Abstract
Objective
A resting-state functional magnetic resonance imaging study (fMRI) to investigate pretreatment regional differences in brain function, in patients with early treatment responsive (ERD) and early treatment nonresponsive (END) major depressive disorder (MDD).
Methods
Patients with MDD and healthy control subjects underwent fMRI. Intrinsic neural activity at baseline was evaluated via amplitude of low-frequency fluctuations (ALFF). Antidepressant treatment was initiated after MRI. All patients received selective serotonin reuptake inhibitor type antidepressants at the minimum effective dose.
Results
There were significant differences in brain activity between patients (n = 56) and control subjects (n = 33). Brain activity in patients with ERD (n = 26) differed from those with END (n = 30) in the lingual gyrus and cerebellum. There was a significantly correlation between activity in these regions and disease duration in patients with ERD, and with amelioration of depressive symptoms in patients with END.
Conclusions
Brain regions related to the neural mechanism of MDD early treatment outcome were identified. These regions may have important implications for the treatment of MDD.
Keywords
Introduction
Major depressive disorder (MDD) is a serious mental illness with substantial functional disability and economic burden. 1 It is characterized by persistent and overwhelming feelings of guilt, sadness, anhedonia, worthlessness and hopelessness. Around 30% of patients with MDD do not respond to standard antidepressant treatment, 2 despite several decades of drug research and development. Standardized systems can identify improvements in MDD symptoms as early as 2 weeks after initiation of antidepressant treatment,3–5 a timescale that is not specific to any particular medication. 5 Importantly, early improvement in MDD symptoms can predict subsequent treatment outcome, 3 as early nonresponse is a highly specific predictor for final nonresponse (81–98%) and nonremission (87–100%).3,4,6
Neuroimaging studies of patients with MDD have evaluated the relationship between treatment response and abnormal brain function.7,8 Resting-state functional magnetic resonance imaging (RS-fMRI) has been used for investigating brain function under normal and pathological conditions, 9 with the amplitude of low-frequency fluctuations (ALFF) thought to represent regional neural function. 10 ALFF has been used to explore abnormal brain activity in neuropsychiatric disorders such as unipolar depression, 11 bipolar depression 12 and schizophrenia. 13 In addition, ALFF has identified differences in the cerebellum, visual recognition circuit and default circuit between patients with treatment-sensitive depression (TSD) and those with treatment-resistant depression (TRD). 14 More specifically, lower coherence-based regional homogeneity values in the cerebellum differentiated TRD from TSD with high sensitivity and specificity (>80%, respectively). 15 To the best of our knowledge, there have been no studies investigating brain function in the early phase of treatment (2 weeks).
Since both early nonresponders and responders have similar clinical pictures but disparate clinical outcomes, we speculated that there could be differences in pretreatment brain regional function. We hypothesized that differences between these two groups of patients in regions of abnormal function could help to predict early improvement and provide potential targets for new antidepressant drugs. The present study, therefore, used ALFF to assess brain function changes in first-episode, drug-naïve patients with MDD, and investigated their correlation with antidepressant efficacy.
Patients and methods
Study population
The study recruited consecutive, drug-naïve patients with first-episode MDD attending the Mental Health Centre, West China Hospital of Sichuan University, Chengdu, China, between December 2011 and December 2013. MDD was diagnosed by experienced psychiatrists (W.H.K. and Y.C.Y.), using the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID). 16 Exclusion criteria were: (i) other Axis I psychiatric disorders and symptoms; (ii) a history of organic brain disorder, neurological disorder or cardiovascular disease; (iii) pregnancy or any physical illness as assessed by personal history and laboratory analysis; (iv) age <18 or >60 years; (v) the inability to undergo an MRI. Patients were required to be right-handed (based on the Annett handedness scale 17 ) and to have a Hamilton Depression Rating Scale (HAMD-17) score ≥18 on the day of MRI scanning. 18
Right-handed healthy control subjects were recruited from the local area by poster advertisements. Subjects were screened using the nonpatient edition of SCID 16 to confirm a lifetime absence of psychiatric or neurological illness and were interviewed to exclude any family history of psychiatric illness.
