Abstract
Objectives
To detect microRNA (miR)-126 expression and its correlation with vascular endothelial growth factor (VEGF) levels in placentas from patients with pre-eclampsia compared with those from normotensive pregnancies.
Methods
miR-126 expression, and VEGF mRNA and protein levels, in placentas collected sequentially from patients with pre-eclampsia and normotensive pregnancies were measured using real-time reverse transcription–polymerase chain reaction and Western blot, respectively. The relationship between miR-126 and VEGF expression was analysed statistically. The regulatory effect of miR-126 on VEGF expression in human placental choriocarcinoma (BeWo) cells in vitro was also investigated.
Results
miR-126 expression was decreased, and VEGF mRNA and protein levels were significantly lower, in placentas from patients with pre-eclampsia (n = 115) compared with placentas from normotensive pregnancies (n = 115). A positive correlation was found between VEGF mRNA and miR-126 expression (r = 0.79). In addition, miR-126 overexpression significantly upregulated VEGF expression in BeWo cells, whereas miR-126 downregulation decreased VEGF expression.
Conclusions
miR-126 was downregulated in placentas from patients with pre-eclampsia and this correlated with decreased VEGF expression. These findings indicate that miRNA-126 may be involved in pre-eclampsia pathogenesis and could be a potential biomarker for this disease.
Introduction
Pre-eclampsia is a disease of pregnancy, characterized by hypertension and proteinuria, which develops after 20 weeks’ gestation. It has been estimated that 5–7% of pregnancies worldwide are complicated by this disorder, resulting in a very large disease burden. 1 There is much evidence to show that pre-eclampsia originates in the placenta, and thus the placenta is believed to be central to the pathogenesis of this disease. 2 The molecular basis for placental dysregulation is not completely understood. Many hypotheses have emerged that attempt to piece together a causal framework for the disease, resulting in pre-eclampsia being named the ‘disease of theories’. 3
Vascular growth during implantation and placentation is critical for successful gestation, and vascular insufficiencies during placentation are thought to contribute to a number of obstetrical complications. 4 Pre-eclampsia is suggested to be related to an imbalance of angiogenic factors that result in endothelial dysfunction, the implication being that pre-eclampsia is primarily a disease of the vascular endothelium.5,6 Angiogenic factors are essential for normal placental vascular development. 4 Alterations in the regulation and signalling of angiogenic pathways in early gestation contribute to the inadequate cytotrophoblast invasion seen in pre-eclampsia. 4 Vascular endothelial growth factor (VEGF) is a disulphide-linked homodimeric glycoprotein that is selectively mitogenic for endothelial cells and appears to play a major role in the mediation of vasculogenesis and angiogenesis, and in the control of microvascular permeability and vasodilatation.7–9 Studies have shown decreased placental and serum VEGF expression in women with pre-eclampsia, and have indicated its involvement in endothelium dysfunction in this condition.10–12 The molecular mechanisms leading to VEGF dysregulation, however, remain largely unknown.
MicroRNAs (miRNAs) are small noncoding RNAs that are highly conserved post-transcription regulators of gene expression. 13 Through inhibition of protein translation or promotion of mRNA degradation, miRNAs have roles in physiological and pathological processes including cell differentiation, proliferation and growth, apoptosis, angiogenesis, inflammation, redox signalling and other endothelial cell functions.13–15 Since these processes are disrupted in pre-eclampsia, miRNAs could potentially play substantial roles in pre-eclampsia pathogenesis. Some investigators have studied global placental miRNA profiling in pre-eclampsia, and aberrant placental expression of several miRNAs has been confirmed in association with the pathogenesis of this disease.16–20
One of the endothelium-relevant miRNAs is miR-126, which is found highly enriched in endothelial cells, and plays a pivotal role in maintaining endothelial homeostasis and vascular integrity. 21 miR-126 facilitates VEGF signalling by repressing two negative regulators of the VEGF pathway, namely sprouty-related, EVH1 domain-containing protein 1 (SPRED1) and phosphoinositol (PI)-3 kinase regulatory subunit β (PIK3R2). 22 One study demonstrated that miR-126 may be used as a marker for endothelial cell destruction. 23 To the authors’ knowledge, however, there have been no reports investigating miR-126 expression and its possible correlation with VEGF levels in pre-eclampsia.
