Abstract
Objective:
This study investigated associations between polymorphisms in the vascular endothelial growth factor (VEGF) gene and susceptibility to acute mountain sickness.
Methods:
Two hundred Han Chinese soldiers who developed acute mountain sickness after rapidly ascending to an altitude of < 3600 m and 200 control soldiers (who did not develop the condition) were enrolled in the study. Twelve single nucleotide polymorphisms (SNPs) of the VEGF gene were genotyped in all the study participants. Plasma VEGF concentrations were measured by enzyme-linked immunosorbent assay in 40 subjects with acute mountain sickness and 40 controls before and after exposure to high altitude.
Results:
The frequencies of the rs3025039 genotype and allele were significantly different between the groups. Two SNPs, rs3025039 (which involves a C→T allele variation at position 936 in the 3′ untranslated region) and rs3025030 (which involves a G→C allele variation in the intronic sequence), were associated with a decreased risk of acute mountain sickness.
Conclusion:
The SNPs rs3025039 and rs3025030 of the VEGF gene may be associated with a decreased risk of acute mountain sickness development.
Introduction
Acute mountain sickness occurs at high altitude and presents as a collection of nonspecific symptoms that typically develop within 24 h, but may be evident even within the first few hours of hypoxic exposure. 1 The main symptoms include headache, anorexia, nausea, vomiting, fatigue, dizziness and sleep disturbance. The diagnosis is usually made on the basis of the Lake Louise scoring system, 2 which assigns scores to symptoms according to severity. A Lake Louise score of ≥ 3 points in the presence of headache is required for the clinical diagnosis of acute mountain sickness. 2 When individuals ascend rapidly to 2500 m, ∼10% will develop acute mountain sickness; when ascending to 4500 m, the incidence of this condition exceeds 60%. 3 Furthermore, potentially fatal cerebral oedema and pulmonary oedema can occur in individuals exposed to high altitudes.
In China, there are four high plateaus (Qinghai-Tibet, Inner Mongolia, Yun-Gui and the Yellow Land plateau), with a total population of nearly 60 million living at altitudes > 3000 m. In addition, a growing number of people travel to high altitudes for work or leisure. Consequently, determining the susceptibility of individuals to acute mountain sickness before high-altitude exposure is becoming increasingly important.
Although the exact mechanism for the development of acute mountain sickness is unknown, the basic pathophysiological changes include hypoxaemia (due to a decrease in the fractional concentration of inspired oxygen) and consequent vascular changes in the body, especially in the brain and lungs. It is known that some individuals are more susceptible to developing acute mountain sickness than others.4,5 Thus, individual differences in the tolerance to hypoxia and the ability to acclimatize may explain the altitude-dependent increase in acute mountain sickness incidence. Experience of previous high-altitude exposure probably represents the most reliable predictor of susceptibility. However, for the many individuals visiting high altitudes for the first time, advance knowledge of their likely susceptibility to acute mountain sickness would be beneficial.
Vascular endothelial growth factor (VEGF) is a major angiogenic factor that plays an important role as a regulator of endothelial cell proliferation and vascular permeability. VEGF has been shown to be markedly upregulated in hypoxic conditions. 6 Hypoxia stimulates the expression of VEGF, in addition to VEGF receptors in human endothelial and tumour cells. 7 VEGF exerts its effect by disrupting basement membrane ligands and increasing capillary permeability, leading to plasma leakage from capillaries. Overexpression of VEGF in the lungs induces increased pulmonary vascular permeability, resulting in pulmonary oedema. 8 Animal and in vitro studies have demonstrated a consistent upregulation of VEGF in endothelial tissues that are subjected to hypoxic conditions. 9 It is also of interest that corticosteroids (which are potent downregulators of VEGF expression in vitro 10 ) are highly effective in the prevention and treatment of acute mountain sickness. 11
The VEGF gene is located on chromosome 6p21.3 and is organized into eight exons and seven introns. At least 30 single nucleotide polymorphisms (SNPs) have been described in this gene, 12 some of which have been correlated with variations in VEGF protein production. VEGF gene expression is regulated by a variety of growth factors, cytokines and hormones as well as hypoxia. In addition, VEGF polymorphisms have been demonstrated to contribute to interindividual variations in VEGF expression. 13 There have been a number of studies investigating the biological properties and functions of VEGF in hypoxic conditions 14 and plasma VEGF levels in acute mountain sickness;15,16 however, there are few published data on the role of VEGF polymorphisms in relation to acute mountain sickness.