The Ethics Committee of West China Hospital of Sichuan University approved the study protocol; all participants provided written informed consent according to the guidelines set by the MRI Centre and Mental Health Centre of Sichuan University.
Resting-fMRI data acquisition
A 3 Tesla MR scanner with a 12-channel phased array coil (Trio; Siemens, Erlangen, Germany) was used to acquire MRI data. A total of 175 whole brain volumes were collected on 30 oblique slices (5-mm thick), using a T2*-weighted echo planar imaging sequence sensitive to blood oxygen level-dependent contrast with the following acquisition parameters: TR = 2000 ms, TE = 30 ms, flip angle = 90, field of view (FOV) = 24 × 24 cm2, matrix size = 64 × 64, in-plane resolution = 3.75 × 3.75 mm2.
ALFF
Data were preprocessed using Statistical Parametric Mapping (SPM8, http://www.fil.ion.ucl.ac.uk/spm), Data Processing Assistant for Resting-State fMRI (DPARSF, http://www.rest.restfmri.net) 19 and Resting-State fMRI Data Analysis Toolkit (REST, http://www.rest.restfmri.net). 20 The first five volumes of each functional time series were discarded for signal equilibrium and participants’ adaptation to scanning noise. The 30 volume images were automatically realigned to the first image of the time series, to correct for head movements between scans. All subjects with head movement exceeding 1.5° (regardless of rotation) and translation exceeding 1.5 mm were excluded. Realigned images were then normalized and resampled to 3 × 3 × 3 mm3 voxels in Montreal Neurological Institute (MNI)-labelled space. Spatial and temporal smoothing were performed with a Gaussian filter of FWHM = 8 × 8 × 8 mm3.
The ALFF was calculated using DPARSF. After band-pass filtering (0.01–0.08 Hz) and linear trend removing, the time series were transformed to frequency domains using fast Fourier transformations (FFTs), the square root of power spectrum was calculated, and the mean was calculated across 0.01–0.08 Hz for each voxel. 9 This mean square root was referred to as ALFF. The ALFF of each voxel was standardized by dividing by global (within the brain) mean ALFF value for further statistical analysis.
Treatment
Antidepressant treatment was initiated for all patients after MR imaging. All patients received selective serotonin reuptake inhibitor (SSRI) type antidepressants at the minimum effective dose (fluoxetine 20 mg/day, paroxetine 20 mg/day, sertraline 50 mg/day, citalopram 20 mg/day or escitalopram 10 mg/day, consistent with other studies21,22), empirically prescribed according to the clinical judgement of the treating psychiatrist (W.H.K. and Y.C.Y.). At 2 weeks after initiation of treatment, patients were classified as early treatment-responsive (ERD; reduction in HAMD score of ≥20% compared with baseline) or early treatment-nonresponsive (END), according to changes in depression severity. 5 Decrease ratio of HAMD score was calculated as (baseline score – 2 week score)/baseline score.
Statistical analyses
Data were presented as mean ± SD or n. Differences in demographic characteristics between patients and controls were analysed using one-way analysis of variance (ANOVA) or χ 2 -test, with P < 0.05 considered statistically significant. Data were analysed using SPSS® version 17.0 (SPSS Inc., Chicago, IL, USA) for Windows®.
Differences in ALFF between the three study groups (END, ERD and control) were determined by ANOVA at each voxel, followed by post hoc t-test to identify differences between each pair of groups. For ALFF comparison, a combined threshold of P < 0.001 and a minimum cluster size of 540 mm3 was used, resulting in a corrected threshold of P < 0.05 (determined by AlphaSim software; http://afni.nih.gov/afni/docpdf/AlphaSim.pdf). Local cluster peaks were identified in T-value maps resulting from comparing ALFF between patient groups and controls (with sex and age as covariants), using the MarsBaR toolbox (version 0.42) (http://marsbar.sourceforge.net/) to gain the mean value in each cluster. Relationships between aberrant brain activity and clinical symptoms and illness duration in patient groups were evaluated with Spearman’s correlation coefficient analysis, with P < 0.05 considered statistically significant.