The aim of the present study was to measure miR-126 and VEGF expression in placentas from patients with pre-eclampsia and in placentas from normotensive pregnancies. The relationship between miR-126 and VEGF expression was assessed.
Patients and methods
Study population
Placental samples were sequentially collected from patients with pre-eclampsia undergoing caesarean section, and from gestational age-matched normotensive pregnancies undergoing elective caesarean section (controls) at the Department of Obstetrics and Gynaecology, The Second Hospital of Shandong University, Jinan, China between January 2009 and December 2012. Pre-eclampsia was defined as gestational hypertension (systolic pressure ≥140 mm Hg and/or diastolic blood pressure ≥90 mm Hg on at least two occasions after gestational week 20), with proteinuria (≥0.3 g/24 h), according to American College of Obstetricians and Gynecologists guidelines. 24
Patients with chronic hypertension, renal disease, collagen vascular disease and other pregnancy complications (such as fetal anomalies or chromosomal abnormalities) were excluded from this study. The Ethics Committee of The Second Hospital of Shandong University approved the consent forms and procedures necessary to utilize the tissues, and written informed consent was obtained from all participants prior to surgery.
Placental tissue collection
Caesarean section was performed with a low transverse incision under combined spinal and epidural anaesthesia, which comprised 2% lidocaine (5 ml) and 0.75% ropivacaine (2 ml). The skin, subcutaneous tissue and rectus sheath were consecutively cut. The rectus abdominis was then isolated followed by opening of the peritoneum, exposure of the lower uterine segment, opening of the bladder peritoneal fold and pushing down the bladder. A transverse incision of the lower uterine segment was made, to form an opening of ∼1 cm in diameter, followed by blunt dissection of the uterus to a diameter of ∼10 cm, and delivery of the fetus. The umbilical cord was then cut before injection of the uterus with oxytocin (20 IU) to promote uterine contraction and reduce bleeding. Next, the placenta was delivered, the uterine cavity was wiped, the cervix was expanded using an oval clamp, followed by continuous suture of the uterine incision and mattress buried suture of the serosa. The bilateral annex was checked to ensure no bleeding and the abdomen was closed layer by layer.
For RNA and protein isolation, only chorionic tissue from the central part of the placental maternal phase was collected. After cleansing in 0.01 mol/l phosphate buffered saline (pH 7.4), tissue samples were frozen in liquid nitrogen and stored at −80℃ until further use. All placental samples were frozen within 30 min following delivery.
Cell culture and transfection
Human placental choriocarcinoma cells (BeWo cells; American Type Culture Collection, Manassas, VA, USA) were cultured in Ham’s F12 media (Gibco BRL, Grand Island, NJ, USA) supplemented with 10% fetal bovine serum (FBS) and 100 IU/ml penicillin. For routine culture, cells were seeded at 4 × 105 cells/ml into six-well plates and maintained at 37℃ in a humidified atmosphere with 5% CO2. Cells were passaged when they reached 70% confluence. Transfection of miR-126 mimics (sense, 5′-UCGUACCGUGAGUAAUAAUGCG-3′; antisense, 5′-GAUUAUUACUCACGGUACGAUU-3′), miR-126 inhibitor (anti-miR-126, 5′-CGCAUUAUUACUCACGGUACGA-3′), or negative control (miR-NC or anti-miR-NC) (GenePharma, Shanghai, China) were performed using Lipofectamine® 2000 (Invitrogen, California, USA) according to the manufacturer’s instructions. Briefly, 1 day prior to transfection, cells were seeded at 4 × 105 cells/ml into six-well plates to provide 70% confluence and allowed to adhere at 37℃/5% CO2 in a humidified atmosphere. miR-126 mimic, miR-126 inhibitor and negative control oligonucleotides were transfected at a final concentration of 20 nmol/l, then cells were incubated at 37℃/5% CO2 for 48 h prior to further analysis.
Quantitative real-time reverse transcription (RT)–polymerase chain reaction (PCR) for miR-126 mRNA analysis
Total RNA was isolated using TRIzol® reagent (Invitrogen) according to the manufacturer’s protocol. Briefly, 1 ml TRIzol® reagent was added to 50 mg placental tissue or 1 × 107 cultured cells for total RNA extraction. RNA concentration and purity were measured using a NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies, Houston, TX, USA). Only samples with an OD A260/A280 ratio of 1.9–2.1 were subsequently analysed.