Functional SNPs in the VEGF gene may have an important role as genetic susceptibility markers for acute mountain sickness, given the important role of VEGF in hypoxia. During exposure to high altitudes, hypoxia-induced stimulation of VEGF expression may contribute to the development of AMS. The demonstration of an association between genetic polymorphisms of VEGF and the risk of developing acute mountain sickness would enable identification of those with a genetic predisposition for this condition. In this case– control study in China, VEGF gene polymorphisms and susceptibility to acute mountain sickness were studied in soldiers after exposure to high altitude.
Subjects and methods
Subjects
Study participants were selected from 2000 healthy male soldiers of Han Chinese ethnicity who lived at low altitudes (200 – 600 m above sea level). Those with a history of previous altitude exposure or acute mountain sickness, or who had taken prophylaxis for the condition, were excluded. After being assembled at Chengdu in Sichuan Province, China, which is located at sea level, the soldiers were flown to Lhasa, Tibet, which is located at an altitude of 3658 m. The study was conducted in December 2009.
All 2000 soldiers completed a Lake Louise acute mountain sickness self-assessment score sheet within 24 – 48 h after arrival at high altitude. The presence of acute mountain sickness was defined as a total score of ≥ 3 in the presence of headache, according to the Lake Louise scoring consensus. 2
A total of 200 subjects with acute mountain sickness (AMS group) and 200 subjects who had not developed the condition (control group) were randomly selected using computer-generated random numbers. Information on age, height, weight, medical history (including history of acute mountain sickness), previous altitude exposure, prophylactic medicines for acute mountain sickness, living environments and lifestyle factors (including smoking) were obtained for all the subjects.
The study protocol was approved by the General Hospital of Chinese People's Armed Police Forces Ethics Committee for Human Subject Research. Written informed consent was obtained from all participants.
SNP Selection for Analysis
The presence of SNPs in the VEGF gene was analysed using a subset of tag SNPs that are predictive of other SNPs in the gene. Tag SNPs were identified using genotype data for Han Chinese from the International HapMap Project (Phase 3) (available from http://hapmap.ncbi.nlm.nih.gov/index.html.en). Haploview software version 4.0 (available from http://www.broad.mit.edu/mpg/haploview) was used to examine the downloaded data across a 16.27 kb locus of VEGF with an average density of one SNP every 0.5 kb. Based on pairwise linkage disequilibrium analysis, tag SNPs with a minor allele frequency (MAF) > 0.05 were selected to capture unmeasured SNPs with a minimum r2 > 0.8. In addition, a few SNPs that have been reported to be associated with diseases or to show potential functional significance, with an expected MAF > 0.05 in Han Chinese, were also selected. A total of 12 SNPs from the VEGF gene were chosen for analysis: rs3025000; rs3025039; rs833070; rs1413711; rs45533131; rs3025053; rs3025040; rs10434; rs2010963; rs25648; rs699947; rs833061.
SNP Detection and Genotyping
Whole blood was drawn from all 2000 soldiers at low altitude, and 24 – 48 h after arrival at high altitude. Genomic DNA was extracted from 5 ml of whole blood in ethylene diamine tetra-acetic acid (EDTA) using the PAXgene Blood DNA kit (Qiagen, Hilden, Germany) according to the manufacturer's instructions. DNA was then stored at 4°C until used. A DNA fragment (100 – 300 base pairs) encompassing the SNP sites was amplified by polymerase chain reaction (PCR) using glyceraldehyde 3-phosphate dehydrogenase as the internal control gene. Briefly, 50 ng/μl of DNA was mixed with 5 U/μl HotstarTaq DNA polymerase (Qiagen), 2.5 mM of each deoxynucleoside triphosphate (Takara Biotechnology, Dalian, China), 1.25 μM of primers, PCR buffer (Sequenom®, San Diego, CA, USA) and 2.5 mM MgCl2. The cycling programme involved preliminary denaturation at 94°C for 30 s, followed by 45 cycles of denaturation at 94°C for 20 s, annealing at 56°C for 30 s and elongation at 72°C for 60 s, followed by a final elongation step at 72°C for 3 min. After PCR amplification and neutralization of the deoxyribonucleotide triphosphate, primer extension was performed by adding a one-base anchored primer. Extension products were differentiated using matrix-assisted laser desorption/ionization with time-of-flight mass spectrometry (Sequenom®). SNP data were directly uploaded from the sequence output files and analysed by allelic testing, to establish potential SNP allele– disease associations. The SNP assay was designed at Shanghai Benegene Biotechnologies, Shanghai, China.
