 Surgical incisions disrupt normal perfusion, which may lead to ischemia in the surrounding tissues. However, ischemic tissues often remain viable long enough to benefit from angiogenesis, which may re-establish perfusion to overcome the ischemic condition. The blood supply of pedicled flaps or even more complex procedures such as free flaps as well as skin grafts (partial and full-thickness skin) is partially (flaps) or completely (skin grafts) interrupted. Whereas the skin graft is completely hypoxic, the transfer of the flap may induce a more or less extended hypoxic area.3,4 In these situations, the viability of hypoxic tissue is short lived, and it will rapidly become necrotic without correcting the oxygen deficit. Hyperbaric oxygen has been used in some circumstances to prevent necrosis of surgically-induced hypoxia in tissues. 5 However, such an intervention would necessitate an early recognition of the problem, followed by quick intervention to prevent inevitable tissue loss. Preserving tissue until perfusion is re-established could be addressed by providing a reliable method to supply oxygen directly to the tissues at risk following the surgical procedure.
Oxygen is essential for cellular function, and the human body depends on capillary circulation to deliver oxygen to cells. When capillary circulation is compromised, ischemic conditions develop and cells begin to deteriorate. Chronic wounds may have compromised circulation that decreases or prevents oxygen delivery to healing cells. In surgical and trauma wounds, impairment of oxygen delivery may be due to either a disruption of the blood flow due to severed vessels or the disruption of oxygen distribution from capillaries to cells due to increased edema. 6 In either case, impaired wound healing may be the overt clinical indicator of this lowered oxygen tension.7,8 Providing dissolved oxygen directly to the cells through the skin may promote wound healing in tissue with compromised blood flow. The oxygen tension in injured tissues is important in the healing process. Tissues are capable of surviving for prolonged periods of time with restricted nutrition, but they must have oxygen to carry out the energy-dependent processes of cellular division and extracellular matrix protein synthesis that are necessary to the healing process.
This study evaluated tissue preservation in a porcine skin flap model emphasizing compromised circulatory support. The intent of this study was to create an ischemic flap model that would lead to necrotic tissue. With the application of a dissolved oxygen dressing, it was hypothesized that the wounds in the treatment group would show improvements in healing and tissue survival compared with the control group.
Materials and methods
Surgical procedures
Flap assignment
Flaps were randomly assigned after surgery to either the experimental treatment group using a dissolved oxygen dressing (OxyGenesys™; AcryMed Inc, Beaverton, OR, USA) or the control group using a hydrogel dressing (FlexiGel®; Smith & Nephew, Largo, FL, USA). OxyGenesys™ is a closed-foam hydrogel dressing made from polyacrylamide polymer that incorporates gaseous oxygen into the closed-cells during the manufacturing process. FlexiGel® is an absorbent hydrogel dressing made from polyacrylamide matrix similar in formulation to OxyGenesys™. Two skin flaps were elevated on the left side of the animal and two on the right side of the animal for a total of four random skin flaps per animal (Figure 1). One entire side (left or right) of each animal was assigned to one treatment group (experimental treatment group or control group) to prevent possible overlap of the two different dressings and thus prevent cross benefit of the dressings between sites.
Map of surgical site and treatment group assignments with skin flap identification numbers (site IDs) for all four animals. Flaps were oriented with the pedicles at the animal ends to ensure secure flap attachment. The odd-numbered animals were oppositely treated from the even-numbered animals to account for ‘sidedness’. Site IDs refer to the type of treatment dressing used for the animals.
Flap elevation
These preclinical studies were performed at American Preclinical Services, Minneapolis, MN, USA, which is an ISO 17025 accredited, Association for Assessment and Accreditation of Laboratory Animal Care accredited, United States Department of Agriculture registered (Class R Facility license and Class A breeding license), and Good Laboratory Practice compliant Contract Research Organization. Four Yorkshire cross adult pigs weighing 50–80 kg were used for the study. The pigs were housed individually and fed a fixed-formula diet and water ad libitum. The study was reviewed and approved by the Institutional Animal Care and Use Committee at American Preclinical Services.