Results
Demographic and clinical characteristics of drug-naïve patients with first-episode major depressive disorder (MDD) and healthy control subjects, included in a study to investigate resting state brain activation and short-term antidepressant treatment outcome.

Data presented as n or mean ± SD.
χ2-test.
One-way analysis of variance.
Two-sample t-test.
(baseline score – 2-week score)/baseline score.
END, early treatment-nonresponsive MDD; ERD, early treatment-responsive MDD; NS, not statistically significant (P ≥ 0.05); BMI, body mass index;HAMD-17, Hamilton depression rating scale 18 .
Data regarding region-specific differences in ALFF between patients with END or ERD and healthy controls are shown in Table 2 and Figure 1. Patients with ERD and those with END both exhibited several differences in ALFF, compared with control subjects, and there were also several differences between the ERD and END groups.
Functional magnetic resonance images showing brain activation in drug-naïve patients with first-episode major depressive disorder (MDD) and healthy control subjects. Patients were classified as having early treatment-nonresponsive (END) or early treatment-responsive (ERD) MDD. R, right; L, left; circles, increased brain activation regions; squares, decreased brain activation regions; colour bars indicate T-values from post hoc analysis between each pair of groups. Values at the left top of each image refer to z co-ordinates (mm) in Montreal Neurological Institute space. (a) END vs ERD; (b) ERD vs control; (c) END vs control. The colour version of this