Measurement of Plasma VEGF
Whole blood was drawn from all 2000 soldiers at low altitude and within 24 – 48 h after arrival at high altitude, collected into tubes containing EDTA and centrifuged at 2000
Plasma VEGF concentrations were measured in blood samples from 40 subjects with acute mountain sickness and 40 controls, randomly selected using computer-generated random numbers. Plasma was withdrawn and aliquoted to 0.5 ml. The specimens were then placed on dry ice and transported to Beijing for final analysis. VEGF plasma levels were determined by enzyme-linked immunosorbent assay using the Human VEGF QuantiGlo ELISA kit (R&D Systems, Minneapolis, MN, USA) according to the manufacturer's instructions.
Statistical Analyses
The genotype frequencies of VEGF were tested for Hardy-Weinberg equilibrium in both the acute mountain sickness and control groups using the χ2-test. Allele frequencies and genotype distribution were calculated in the acute mountain sickness and control groups and were analysed for association with the development of acute mountain sickness using the χ2-test. Adjusted odds ratios (ORs) with 95% confidence intervals (CI) were computed to test the association between AMS risk and genotype by logistic regression analysis. Student's t-test was used for analysis of plasma VEGF concentrations. For all statistical tests, the criterion for statistical significance was P ≤ 0.05. Analyses were conducted using SPSS® software, version 13.0 (SPSS Inc., Chicago, IL, USA).
Results
Characteristics of the study groups are shown in Table 1. Mean age, height, weight and smoking frequency were similar between the groups: 718/2000 soldiers (35.9%) developed acute mountain sickness at high altitude.
Demographic characteristics and acute mountain sickness scores 2 in subjects with scores ≥ 3 (AMS group) and controls (scores < 3)
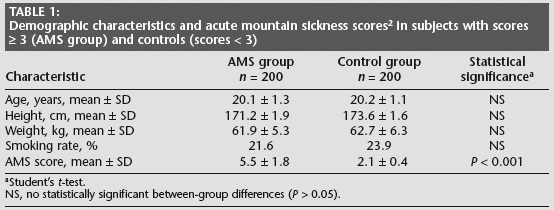
Student's t-test.
NS, no statistically significant between-group differences (P > 0.05).
SNP Analyses
All 12 SNPs from the VEGF gene were in Hardy-Weinberg equilibrium at a significance level of 0.01, and the average genotyping success rate was 99.18%.
Two SNPs of the VEGF gene, rs3025039 and rs3025030, showed evidence of an association with acute mountain sickness. The SNP rs3025039 involves a C→T allele variation at position 936 in the 3′ untranslated region (UTR) of the VEGF gene, whereas the SNP rs3025030 involves a G→C allele variation in the intronic sequence. Distribution of rs3025039 and rs3025030 alleles was different between the study groups (P = 0.012 and P = 0.019, respectively) (Table 2).
Genotype distributions and allele frequencies of single nucleotide polymorphisms rs3025039 and rs3025030 of the vascular endothelial growth factor gene in subjects with acute mountain sickness scores-2 ≥ 3 (AMS group) and controls (scores < 3)

Data presented as n (%) of subjects; data on rs3025039 unavailable for one subject in the AMS group.
Student's t-test.
NS, no statistically significant between-group differences (P > 0.05).