Sixteen full thickness skin flaps were elevated by an experienced research surgeon in four pigs; two skin flaps on each side of each animal (Figure 2A). Each flap measured 16 cm × 4 cm to create a 4:1 aspect ratio (Figure 2B). Wound drains were placed and sutured onto the muscle beneath the raised flaps and tunneled underneath the skin at the most distal end of the flap where it was sutured and connected to the closed suction drainage bulb. Flaps were then stapled back into the surrounding tissue over a silicone membrane with small hash marks to allow fluid communication through the silicone layer to the wound drain beneath it (Figure 2C). Each flap was created at a consistent depth to include the dermal layer, the subcutaneous fat, and the panniculus carnosus, a layer of muscle particular to the porcine model. The width of the base and anatomical depth of the flap allowed a consistent cross-sectional area of intact perfusion to each flap. All flaps were closed using surgical wound staples and dressed according to the randomly assigned treatment group. Each wound was dressed using two dressings approximately 10.2 cm × 10.2 cm, each then covered by a large clear dressing (3M™ Tegaderm™ 15.2 cm × 15.2 cm; 3 M, St Paul, MN, USA) (Figure 2D). Both the OxyGenesys™ and the FlexiGel® dressings were premoistened with 1 ml of sterile normal saline solution (0.9% sodium chloride solution) prior to applying the dressing over the skin flaps. After covering the wounds with the dressings, the animals were loosely wrapped with gauze dressing and covered with an elastic adhesive dressing wrap to protect the incisions from contact with cages, urine, fecal waste, or other debris in the animal environment between dressing changes (Figure 2E). The closed wound drainage bulbs were secured along the spine at the most external portion of the dressing (Figure 2F).
(A) Marking the locations for the surgically-created skin flaps prior to surgery using a standard template. (B) After the flaps were lifted, wound drains were placed three-quarters of the way into the flap bed and tunneled under the most distal end of the flap and sutured into place. (C) A thin layer of silicone was placed over the wound drain to separate the raised flap from the vascular wound bed. The silicone was ‘pie-crusted’ throughout the silicone layer to allow fluid to communicate through the silicone layer to the wound drain. (D) The wounds were dressed and held in place with tape and 3M™ Tegaderm™ dressings. (E) The dressings and animals were wrapped with cotton gauze and then with a secure elastic and adhesive dressing to secure the dressings and keep them intact for 24 h while the animals were housed in individual pens. Openings were created in the dressings to allow for defecation and urination. (F) Wound drains were attached to the outer dressing along the spine of the animal to protect them from damage during daily animal activities.