figure is available at: http://imr.sagepub.com. Region-specific differences in amplitude of functional magnetic resonance imaging low-frequency fluctuation (ALFF) between healthy control subjects (n = 33) and pretreatment drug-naïve patients with first-episode major depressive disorder (MDD), stratified into early treatment-nonresponsive (END; n = 30) and early treatment-responsive (ERD; n = 26) groups. Co-ordinates of primary peak locations in the Montreal Neurological Institute space. T-statistical value of peak voxel showing ALFF differences among three groups. Data corrected for multiple comparisons with AlphaSim correction. BA, Brodmann area.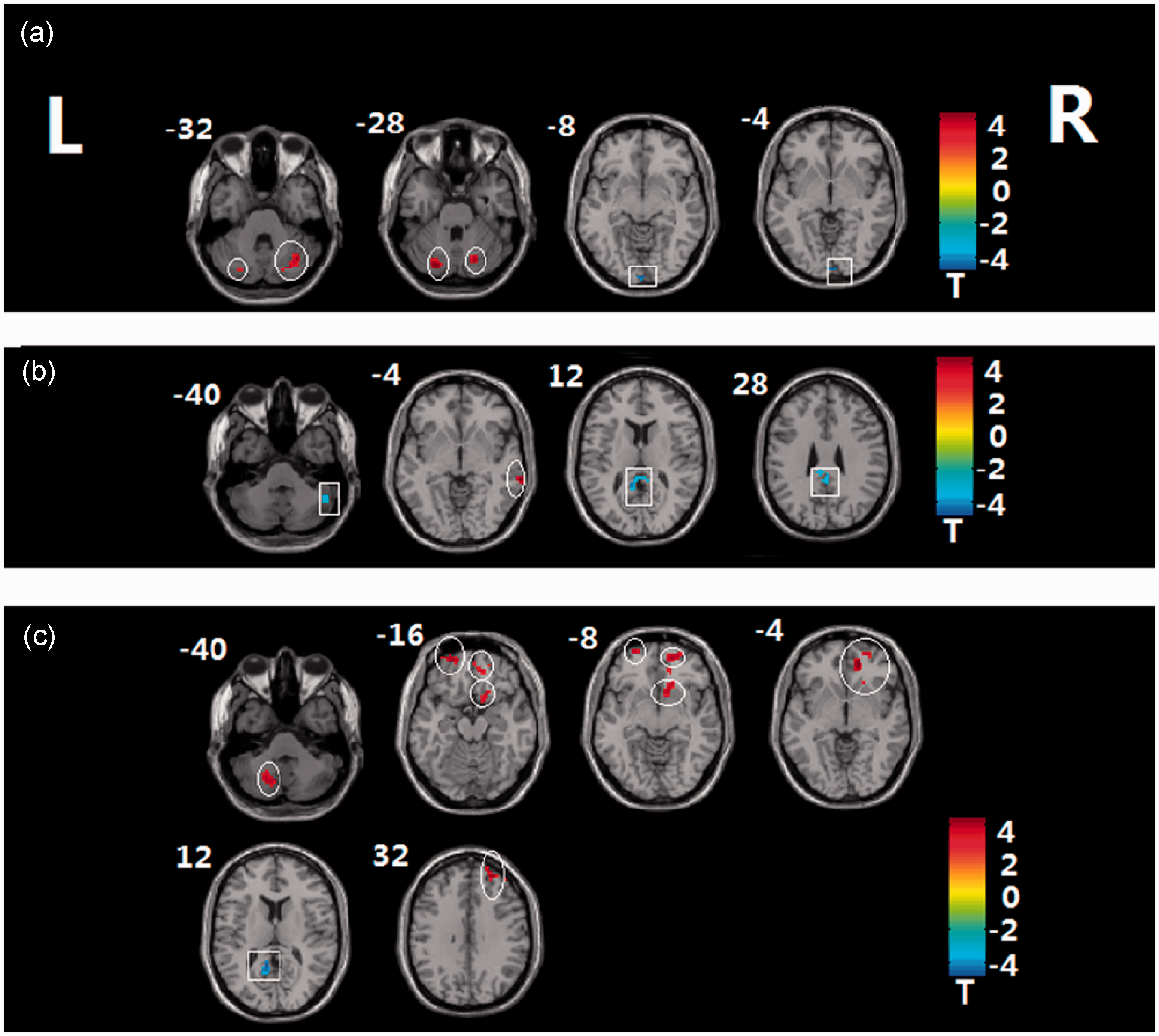

In patients with ERD, disease duration was negatively correlated with mean ALFF of the right lingual gyrus (r = −0.460, P = 0.018) and positively correlated with mean ALFF of the left cerebellum posterior lobe (r = 0.432, P = 0.028). In patients with END there was a positive correlation between decrease ratio of HAMD score and mean ALFF of the right lingual gyrus (r = 0.383, P = 0.037) and of the left cerebellum posterior lobe (r = 0.398, P = 0.046).
Discussion
The use of ALFF to characterize changes in brain activation regions in patients with first-episode MDD could assist in the development of effective early therapy. Altered brain function was found in the right lingual gyrus and left cerebellum posterior lobe in the present study, and these changes correlated with disease duration and short-term changes in symptom severity in patients with ERD and END, respectively.