Expression of miR-126 was assessed by quantitative real-time RT–PCR, and the small nuclear RNA U6 was used as an internal control. The miR-126 and RNA U6-specific cDNA sequences were synthesized from total RNA using gene-specific primers, according to the two-step TaqMan® MicroRNA assay protocol (Applied Biosystems, Foster City, CA, USA). Reverse transcription reactions contained 10 ng of total RNA, 50 nmol/l stem-loop RT primer (part No. 200131; Applied Biosystems), 1 × RT buffer, 0.25 mmol/l each of deoxynucleotide triphosphate (dNTP), 3.33 U/µl MultiScribe® reverse transcriptase, and 0.25 U/µl RNase inhibitor. The 7.5 µl reaction mixture was initially incubated at 16℃ for 30 min, 42℃ for 30 min, and 85℃ for 5 min, then held at 4℃.
Real-time quantitative PCR was performed using an ABI7500 real-time PCR detection system (Applied Biosystems). Each 20 -µl reaction volume contained 1.33 µl of RT product (containing cDNA), 1 × Universal PCR Master Mix and 1 µl gene-specific primers and probes (miR-126 kit catalogue No. 4373269, U6 kit catalogue No. 4427975; Applied Biosystems). PCR amplification was initiated at 95℃ for 10 min, followed by 40 cycles of 94℃ for 15 s, 60℃ for 60 s and 72℃ for 50 s. The threshold cycle (C
Quantitative real-time RT–PCR for VEGF mRNA analysis
Following total RNA extraction, VEGF mRNA was analysed by converting 2 ng of RNA per sample to cDNA using an RNA reverse transcription kit with random hexamer primers (TOYOBA, Osaka, Japan), according to the manufacturer’s instructions. Briefly, reagents were prepared to give a final concentration of 1 × RT buffer (pH 7.4), 100 µM random hexamer primers, 100 µM each dNTP, 5.5 mM MgCl2 and 1.25 U/ml reverse transcriptase in a final volume of 20 µl. The RT reaction was performed by incubating the samples for 10 min at 25℃, 30 min at 48℃ and 5 min at 95℃. Real-time quantitative PCR was performed using an Mx3000P® Real-Time PCR System (Agilent; Palo Alto, CA, USA) and SYBR® qPCR Mix (TOYOBA), using 5 µl of the RT product and 300 nmol/l of each of the following primer pairs per reaction: β-actin control, forward 5′-GCAGGGGGGAGC CAAAAGGGT-3′ and reverse 5′-TGGGTGGCAGTGATGGCATGG-3′; VEGF, forward 5′-TGCAGATTATGCGGATCAAACC-3′ and reverse 5′-TGCATTCACATTTGTTGTGCTGTAG-3′. PCR cycling conditions comprised 95℃ for 1 min, followed by 40 cycles at 95℃ for 15 s, 58℃ for 15 s and 72℃ for 30 s. The relative amount of VEGF to β-actin was calculated using the equation 2−Δ
C
VEGF Western blot analysis
Protein extracts were prepared from placental tissues and cells using ice-cold lysis buffer (50 mM Tris-HCl, pH 6.8, 32 mM 2-mercaptoethanol, 2 % w/v sodium dodecyl sulphate [SDS], 10 % glycerol) supplemented with ethylenediaminetetra-acetic acid-free complete protease inhibitors (Roche, Penzberg, Germany). Following lysis, samples were centrifuged at 20 000
Statistical analyses
Statistical analyses were carried out using SPSS® software, version 16.0 (SPSS Inc, IL, USA); data were presented as mean ± SD. Between-group differences were analysed using Student’s t-test. Pearson’s correlation coefficient was used to analyse the association between miR-126 expression and VEGF mRNA levels. All tests were two-tailed, and the significance level was set at P < 0.05.
Results
This study included placental samples from 115 patients with pre-eclampsia and 115 gestational age-matched participants with normotensive pregnancies (control group). Each study participant was undergoing caesarean section. All patients had normal platelet counts and normal hepatic and renal function (white blood cell count, 5.6 ± 1.3 × 109/l; red blood cell count, 4.2 ± 0.9 × 1012/l; platelet count, 238.2 ± 51.6 × 109/l; alanine transaminase, 21 ± 4 IU/l; total bilirubin, 14.3 ± 4.2 µmol/l; blood urea nitrogen, 2.8 ± 0.5 mmol/l; creatinine, 81.6 ± 10.8 µmol/l). There were no statistically significant between-group differences in other demographic parameters (age and weight). Blood-pressure levels returned to normal and symptoms of proteinuria disappeared in all patients within 6 weeks postpartum.