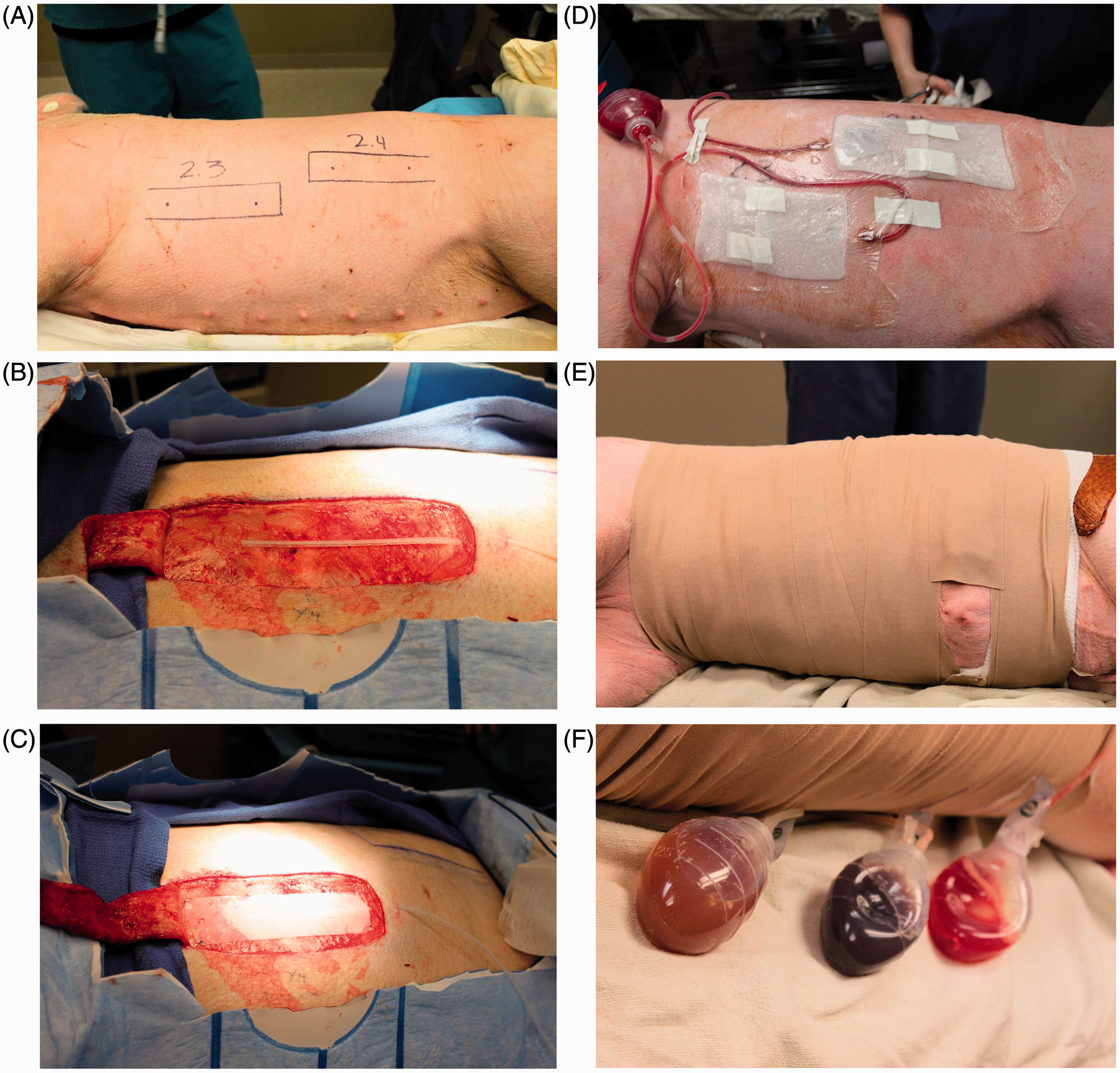
Closed wound drainage system
The wound drains were placed three-quarters of the way into the flap bed, were emptied daily, and were removed on postoperative day 4 (POD4) or when drained volumes fell below 20 ml/day (Figures 2D and 2F). Drained volumes were recorded daily. If fluid accumulation >20 ml was observed beneath the flap, wounds were needle-aspirated by penetrating the skin outside of the wound edges to avoid possible trauma to the primary skin flap. Wound drains were removed if occlusions could not be resolved.
Daily flap observation and dressing changes
Flaps were evaluated daily for 14 days (POD14). At each dressing change, the dressings were carefully removed and the wounds were cleaned, if necessary, using 10.2 cm × 10.2 cm gauze saturated with sterile normal saline solution and gentle scrubbing as needed. The length and width of the flap tissue, and the drain status (intact, patent, or removed) were also recorded along with digital photos being taken of each skin flap every day.
All skin flaps were examined according to an initial clinical assessment checklist with observed erythema (tissue redness that blanched upon pressure), edema (fluid retention resulting in tissue swelling), seroma (serous fluid collection within the flap), odor, skin sloughing, and wound drainage as collected by the closed wound drainage system. The color, consistency, and quantity of drainage were integral to assess flap progression. 9 Based upon the clinical observations, wounds were judged as healing (pass) or deteriorating (fail) according to incidence of the four key clinical outcomes: (i) active wound drainage; (ii) flap erythema; (iii) skin sloughing; and (iv) drainage on old dressings. One observation of any of these four clinical characteristics on POD14 indicated a deteriorating wound, and therefore a skin flap that failed to heal in 14 days. Drainage volumes were tracked as a real-time indication of wound healing, as active drainage indicates unresolved inflammatory processes and incomplete healing. 9 Additionally, sloughing of tissue that was viable underneath was observed, so analysis at the epidermal level was not informative.