The occipital lobe lingual gyrus is part of the visual recognition circuit that plays a role in the perception of facial emotion 23 and is crucial for social functioning, possibly affecting individual emotion. Structural and functional studies of patients with MDD have found decreased grey matter concentrations in the bilateral lingual gyrus. 24 Evidence from first-episode, drug-naïve patients with MDD has demonstrated decreased nodal centralities 25 and white-matter integrity 26 related to occipital regions. These patients exhibit impaired interhemispheric co-ordination in visual recognition regions 27 and decreased bilateral lingual gyrus connectivity with frontal regions. 28 A resting-state fMRI study revealed that abnormal activity of the occipital cortex was involved in the pathophysiology of remitted geriatric depression. 29 In addition, imaging studies have identified reductions in occipital cortex γ-aminobutyric acid (GABA) levels in patients with MDD.30,31 We hypothesize that patients with END may have a greater imbalance in concentrations of GABA than those with ERD, resulting in a poor response to SSRI antidepressants. There were no differences between either of the patient groups and the control subjects in ALFF of the lingual gyrus, in the present study, but there was a difference between patients with ERD and those with END. This is in accordance with other neuroimaging studies, suggesting that functional differences may be present specifically in patients rather than in healthy controls.32,15 Studies comparing patients and healthy subjects naturally reflect the differences related to pathological changes, and it remains unclear as to whether these brain regions may represent new treatment targets. A cross-sectional and longitudinal study reported that the ALFF approach for identifying abnormal brain regions was inconsistent. 32 It is therefore not unexpected that some brain regions may or many not change following treatment. We hypothesize that the lingual gyrus may be a targeted treatment area, but further longitudinal studies are required to establish whether it is related to treatment response. The findings of the present study provide direct evidence that function in this particular brain region may be related to treatment response.
A positive correlation was found between lingual gyrus activity and short-term change in symptom severity in patients with END in the present study, suggesting that this region may require long-term treatment. In patients with ERD, however, brain activity changes of the lingual gyrus were negatively related to disease duration in the current study, suggesting that the duration of the illness itself was responsible for the impairment of this brain region.
Studies have suggested that the cerebellum is important in MDD, 33 although its role in emotion, affect and cognitive processes is not fully understood. 34 Neuroimaging studies of the cerebellum in MDD have found morphological alterations 35 and functional changes during both rest-36,37 and task-related fMRI.3338 Others have found increased/decreased regional homogeneity in large areas of the cerebellar region in refractory MDD, in addition to in patients with MDD 40 and their first-degree relatives. 41 In the present study, reduced right cerebellum activity was related to changes in clinical symptom severity in patients with END, and to disease duration in patients with ERD. Evidence suggests that the pathophysiology of MDD may not be attributable to monoaminergic system dysfunction alone, 42 but also to the glutamatergic system. While high densities of glutamate neurons reside in the cerebellum, 43 studies have demonstrated that glutamate and NMDA receptor activation may cause cerebellar dysfunction. 44 Our findings support the involvement of the cerebellum in MDD, providing additional evidence regarding the involvement of this region and the glutamatergic system in the pathophysiology of MDD and END. The poor response to SSRI antidepressants in patients with END might stem from glutamatergic system dysfunction.
The ALFF of the posterior cingulate cortex was decreased (relative to controls) in patients with ERD and those with END, in the present study. The posterior cingulate cortex is part of the default mode network (DMN), which is important for affective processing. 45 Brain imaging studies have identified abnormal changes in the DMN in patients with MDD. 46 The present findings, therefore, confirmed the importance of this region in disease pathology unrelated to treatment responsiveness.
The present study has some limitations. First, the image data are cross sectional, and longitudinal studies are required to establish whether these altered neural activities change dynamically after therapy. Secondly, patients with MDD were prescribed the minimum effective dose of antidepressants, which may not have been sufficient for all patients. Further studies should select a higher dose and drugs with a fast onset of action, in order to differentiate accurately between END and ERD groups.
In conclusion, we identified brain regions related to the neural mechanism of MDD early treatment outcome. These regions may have important implications for the treatment of MDD and SSRI use, in particular.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported by National Natural Science Foundation of China (Grants No. 81171488, 81030027 and 30900362) and National Basic Research Program of China (973 Program No: 2007CB512302/5).
Acknowledgements
We wish to thank Professor Qi-Yong Gong and Professor Xiao-Qi Huang for assistance and guidance with this article. We thank Lei Li, Ya Luo, Xiu-Li Wang, Xun Yang, Li-Zhou Chen and Ming-Yin Du for help with participant recruitment, and Tao-lin Chen and Li Yao for their assistance with data analysis in the Huaxi MR Research Centre (HMRRC), Department of Radiology, West China Hospital of Sichuan University.