Genotype distributions of the SNP rs3025039 in the acute mountain sickness group were significantly different from those in control group (P = 0.029) (Table 2). Overall, the SNP rs3025039 was associated with a decreased risk of acute mountain sickness (P = 0.008; OR 1.779; 95% CI 1.160, 2.728; CC versus CT/TT) (Table 2).
Genotype distributions of the SNP rs3025030 were not significantly different between the study groups (Table 2). Variant homozygote carriers of rs3025030 (CC) had a reduced risk of acute mountain sickness (P = 0.019; OR 0.641; 95% CI 0.441, 0.931; C versus G). Overall, the SNP rs3025030 was associated with a decreased risk of acute mountain sickness (P = 0.018; OR 1.664; 95% CI 1.088, 2.545; GG versus CC/GC).
There were no statistically significant differences in the genotype distributions or allele frequencies between the study groups for the other SNPs analysed (data not shown). Genotypes of these SNPs were not significantly associated with the risk of acute mountain sickness.
Plasma VEGF
Plasma VEGF concentrations were significantly higher in the acute mountain sickness group than in the control group, both at sea level and at 3658 m (P < 0.01) (Table 3). In addition, plasma VEGF levels at 3658 m were significantly lower than at sea level in both groups (P < 0.01 for both) (Table 3).
Plasma concentrations of vascular endothelial growth factor in subjects with acute mountain sickness scores2 ≥ 3 (AMS group) and controls (scores < 3)

P < 0.01 compared with control group (Student's t-test).
P < 0.01 compared with sea level (Student's t-test).
The only mutation significantly associated with plasma VEGF levels was rs3025039 (936 C/T polymorphism). All study participants carrying this mutation had significantly lower VEGF levels than those carrying the VEGF 936 CC genotype, both at sea level and at high altitude (39.04 ± 21.55 pg/ml versus 65.11 ± 23.45 pg/ml, respectively, at sea level; 21.32 ± 12.15 pg/ml versus 51.34 ± 14.05 pg/ml, respectively, at high altitude; P < 0.01 for both). None of the other SNPs analysed were associated with significant differences in plasma levels of VEGF (data not shown).
Discussion
At altitudes between 2500 and 3000 m, the prevalence of acute mountain sickness is 10 – 30%, depending on the population, and at altitudes of 4000 – 4500 m the prevalence rises to 40 – 60%, 17 which is consistent with the results of the present study. Some individuals appear to be more susceptible to acute mountain sickness than others. 1 This fact has led researchers to exert substantial efforts towards explaining the differences in susceptibility and creating methods that predict the risks of acute mountain sickness development. The little that is known about the genetics of acute mountain sickness comes mostly from studies of relevant traits in high-altitude natives. 18 In the present study, a population-based case-control study of acute mountain sickness was used to examine genetic variation at the VEGF gene locus, and its relationship with plasma VEGF levels and acute mountain sickness risk. The contribution of 12 VEGF SNPs to the risk of acute mountain sickness was assessed, and evidence of an association between reduced risk and two SNPs (rs3025039 in the 3′ UTR and the intronic rs3025030) was found. To the best of our knowledge, this is the first molecular epidemiological study to demonstrate such an association. In addition, a relationship was also observed between plasma concentration of VEGF and presence of acute mountain sickness.
The SNP rs3025039 was first identified by Renner et al. 19 and was denoted as 936 C/T The presence of the T allele was shown to be associated with significantly lower plasma VEGF levels in healthy young men, 19 but the mechanism by which the polymorphism affects VEGF levels is not known. The variants rs3025039 and rs3025030 may cause changes to the potential binding sites of transcription factors, which may result in dysfunction of VEGF expression. This putative molecular mechanism is further supported by the demonstration that the intronic sequence is essential to VEGF expression regulation. 20 The significant association of these two SNPs with acute mountain sickness needs to be validated in further studies.