Using quantitative RT–PCR, relative miR-126 expression in placental tissue from patients with pre-eclampsia (mean ± SD, 1.63 ± 0.29) was found to be significantly lower than in the normotensive control group (mean ± SD, 5.11 ± 0.48; P < 0.001; Figure 1). VEGF mRNA levels were also significantly lower in placental tissue from patients with pre-eclampsia compared with the control group (mean ± SD, 1.97 ± 0.33 versus 3.94 ± 0.58, respectively; P < 0.001; Figure 1). A positive correlation was revealed between VEGF mRNA and miR-126 expression (r = 0.79; P = 0.006; Pearson’s correlation coefficient).
MicroRNA (miR)-126 and vascular endothelial growth factor (VEGF) mRNA levels in placental tissue samples from patients with pre-eclampsia (PE placentas) and gestational age-matched patients with normotensive pregnancies (normal placentas), who underwent caesarean section. Data presented as mean ± SD; aP < 0.001, miR-126 levels in PE placentas versus normal placentas; bP < 0.001, VEGF levels in PE placentas versus normal placentas; Student’s t-test.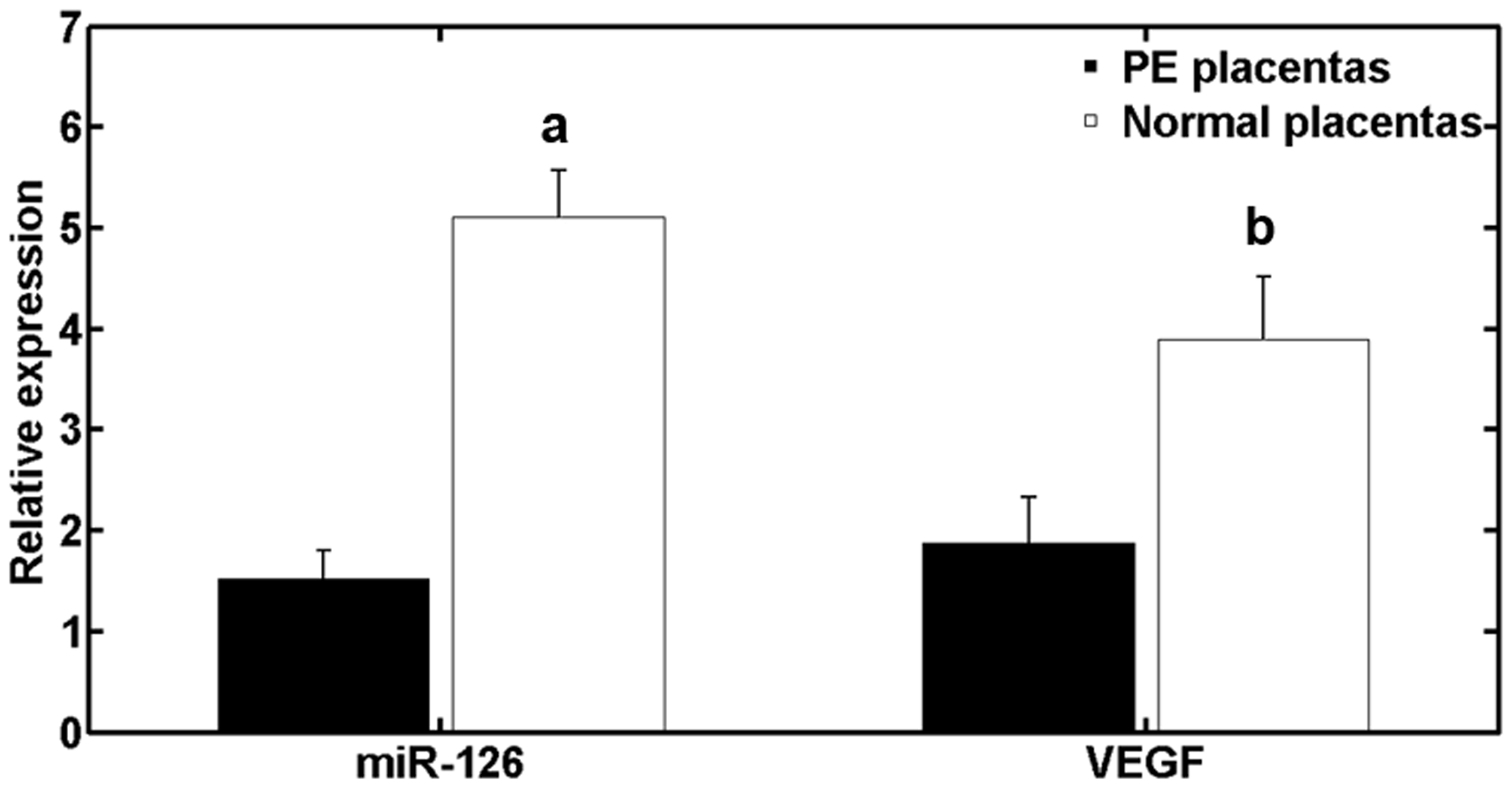
Relative VEGF protein levels were also decreased in placental tissue from patients with pre-eclampsia compared with the control group (P < 0.05; Figure 2).