The OxyGenesys™ and FlexiGel® dressings were applied and secured in place using 3M™ Tegaderm™ clear wound dressings as a secondary dressing. For this study, the OxyGenesys™ dressings were approximately 10.2 cm × 10.2 cm, which covered the 4 cm × 16 cm skin flaps with an overlapping border of approximately 2 cm to cover both the skin flap and the surrounding skin. To keep the dressing-to-dressing overlap consistent, the dressings were secured together with tape. At times when there was a copious amount of wound exudate, the dressings would adhere to the skin and the wound. When this occurred, the dressings were removed with sterile normal saline solution-moistened 10.2 cm × 10.2 cm cotton gauze and gentle scrubbing without incident to the skin flaps.
Biopsy sampling and histology
Under general anesthesia on POD14, 27 8-mm punch biopsies were performed on an isolated skin flap (Figures 3A and 3B). Two columns of three punch biopsies, perpendicular to the long axis of the site, were collected in each of four zones. Three punch biopsies were collected at the distal incision site. Samples were marked with tissue dye to differentiate the source location. The biopsy sites most medial were marked with black tissue dye, the sites in the middle were marked with yellow tissue dye, and the sites most lateral were marked with green tissue dye. The incision site was marked with red tissue dye. Each sample was placed in an individually-labeled container. The remaining sample (skin flap) was placed in fixative. The samples were immersion-fixed in 10% neutral buffered formalin for at least 24 h. The biopsies were bisected along the long axis and placed into cassettes. Each sample was faced-in and cut at 4 µm, and stained with hematoxylin and eosin (H&E). The samples were evaluated for histological parameters by a blinded histopathologist who looked at both dermal and subcutaneous characteristics of necrosis (pyknotic and karyorrhectic nuclei), inflammation (leukocytes and macrophages based on staining and morphology), ischemia, fibrosis (densely packed collagen bundles), and bacterial load (blue-stained coccoid bacterial colonies). The pathological changes of the tissue samples were assessed according to integrity of the skin structure such as dermal infiltration of inflammatory cells and the formation of collagen, capillaries and edema in the epidermis, dermis, and muscle. The biopsies were scored for each parameter using a numeric grading scale based on the percentage of the area of tissue affected, from 0% affected (unaffected and within normal limits) = 0 up to >50% affected = 4 (Table 1).