The mechanism by which the rs3025039 SNP (936 C/T polymorphism) affects the VEGF plasma concentration is currently not known. The 936 C allele carries a potential binding site for activator protein-4 (AP-4) that is abolished in the 936-T allele. 21 AP-4 is a helix–loop–helix transcription factor, enhancing expression of several viral and cellular genes by binding to specific enhancer sites; the loss of this potential binding site could be responsible for decreased VEGF expression. 22 Because of the possible transcriptional regulation linked to the 3′ UTR of the VEGF gene, especially at position 936, the C to T change at position 936 results in the loss of the core binding sequence for this transcription factor; the 3′ UTR and 5′ UTR binding proteins may interact, with the formation of a circular structure of mRNA that can alter mRNA translation. 23 VEGF proteins have the potential to bind the site encompassing position 936 and could interact with the 5′ UTR binding proteins.
The decreased risk of acute mountain sickness that is associated with the SNPs rs3025039 and rs3025030 described in the present study may be at least partially explained by this potential mechanism. Among the 12 SNPs of the VEGF gene studied, only rs3025039 and rs3025030 were significantly associated with acute mountain sickness. The determination of a genetic predisposition for the development of this condition is challenging because of the number of confounding factors. Further studies on the functional relevance of VEGF polymorphisms in acute mountain sickness will be needed to confirm these observations.
The protein VEGF is significantly upregulated by hypoxia in rats24,25 and human lungs, 26 and has been shown to be a powerful angiogenic factor 27 and endothelial cell-specific mitogen. 28 Based on the reported biological properties and function of VEGF in hypoxic conditions,14,26,29 the function of VEGF has been investigated in the pathogenesis of diseases associated with high altitude, including high-altitude pulmonary oedema and acute mountain sickness. Kaner and Crystal 30 reported that levels of VEGF compartmentalized in the alveolar epithelial lining fluid of normal humans were 500-fold greater than plasma levels. They suggested that when alveolar epithelial tight junctions are compromised at high altitudes, the normally high level of VEGF in the alveolar epithelial fluid has access to the pulmonary endothelium and can alter permeability. However, studies on changes in the levels of VEGF in humans exposed to high altitudes remain inconclusive: some authors have reported the enhancing effects of hypoxia on VEGF blood concentrations; 16 others have found a decrease or no change in VEGF levels.31,32 Dorward et al. 15 reported novel data on changes in plasma VEGF levels during acclimatization to high altitudes, but did not find an association between acute mountain sickness and plasma VEGF levels.
In the present study there were large interindividual differences in baseline VEGF levels at sea level; this is consistent with the results reported by Maloney et al., 31 who showed a trend towards decreased plasma VEGF levels at high altitudes but did not demonstrate a significant difference in VEGF levels between subjects with or without acute mountain sickness. In the present study, both groups had significantly lower plasma VEGF levels at an altitude of 3658 m than at sea level, suggesting that the VEGF concentration may be regulated differently from its cellular expression. Decreased plasma VEGF in individuals without acute mountain sickness may in part be due to enhanced renal VEGF clearance: a theory supported by a previous description of renal VEGF clearance and fluid retention in subjects susceptible to the condition. 31 In addition, VEGF receptors increase in many organs (including the brain and lung) during hypoxia; 33 these receptors can bind VEGF and consequently unbound VEGF concentrations decrease. Our present study results suggest that circulating unbound VEGF is not a vital component in the pathogenesis of acute mountain sickness. The reasons could include that VEGF plasma samples were collected during the early stages of the condition; further studies will focus on measuring VEGF plasma samples at different stages. In the present study, VEGF genotype and plasma levels were available for only a small number of subjects, therefore these results need to be confirmed by further studies. However, the statistically significant association between the 936 C→T mutation and the VEGF plasma level was apparent using data from only 80 subjects, which underlines the importance of this genetic marker. The sample size in the present study was relatively small for a genetic case–control association study. Another limitation was that genomic DNA was extracted from the whole blood of all subjects; plasma VEGF concentrations were measured in a limited number of subjects only.
In summary, the present study provides tentative evidence that certain SNPs of the VEGF gene are significantly associated with a reduced risk of acute mountain sickness, and that their effects may be due to altered VEGF expression. These findings may help to clarify the pathophysiology of acute mountain sickness.
Footnotes
Acknowledgements
This work was supported by a grant (No. 20090451581) from the China Postdoctoral Science Foundation Funded Project. The authors would like to thank all the individuals who participated in this study for their support.
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article.