Vascular endothelial growth factor (VEGF) protein expression in placental tissue samples from patients with pre-eclampsia (PE placentas) and gestational age-matched patients with normotensive pregnancies (normal placentas), who underwent caesarean section. (A) Representative Western blot of VEGF protein levels and β-actin (control). (B) Relative VEGF protein expression assessed by densitometry and grey value analysis using Quantity One software, version 4.6.2. Data presented as mean ± SD; *P < 0.05; normal placentas versus PE placentas; Student’s t-test.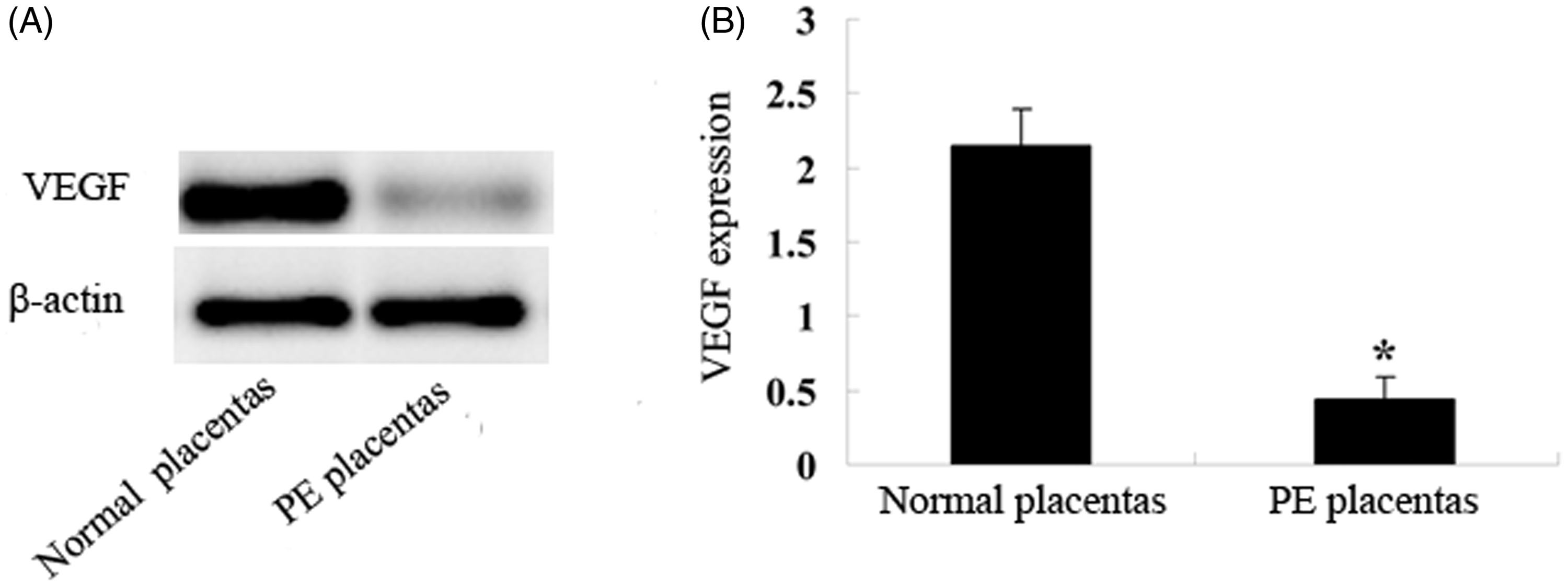
Quantitative RTPCR analysis confirmed increased miR-126 expression following miR-126 mimic transfection and decreased miR-126 expression following miR-126 inhibitor transfection, compared with control cells (miR-NC or Anti-miR-NC; P < 0.001 and P = 0.002, respectively; Figure 3A and B). Overexpression of miR-126 resulted in significantly upregulated VEGF levels compared with controls (P = 0.015; Figure 3C) and miR-126 downregulation resulted in decreased VEGF levels compared with controls (P < 0.001; Figure 3D).