(A) Example of the template used for determining the location of each of the punch biopsies. Each flap was divided into four equal zones where zone 1 represented the proximal 25% of the flap, zone 2 represented the portion of the flap just distal to zone 1, zone 3 represented the portion of the flap just distal to zone 2, and zone 4 represented the most distal 25% of the skin flap. Black punches were located on the medial side of the flap, yellow punches on the middle, and green punches were located at the most lateral location of the flap. (B) Map of the punch biopsy zone assignment and labeling for tracking and repeatable sampling. Black, medial; yellow, middle; green, lateral. The histopathological scoring system used by the histopathologist to blindly score each histological parameter according to the percentage of the area of tissue affected.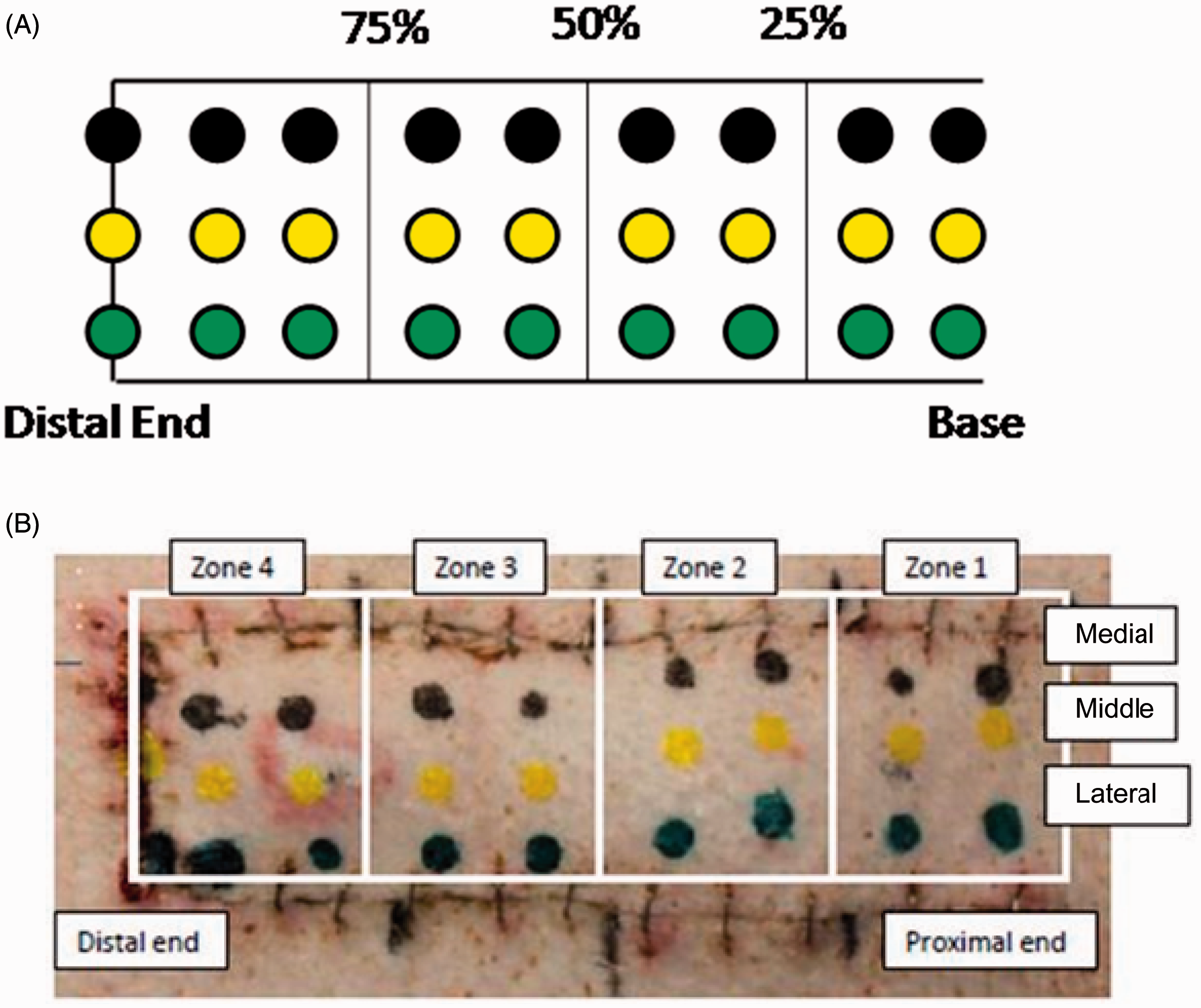

Statistical analyses
All statistical analyses were performed using the JMP® statistical package, version 9.0 (SAS Institute, Cary, NC, USA) for Windows®. Correlation between treatment groups and the clinical observations and histological scoring were carried out using Pearson’s χ2-test. At POD14, a table of frequency data was created that cross-classified the two categorical variables: dressing type and the presence or absence of a particular response of interest (e.g. odor). The χ2-test procedures applied to a contingency table were aimed at determining whether the two categorical variables were associated. χ2-test for 2 × 2 contingency table tests the probability that the number of positive and negative responses in the two dressing groups are from a similar population. A P-value ≤ 0.05 was considered statistically significant.
Results
Clinical results of each skin flap according to the pass or fail criteria in this preclinical study that aimed to determine if skin flap failure rates could be improved with the use of a dissolved oxygen wound dressing (OxyGenesys™) compared with a hydrogel wound dressing (FlexiGel®).

One observation of any of the four key clinical characteristics on postoperative day 14 indicated a deteriorating wound that failed to heal.
Hydrogel dressing, FlexiGel®; dissolved oxygen dressing, OxyGenesys™.
Statistical findings for the zone 4 histological outcomes in this preclinical study that aimed to determine if skin flap failure rates could be improved with the use of a dissolved oxygen wound dressing (OxyGenesys™) compared with a hydrogel wound dressing (FlexiGel®). a
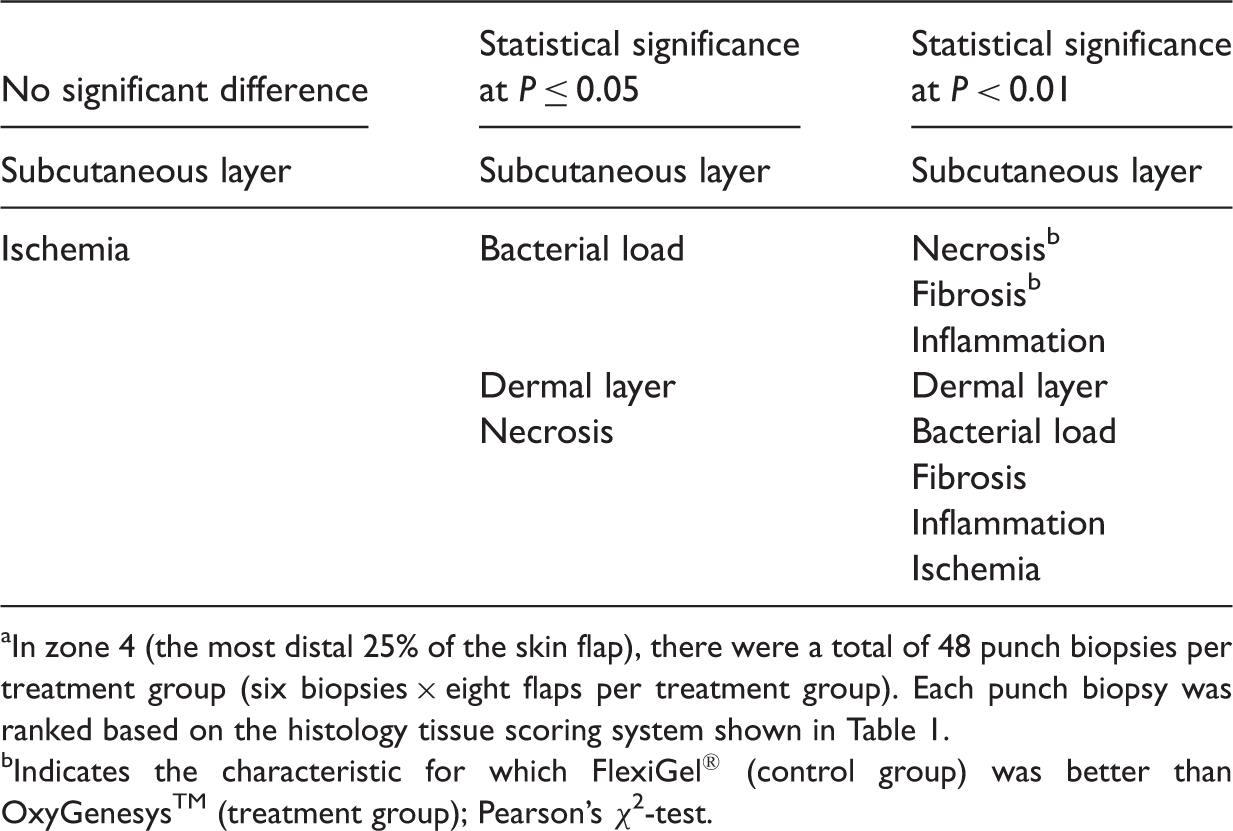
In zone 4 (the most distal 25% of the skin flap), there were a total of 48 punch biopsies per treatment group (six biopsies × eight flaps per treatment group). Each punch biopsy was ranked based on the histology tissue scoring system shown in Table 1.
Indicates the characteristic for which FlexiGel® (control group) was better than OxyGenesys™ (treatment group); Pearson’s χ2-test.
The histopathologist noted the following histopathological changes. When ischemia was present in any of the dermal flaps, there was a sharp interface between necrotic and viable skin. Necrotic skin appeared eosinophilic (red) on H&E and often contained numerous foci of blue-stained coccoid bacterial colonies. Necrotic skin was devoid of inflammatory cells and granulation tissue formation. Viable skin adjacent to ischemic skin had increased epidermal thickness (acanthosis) with thick papillary projections extending into the inflamed and fibrotic superficial dermis. The majority of skin flaps contained moderate to marked amounts of granulation tissue infiltrated by large numbers of neutrophils, lymphocytes, and lesser macrophages. Relatively normal sections of skin often contained minimal perivascular inflammation comprised of eosinophils with minimal granulation tissue formation.