Correlation between microRNA (miR)-126 and vascular endothelial growth factor (VEGF) expression in human placental choriocarcinoma (BeWo) cells in vitro. (A) Increased miR-126 expression in BeWo cells following transfection with miR-126 mimics compared with controls (miR-NC); *P < 0.001. (B) Decreased miR-126 expression in BeWo cells following transfection with miR-126 inhibitors (anti-miR-126) compared with controls (anti-miR-NC); *P = 0.002. (C) miR-126 overexpression significantly upregulated VEGF levels in BeWo cells with overexpressed miR-126 (miR-126 mimics) compared with negative controls (miR-NC); *P = 0.015. (D) Decreased VEGF levels in BeWo cells with downregulated miR-126 (anti-miR-126) compared with controls (anti-miR-NC); *P < 0.001. Data presented as mean ± SD; Student’s t-test.
Discussion
Pre-eclampsia, which is a vascular disorder characterized by hypertension and proteinuria during pregnancy, is a consequence of diverse pathophysiological processes involving endothelial dysfunction and intravascular inflammation.25,26 The placenta is a source (and site) of action of mediators of these processes, and plays a central role in pre-eclampsia pathogenesis.26,27 Conventional and microarray approaches have identified a substantial number of differentially expressed genes, including miRNAs, in human placentas from patients with pre-eclampsia compared with normotensive healthy term controls,19,20,28 and miRNAs clearly possess critical roles in regulating placental development and function. 29 miR-126 is highly expressed in endothelial cells and exerts an important function in maintaining endothelial homeostasis and vascular integrity. The absence of miR-126 during development increases vascular permeability and leakage.21,22 In the present study, placental tissue from patients with pre-eclampsia showed decreased miR-126 expression compared with tissue from the normotensive control group, suggesting that miR-126 may be involved in pre-eclampsia pathogenesis. Further research is needed, however, to clarify the cause of miR-126 downregulation in pre-eclampsia placental tissue.
The present study revealed a positive correlation between VEGF mRNA and miR-126 levels in placental tissue from patients with pre-eclampsia. In addition, a potential regulatory effect of miR-126 on VEGF expression was shown in the placental BeWo cell line in vitro. Incomplete trophoblast invasion and aberrantspiral arterial remodelling, leading to decreased uteroplacental perfusion, are two key links in pre-eclampsia pathogenesis. 30 VEGF is likely to be involved in remodelling of the uterine vessels, and in angiogenesis. 31 VEGF also participates in the regulation of trophoblast invasion, proliferation and differentiation. 32 In addition, decreased VEGF expression has been shown in immunohistochemical staining of placental tissues from patients with pre-eclampsia compared with normal controls who underwent caesarean delivery.10,33 In terms of the possible influence of miR-126 on the VEGF pathway, two papers revealed that miR-126 repressed SPRED1 and PIK3R2, which negatively regulate VEGF signalling via the mitogen-activated protein kinase and PI3 kinase pathways.21,22 In the present study, a correlation was observed between VEGF and miR-126 expression, however, the function of miR-126 in pre-eclampsia pathogenesis may not only be limited to the regulation of VEGF signalling. Other elements regulated by miR-126 may also contribute to the occurrence of pre-eclampsia, and further investigation is required to understand fully the role of miR-126 in this respect.
In conclusion, the present study indicates that miR-126 is downregulated in placental tissue samples from patients with pre-eclampsia compared with normotensive placental tissue samples, and that downregulated miR-126 expression correlates with decreased VEGF expression. These findings suggest that miRNA-126 may be involved in pre-eclampsia pathogenesis, and could be a potential biomarker for this disease.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