Discussion
This study aimed to create an ischemic skin flap model that emphasized compromised circulation that would lead to tissue necrosis. Ischemic tissue isolated from the general circulation is at risk of necrosis and can compromise the overall wound healing process. The lack of oxygen may hinder many essential healing processes, including free-radical production for antibiotic potential10,11 and fibroblast collagen synthesis.12,13 Low oxygen tension in a wound bed is very often a precursor of necrosis. The skin pedicle flap is an established model for studying tissue necrosis and ischemic wound healing in animals. 14 While the pedicle vasculature supplies the flap with oxygen, which is critical to flap survival and overall wound healing, delivery of blood to the tissue at the distal end of the flap is often insufficient to promote proper wound healing and results in necrosis.15,16 To prevent ischemic conditions in the ends of random skin flaps that lack a major blood vessel supply, it is customary to limit the flap length-to-width ratio to 3:1. 1 In this present study, skin flaps with a 4:1 length-to-width ratio were created on porcine subjects and monitored over a 14-day course of healing. The results of the present study demonstrated that this porcine skin pedicle flap model was an effective and informative model in which to study ischemic tissue necrosis and wound healing.
It has long been a goal to better deliver oxygen to wounded tissue to improve cellular and tissue healing. Early studies to increase blood flow with vasodilating drugs largely prevented distal flap necrosis in rats. 17 For decades, though, hyperbaric oxygen treatment has been the most common way to increase the oxygen tension in hypoxic tissue in an attempt to improve wound healing. 18 For skin flaps specifically, hyperbaric oxygen therapy improved healing and decreased necrosis in a porcine pedicle flap model 19 and decreased inflammation in a bi-pedicle dermal rat model. 20 Because hyperbaric therapy is not always available or practical in all situations, recent research has sought to deliver oxygen topically, as in a wound dressing. A previous study demonstrated that dissolved oxygen in a gel matrix, OxyGenesys™, is an effective method of transcutaneous oxygen delivery, with a penetrative capacity of 700 µm in human skin. 21
The current study aimed to evaluate this OxyGenesys™ technology in a porcine model of ischemic wound healing. The hypothesis was that transcutaneous dissolved oxygen in a wound dressing covering poorly-perfused tissue would supplement the hypoxic environment, promote wound healing and limit necrosis. The results of the present study demonstrated that the distal ends of the skin pedicle flaps (zone 4) treated with OxyGenesys™ displayed better overall wound healing characteristics than control FlexiGel®-treated flaps after 14 days, as evidenced by fewer clinical failures and improved scoring on most histological markers.
Evaluating the histological characteristics can be complicated and is subject to the experience of the pathologist. Similar histopathological results were observed in zones 1–3 when comparing each parameter using these two groupings. In zone 4, dermal necrosis, bacterial load, and inflammation were significantly more evident in the control FlexiGel®-treated group than in the OxyGenesys™-treated group due to overall dermal breakdown in the FlexiGel®-treated flaps. This was similarly reflected in the subcutaneous levels in zone 4, where both bacterial load and inflammation were less evident in skin flaps treated with OxyGenesys™ compared with those treated with FlexiGel®. Unexpectedly, in the subcutaneous levels of the skin flaps, there was significantly less necrosis and fibrosis in the FlexiGel®-treated skin flaps compared with the OxyGenesys™-treated flaps.
In conclusion, this pilot study in a porcine skin flap model demonstrated significant differences between skin flaps treated with either OxyGenesys™ or FlexiGel® in terms of the extent of dermal necrosis, dermal bacterial load, dermal fibrosis, dermal inflammation, dermal ischemia, subcutaneous bacterial load, subcutaneous necrosis, subcutaneous fibrosis, and subcutaneous inflammation. All of the characteristics, with the exception of subcutaneous necrosis and subcutaneous fibrosis, favored the use of the topical dissolved oxygen dressing OxyGenesys™. These results suggest that the environment created by the OxyGenesys™ dressing improves ischemic wound repair and should be further investigated as a method for delivering oxygen to healing wounds with the aim of improving clinical outcomes.
Footnotes
Declaration of conflicting interest
Susan Zellner received a salary from Kimberly Clark Healthcare, Roswell, GA, USA; Rowena Manabat and David F Roe, receive salaries from AcryMed Inc, Beaverton, OR, USA (a wholly owned subsidiary of Kimberly Clark).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
